3.1.4a - General Properties of Proteins
Thursday 28th September ‘23
Functions
Enzymes: catalyse biological processes. eg pepsin.
Hormones: regulate body processes. eg insulin.
Storage proteins: store nutrients. eg ferritin.
Transport proteins: transport oxygen and other substances through the body. eg haemoglobin.
Structural proteins: form an organism’s structure. eg collagen.
Protective proteins: help fight infection. eg antibodies.
Contractile proteins: form muscles. eg actin, myosin.
Toxic proteins: serve as a defence for the plant or animal. eg snake venoms.
Amino Acids
These are the monomers which make up proteins. There are 20 different amino acids present in all living organisms. They are composed of an amine group, carboxyllic acid group and R group (and a hydrogen).
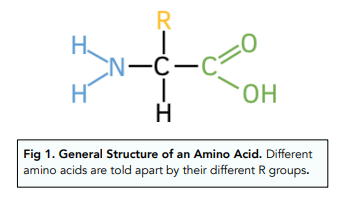 These form proteins through a condensation reaction where a peptide bond forms between the amine group of one amino acid and the hydroxyl group of another.
These form proteins through a condensation reaction where a peptide bond forms between the amine group of one amino acid and the hydroxyl group of another.
Structural levels
Primary - the number and order of amino acids in the polypeptide chain.
Secondary - initial shape the chain form (local folding). Hydrogen bonds form between amino acids in the chain causing it to either coil into an alpha helix or fold into a beta pleated sheet.
Tertiary - overall 3D shape of the protein. Types of bonds involved: hydrogen, ionic (between any carboxyl and amine group not involced in a peptide bond), disulfide bridges (strong, occur between sulphur atoms)
Quaternary - shows how the individual subunits (the polypeptide chains which form a large molecule) are arranged to form complex protein molecules. This also shows prosthetic groups (which make it become classed as a conjugated protein).
Globular proteins - spherical shape; hydrophobic centre with a hydrophilic outer; soluble due to charged R groups on outer surface allowing water to surround them; examples: haemoglobin, insulin, lysozyme.
Fibrous proteins - long, rope-like chains; made of a series of straight-chain polypeptides, which lie side-to-side and wrap round one another, held in place by hydrogen bonding; insoluble and presence of lots of disulphide bridges makes them relatively resistant to physical and chemical attack.