Lecture Notes on Completing Oxidation of Glucose and Anabolism
Completing Oxidation of Glucose/Other Carbohydrates/Anabolism from Catabolic Products
Products of Cellular Respiration after Glycolysis
If a final electron acceptor is present, the complete oxidation of pyruvate occurs in the matrix of the mitochondria.
Captured electrons are used to create a proton gradient.
Before further oxidation, pyruvate must cross two lipid bilayers into the matrix of the mitochondria.
Electrons captured from glycolysis may also enter the mitochondrial matrix.
Mitochondria Structure
In eukaryotes, pyruvate oxidation, the citric acid cycle, and oxidative phosphorylation take place in mitochondria.
Transport of Pyruvate and NADH
The outer membrane of mitochondria has nonselective channels (pores) which allow free access of pyruvate and NADH.
The inner membrane has specific transporters for pyruvate.
NADH does not pass through the membrane but donates electrons to a substrate which is transported through the inner membrane and reoxidized in the matrix of the mitochondria.
The electrons are donated to or .
Oxidation of Pyruvate to AcetylCoA
The reaction is an oxidative decarboxylation of pyruvate and attachment of the cofactor, CoA, to the remaining carbons of pyruvate.
It is a coupled reaction that utilizes the energy released by oxidation (exergonic) to the attachment of CoA to the remaining two-carbon fragment (acetyl) of pyruvate (endergonic).
There was a similar coupling by triose phosphate dehydrogenase in glycolysis sans decarboxylation.
There are two similar reactions in the citric acid cycle.
All these steps are accomplished by a huge multienzyme complex, pyruvate dehydrogenase, which contains the vitamins thiamine and lipoic acid.
Acetyl CoA Synthesis
Mechanism of Pyruvate Dehydrogenase
Structure of Acetyl-CoA
Oxidized carboxyl (COOH) group is removed and released as CO2 (decarboxylation)
The remain 2 carbon fragments is oxidized to acetate and extract electrons are passsed to NAD+ (redox)
CoA is attached to the acetate by a hight energy bond ( activation using redox energy)
Products of Cellular Respiration after Pyruvate Oxidation
The citric acid cycle (CAC) has eight steps, each catalyzed by a specific enzyme.
The acetyl group (2 carbons) of acetyl CoA joins the cycle by combining with oxaloacetate (4 carbons), forming citrate (6 carbons) catalyzed by citrate synthase.
The next seven steps oxidatively decompose the citrate back to oxaloacetate, making the process a cycle with the loss of two carbons per acetyl group.
The four carbons added to the CAC from 2 acetyl are released as completing the oxidation of the original glucose.
Thermodynamics of the Reactions of the Citric Acid Cycle
Products of Cellular Respiration after Citric Acid Cycle
Oxidative Phosphorylation
The creation of a proton () gradient by the electron transport chain.
The energy in the proton gradient is utilized to drive ATP synthesis as the proton returns into the mitochondrial matrix through the ATP synthase complex.
Other Carbohydrates
Polysaccharides (Macromolecules)
~100 or more monosaccharides covalently joined by dehydration reactions
Common polysaccharides are amylose, glycogen, and cellulose. All these are polymers of glucose that differ in source or bonding difference between monosaccharides.
Disaccharides
Compounds containing 2 monosaccharides covalently joined by dehydration reactions
Common disaccharides are sucrose, maltose, and lactose
Polysaccharides (“many sugars”)
Macromolecules formed by the polymerization of many monosaccharide subunits (monomers)
Two common energy storage polysaccharides:
Starch and glycogen
Two common structural polysaccharides:
Cellulose and chitin
Storage Polymers of Glucose
If glucose is not required by a cell it can be stored as the polysaccharides glycogen in animals or amylose and amylopectin in plants.
The bonds covalently linking the glucose monomer are termed glycosidic bonds.
The glycosidic bonds in all of these polysaccharides are always (1->4) and in amylopectin and glycogen (1->6).
The amylose can be hydrolyzed to glucose monomers by amylase.
The glucose monomers from glycogen are accessed by phosphorolysis to give glucose-1-phosphate. A mutase converts G-1-P to G-6-P.
Monosaccharide Anomeric Isomers
Monosaccharides with five or more carbons can change from the linear form to a ring form
Storage Polysaccharides I
Glycogen is made by animals to store energy, usually in liver and muscle tissues.
Highly branched
Storage Polysaccharides II
Starch is made by plants to store energy
Amylose = linear, unbranched
Amylopectin = branched
Structural Polymers of Glucose
Glucose polymers can be used for structural purposes in cellulose and chitin.
The glycosidic bonds in all of these polysaccharides are always b (1->4).
In chitin, the 2’ position contains an aminoacetyl group.
These b linkages are highly resistant to hydrolysis and very few organisms have the required enzymes to catalyze the reaction.
Structural Polysaccharides
Cellulose is made by plants as a structural fiber in cell walls
Unbranched chain of glucoses connected by -linkages
Extremely strong
Structural Polysaccharides II
Chitin is tough and resilient, used for cell walls of fungi and exoskeletons of arthropods
Similar structure to cellulose, but glucose sub-units modified with nitrogen-containing groups
Enter of Fructose and Galactose into Glycolysis
Fructose can be phosphorylated to fructose-6-phosphate and enter glycolysis.
Galactose is converted to glucose in a reaction that requires an NTP.
Other Monosaccharides in Glycolysis
The Versatility of Catabolism
Catabolic pathways funnel electrons from many kinds of organic molecules into cellular respiration
Glycolysis accepts a wide range of carbohydrates
Proteins must be digested to amino acids; amino groups removed, and the remainder can feed glycolysis or the citric acid cycle
Fats are digested to glycerol (used in glycolysis) and fatty acids (used in generating acetyl CoA)
An oxidized gram of fat produces more than twice as much ATP as an oxidized gram of carbohydrate
Cellular Respiration can have Alternate Sources of Electron Donors and Electron Acceptors
Chemolithotrophs: The electron donors is an inorganic molecules (e.g., H2S, H2, NH4).
Anaerobic Respiration: The final electron acceptor may be either inorganic molecules (e.g. Fe3+, CO2, SO4 -)or organic molecules (i.e. trimethylamine N-oxide, fumarate).
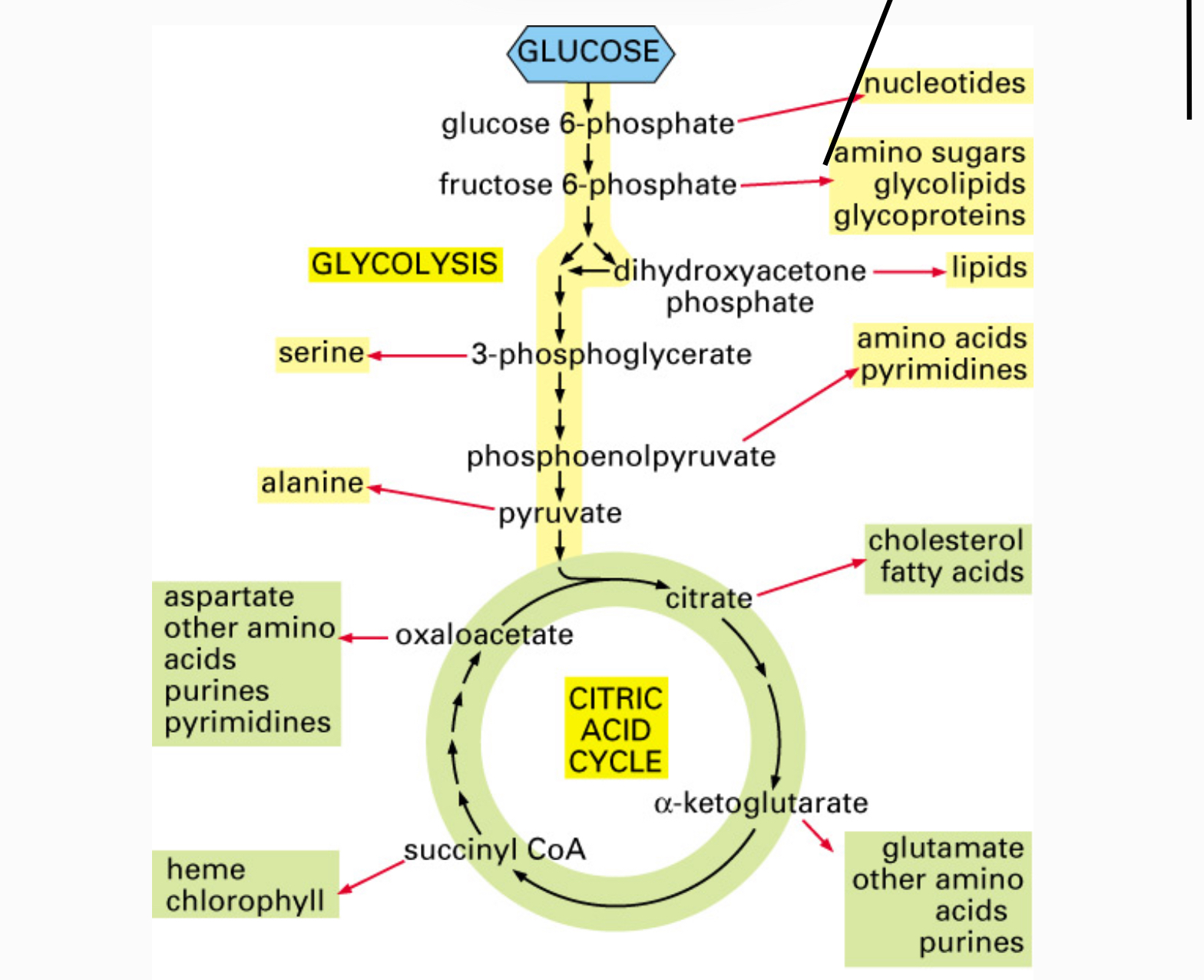
Biosynthesis (Anabolic Pathways)
The body uses small molecules to build other substances
These small molecules may come directly from food and cannot be made by the organism (i.e vitamins, essential nutrients)
Many precursors for biosynthesis are intermediates from cellular respiration
Anabolic Precursors from Catabolism
Anabolic Products from Catabolic Products: DHAP can be Reduced to form Glycerol 3 –phosphate
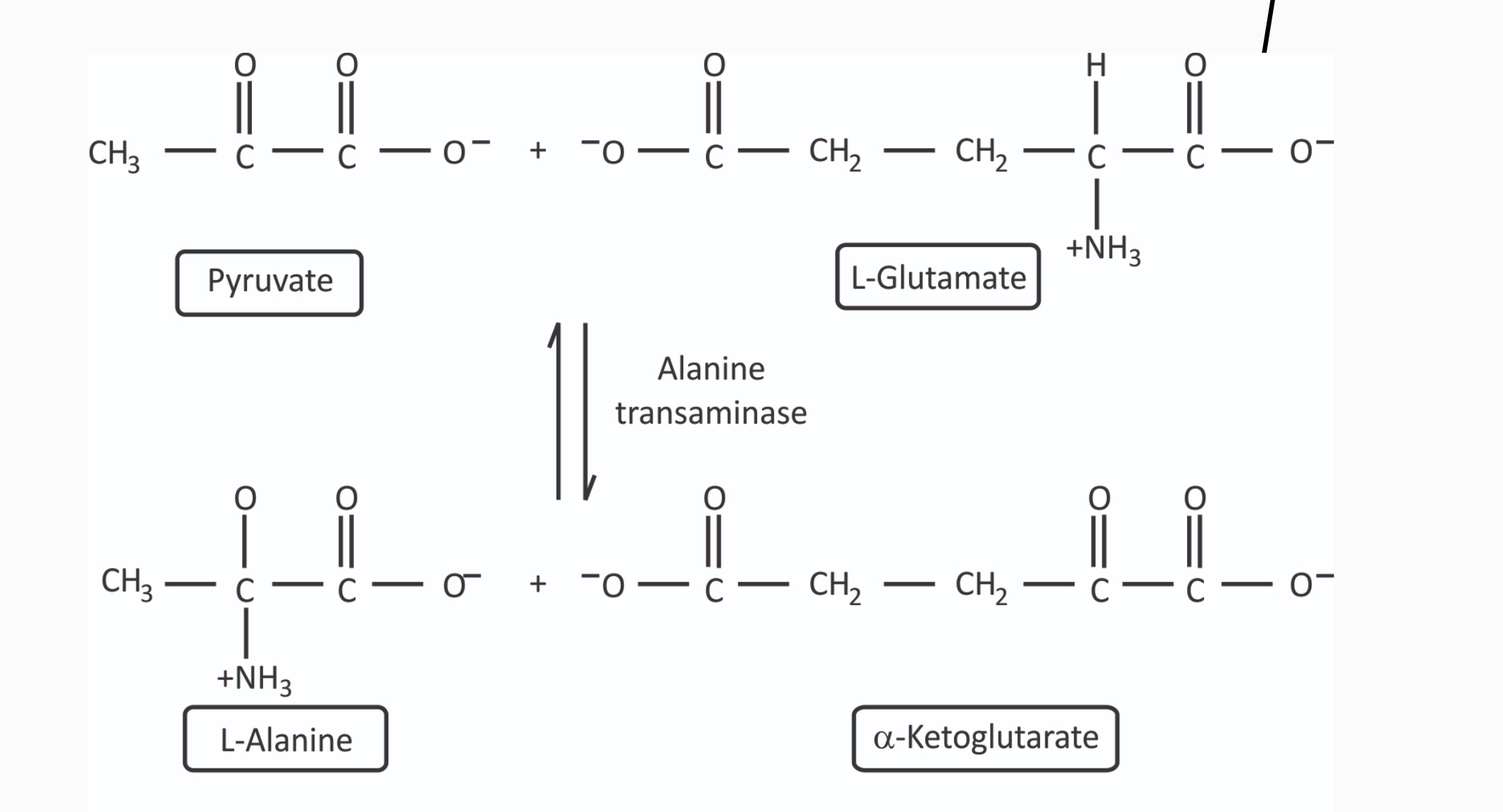
Anabolic Products from Catabolic Products: Pyruvate Amidated to form L-alanine