12-04: Nucleic Acids
Nucleic Acids
- Nucleic acids: molecules that are polymers made of nucleotides * When we link many nucleotides together, we get a nucleic acid
- They store hereditary information and can produce identical copies of themselves
1. DNA, deoxyribonucleic acid: double stranded 2. RNA, ribonucleic acid: single stranded
\
Nucleotides
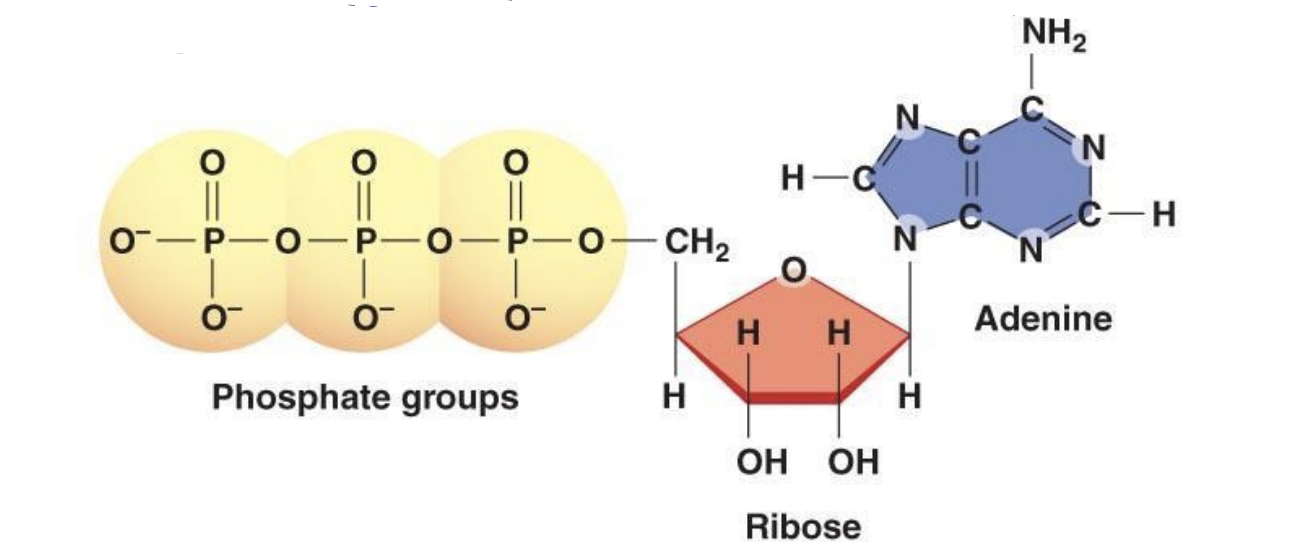
- Nucleotides are made of 3 components (in a single nucleotide)
1. Pentose * 5 carbon sugar * Either a ribose sugar in RNA or deoxyribose sugar in DNA 2. Phosphate group * Bonded to C5 with a phosphate ester linkage 3. Nitrogenous base - 1 of 4:
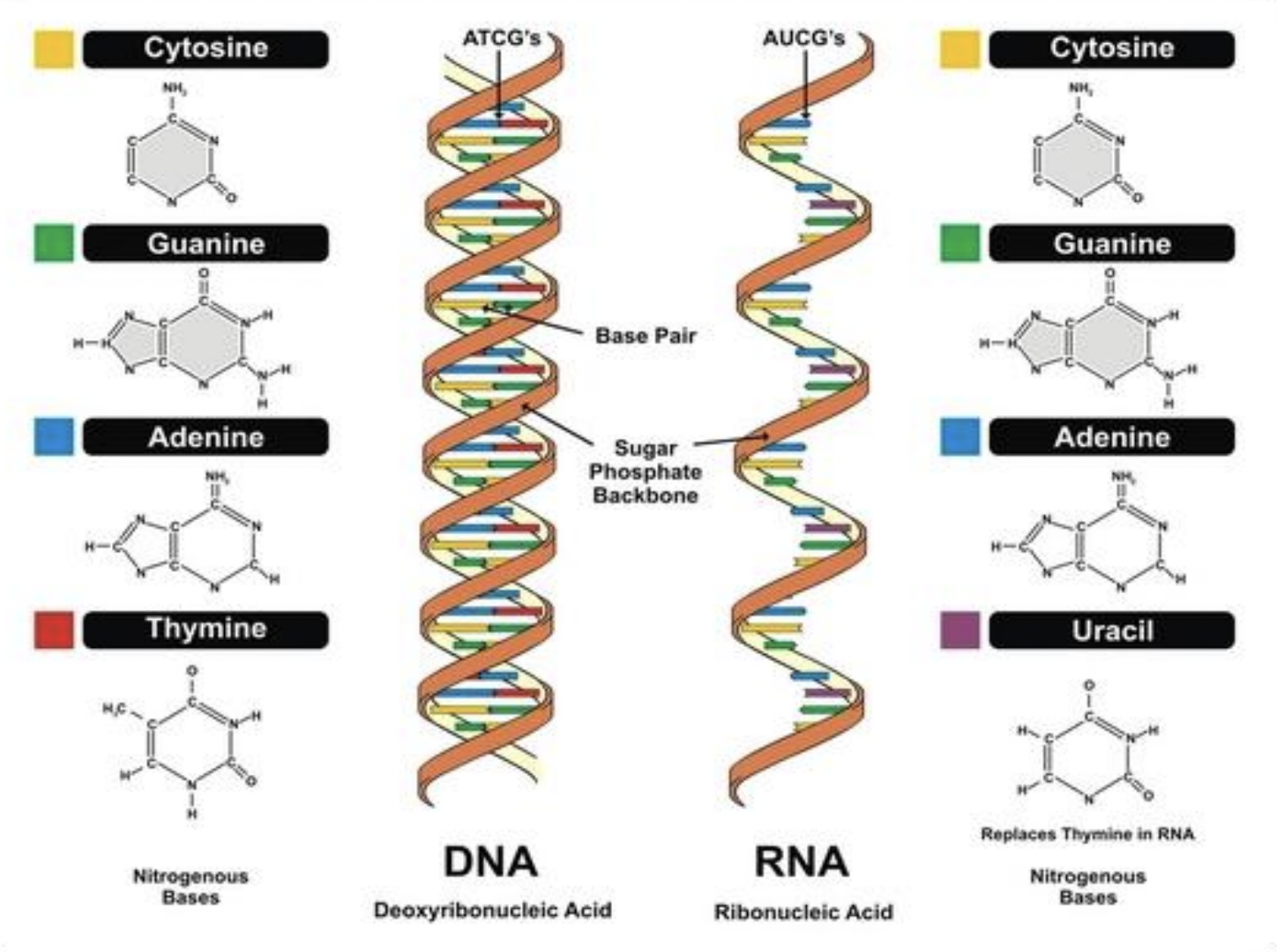
1. Adenine (A) 2. Guanine (G) 3. Cytosine (C) 4. Thymine (T) in DNA, or Uracil (U) in RNA * a. b. and c. are possessed by both RNA and DNA
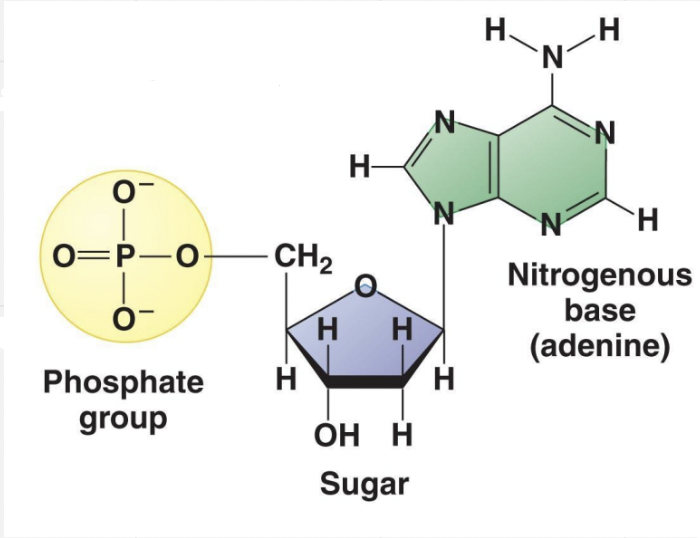
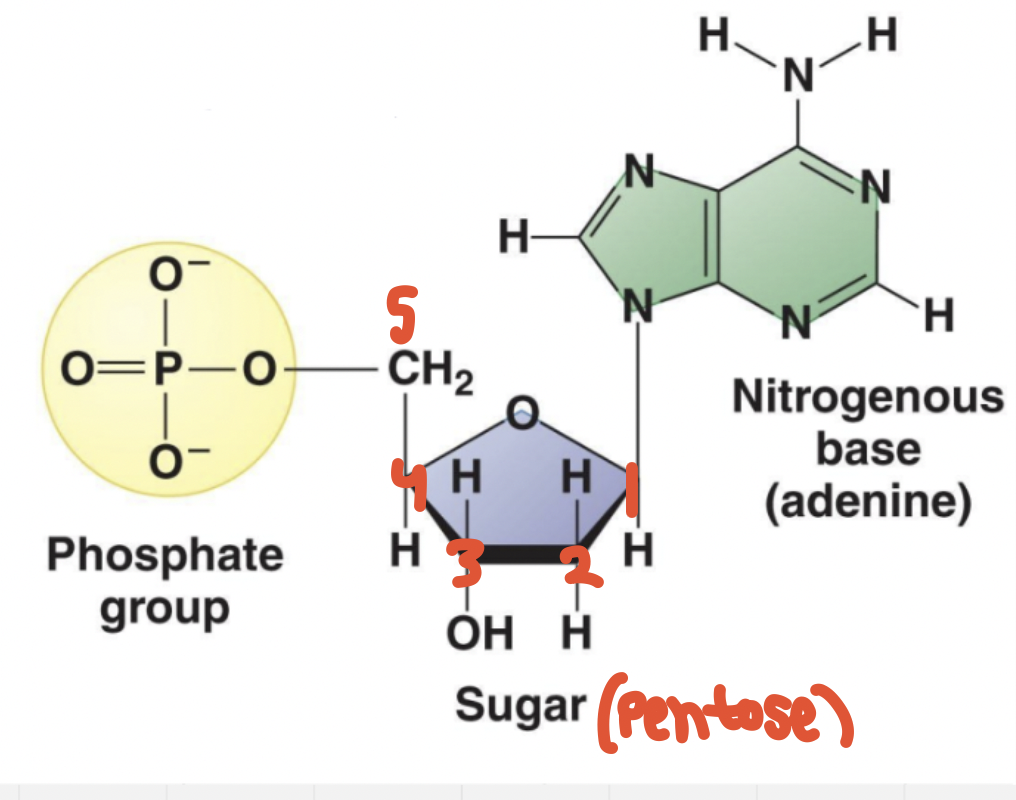
Phosphates can have OH’s, but in an aqueous environment, H and O ionize into H⁺ and O⁻ as H⁺ dissociates
\
Formation:
- The sugar from one carbon and the phosphate from another carbon form a phosphodiester bonds to make a long backbone
- The phosphate has 2 ester linkages to 2 different carbons: C5 on the original/first sugar to C3 on the new/second sugar
- Covalent bonds between the phosphate group on nucleotide on the hydroxyl of the C3 hydroxyl of a sugar on the nucleotide
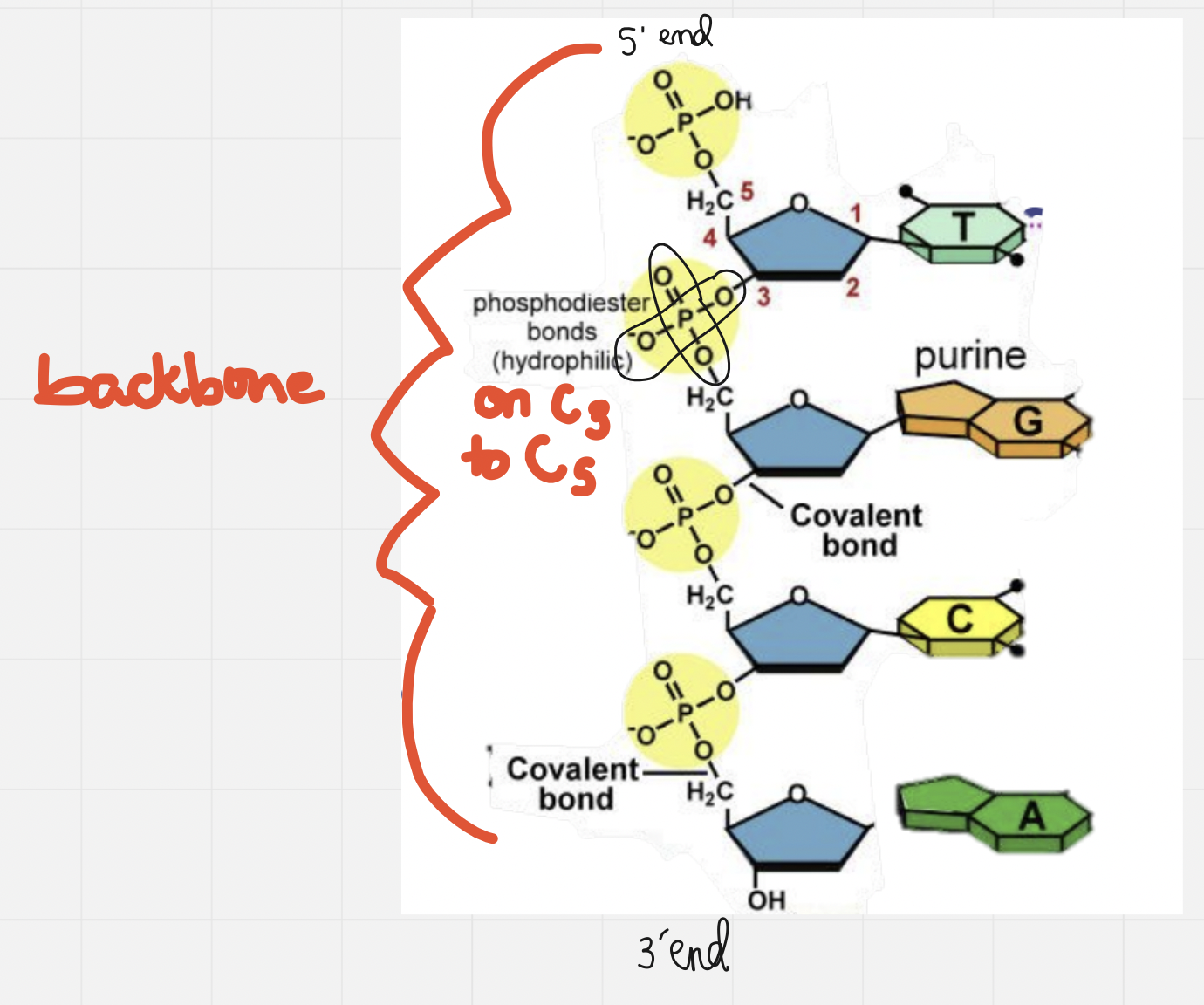
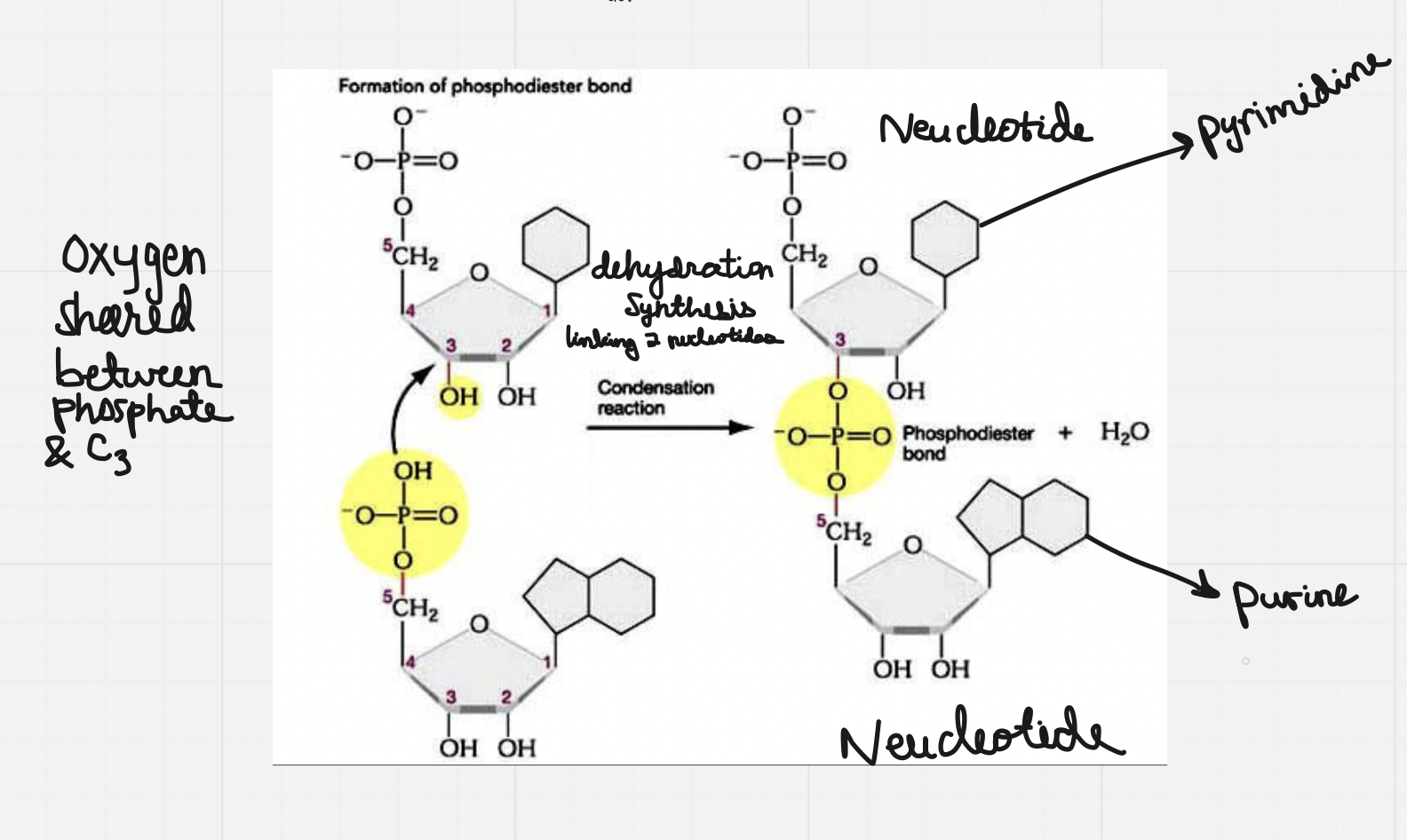
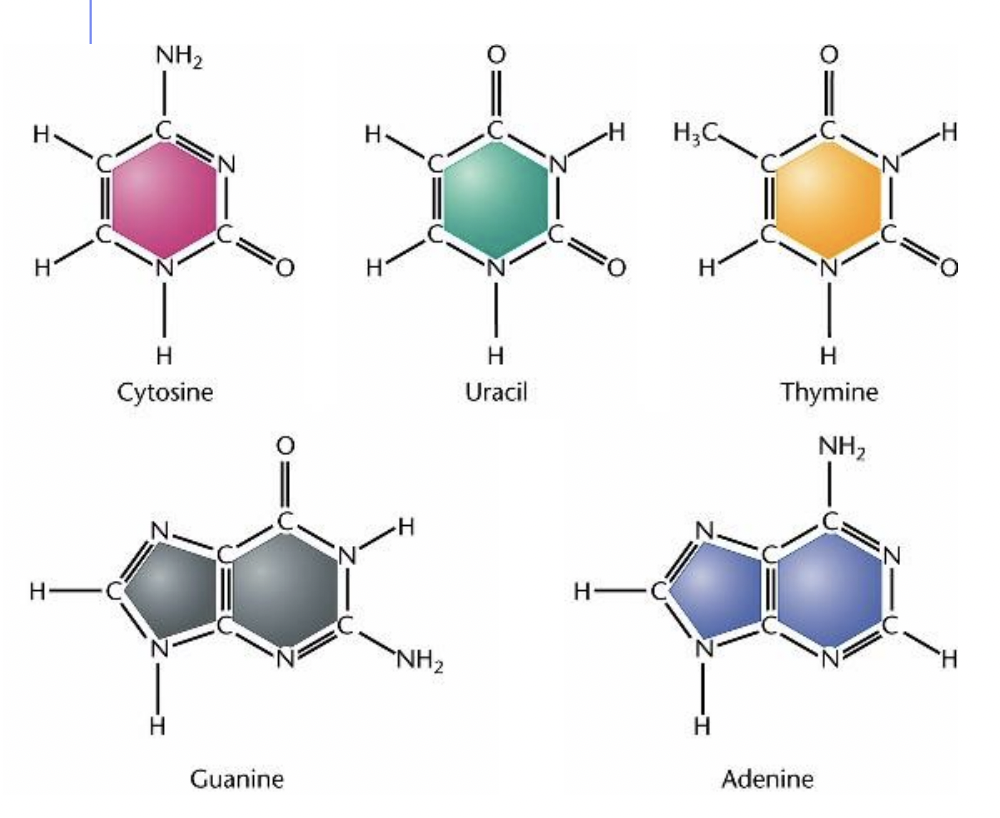
Nitrogenous bases
- Nitrogenous bases point towards the inside of the polymer, away from the backbone
- Two types of nitrogenous bases
1. Pyrimidines * Single ringed * Cytosine (C), Uracil (U), Thymine (T) * CUT are the pyrimidines since they are a double ringed “CUT” in half! 2. Purines * Double ringed * Adenine (A) and Guanine (G)
\
DNA
- 2 strands of nucleotides together – bigger structure, ladder that twists into a helix
- Double stranded
- Bases point towards each other
- Cytosine (C) and Guanine (G) are complementary base pairs and join together with 3 hydrogen bonds (bonded to the N)
- Adenine (A) and Thymine (T) are complementary base pairs and join together with 2 hydrogen bonds
- Each strand of DNA has a free phosphate at one end and a free sugar at the other – one strand runs backwards relative to the other strand * They run “anti-parallel” (parallel but in opposite directions)
\
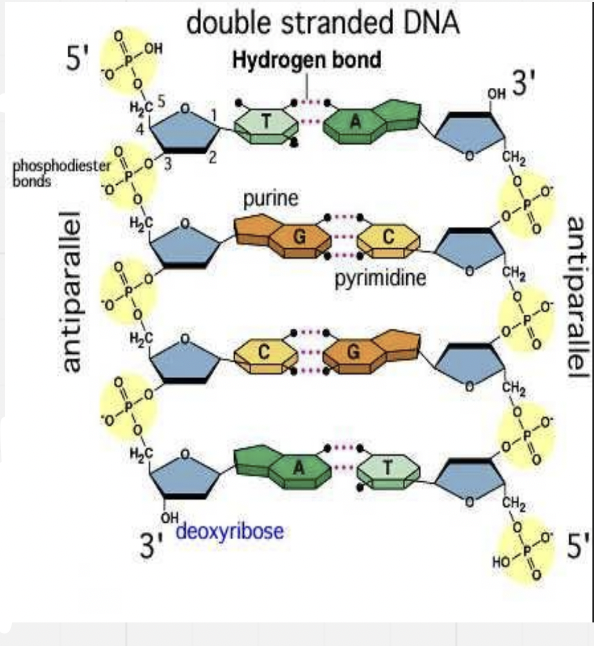 Must be antiparallel in order for the H bonds to occur
Must be antiparallel in order for the H bonds to occur
\
- Coils into a double helix * Twisted ladder
- Phosphate sugar backbone around the outside and bases point towards the inside
\
RNA
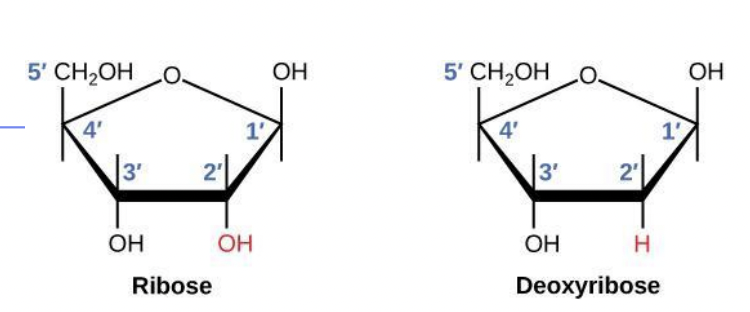 Difference is on C2
Difference is on C2
RNA has many different functions (e.g. mRNA (Messenger RNA), tRNA, rRNA)
\
Comparing DNA and RNA
| DNA | RNA | |
|---|---|---|
| Sugar | Deoxyribose | Ribose |
| Bases | A, G, T, and C | A, G, U, and C |
| Strands | Double stranded | Single stranded (but can be double stranded if needed) |
\
\
\
Other nucleotides
ATP
- ATP: adenosine triphosphate * Has all of the features of a nucleotide but rather than 1 phosphate, it has a triphosphate (3)
- Adenine and ribose + 3 phosphates
- Cells use it as a source of energy – mitochondria synthesizes ATP which is useful for other cellular chemical processes
- Used for energy transport within the cell
- The only format of energy out cells can use (“currency”) * The removal of 1 phosphate group releases energy to be used
\
Nucleotide Derivatives
- Derivatives of nucleotides are used in the production of ATP
- Used to transport H⁺ and electrons from one reaction site to another
e.g. * nicotinamide adenine dinucleotide (NAD+) * flavin adenine dinucleotide (FAD) * cyclic adenosine monophosphate (cAMP)
\ Bottom line: not every nucleotide has to be in DNA & RNA
\