Osmosis & Tonicity (Passive Transport)
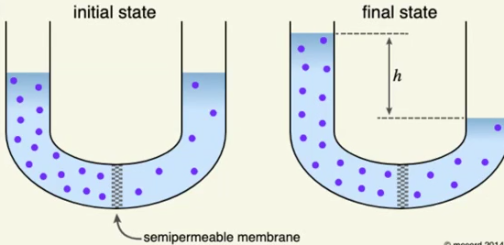
Osmosis: the diffusion of water across a semi-permeable membrane
Passive Transport
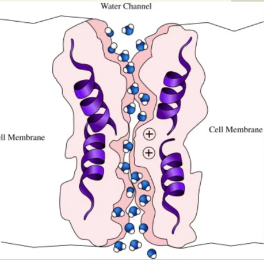
water is a small, uncharged, polar molecule ——> only a little bit can pass through the membrane (simple diffusion)
Aquaporins: channel proteins that are specific to water ——> allow for greater volumes of Osmosis to occur (facilitated diffusion)
Solution = solvent + solute
Solvent: does the dissolving
Solute: what’s being dissolved
NET direction of water movement in Osmosis: water moves from a low concentration of solute to a high concentration of solute
high concentration of solute = low concentration of solvent (water)
low solute concentration = high solvent concentration (water)
Tonicity: ability of a solution to make water move into or out of a cell by Osmosis
Isotonic: same solute concentration ——> A is isotonic to B if A has the same solute concentration as B
B is also isotonic to A
same solvent concentration too
NO NET MOVEMENT of water molecules (same solvent concentration ——> Dynamic Equilibrium)
Hypertonic: higher solute concentration ——> A is hypertonic to B if A has a higher solute concentration compared to B
B is hypotonic to A
NET MOVEMENT of water molecules into solution A
Hypotonic: lower solute concentration ——> A is hypotonic to B if A has a lower solute concentration compared to B
B is hypertonic to A
NET MOVEMENT of water molecules into solution B
Water always moves toward the HYPERtonic space
Cell Walls make plants behave differently than animal cells
Animal Cells:
In isotonic solution: behave normally ——> NO NET MOVEMENT of water, so the cell stays the same size
In hypotonic solutions: NET MOVEMENT of water into the cell ——> bc cell is hypertonic compared to the solution ——> cell will swell up/burst: lyse
In hypertonic solution: NET MOVEMENT of water out of the cell ——> bc cell is hypotonic compared to the solution ——> cell will shrink/shrivel: crenate
Plant Cells:
In isotonic solution: NO NET MOVEMENT of water (flaccid) ——> w/o net diffusion of water into the cell, it will start to wilt ——> bc there is no turgor pressure to keep it upright
In hypotonic solution: turgid (normal) bc water diffuses into the cell
-Turgor Pressure: pressure from cell swelling w/in the cell wall (cell membrane can’t spread bigger than the wall) ——> keeps plants upright/prevents wilting
In hypertonic solution: cells will plasmolyze ——> membrane shrivels up & pulls away from the cell wall as the cell loses water
Amoeba and Paramecium:
Amoeba & Paramecium are unicellular organisms that live in freshwater (hypotonic environments)
Water will diffuse into the cell from the freshwater/hypotonic environment —> -must have some way to remove excess water to avoid bursting
Contractile Vacuole: collects & expels water from the cell to maintain an internal osmotic balance
-relates to the Homeostasis process of life ——> maintaining internal water condition/stable water concentration
Medical Applications of Osmosis:
IV Fluids & organ transplant fluids must be isotonic to the human body cells to prevent damage to the patient/organ
Must be isotonic: we are animals & don’t have cell walls ——> our cells would be damaged in a hypertonic/hypotonic solution
in hyper: our cells would shrivel
in hypo: our cells would swell up & burst (lyse)