C9: Metals
Properties of metals

Metal + dilute acid → salt + hydrogen
metal + water (steam) → metal oxide + hydrogen
metal + (cold) water → metal hydroxide + hydrogen
Uses of metals
Aluminum in the manufacture of aircraft:
high strength-to-weight ratio
low density
resists corrosion
aluminum in overhead cables
good electrical conductivity
low density
ductile
aluminum in food containers:
corrosion resistant from acidic foods
non-toxic
copper in electrical wiring
good electrical conductivity
ductile
Alloys and their properties
alloys: mixtures of metal with other elements
E.g: brass - copper + zinc
stainless steel - iron + other elements (chormium, nickel & carbon)
Alloys can be harder, stronger & more useful than pure metal
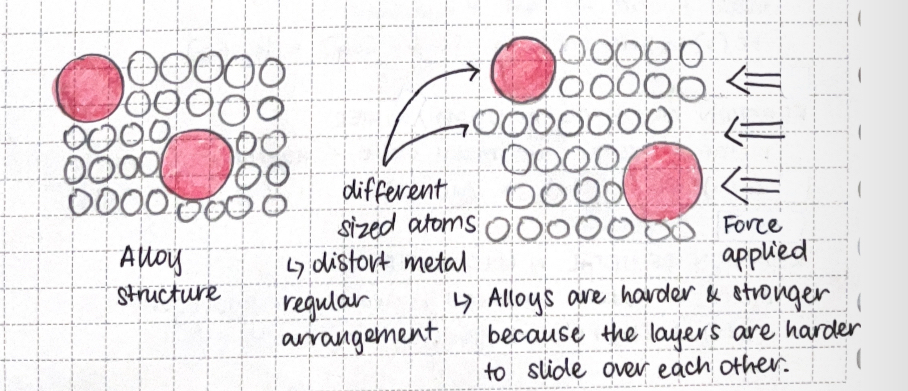
uses:
Stainless steel: cutlery due to its hardness & resistance to rusting
Reactivity series
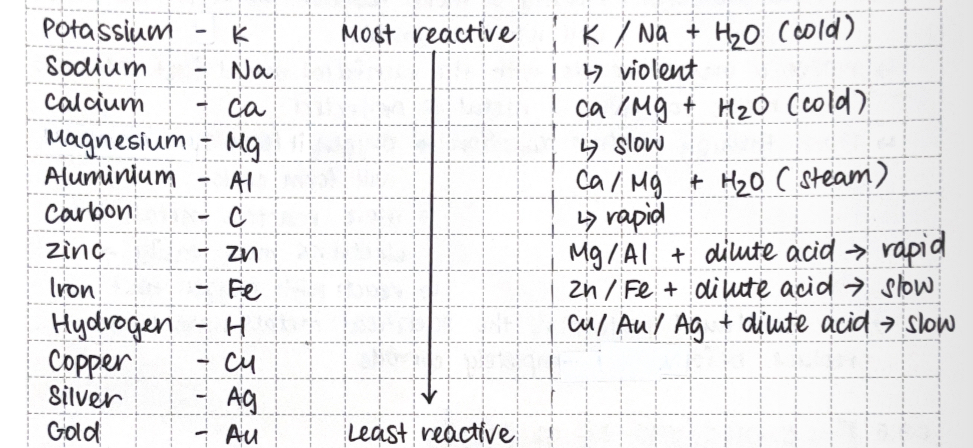
metals that lose electrons more readily to form cations → more reactive
displacement reaction: more reactive metals lose their electrons more easily
Corrosion of metals
corrosion: metals react with substances in their environment and are gradually destroyed
conditions for rusting: presence of oxygen & water
only happens on the surface
Barriers method:
painting, greasing & coating in plastic: prevents oxygen & water to come in contact with the metal
galvanizing steel: coating steel/iron with a thin layer of zinc as protection
sacrificial protection
if coating gets scratched → coating corrodes instead of iron/steel
sacrificial protection: placing a more reactive metal on the metal you want to protect
water + oxygen reacts with sacrificial metal first (more reactive)
works by electron donation:
oxygen + metal → cation
more reactive metals lose electrons more easily
reacts with oxygen first
needs to be replaced continuosly
Extraction of metals
more reactive metals are found in minerals as compunds called ors
high tendency to lose their electrons & form cations
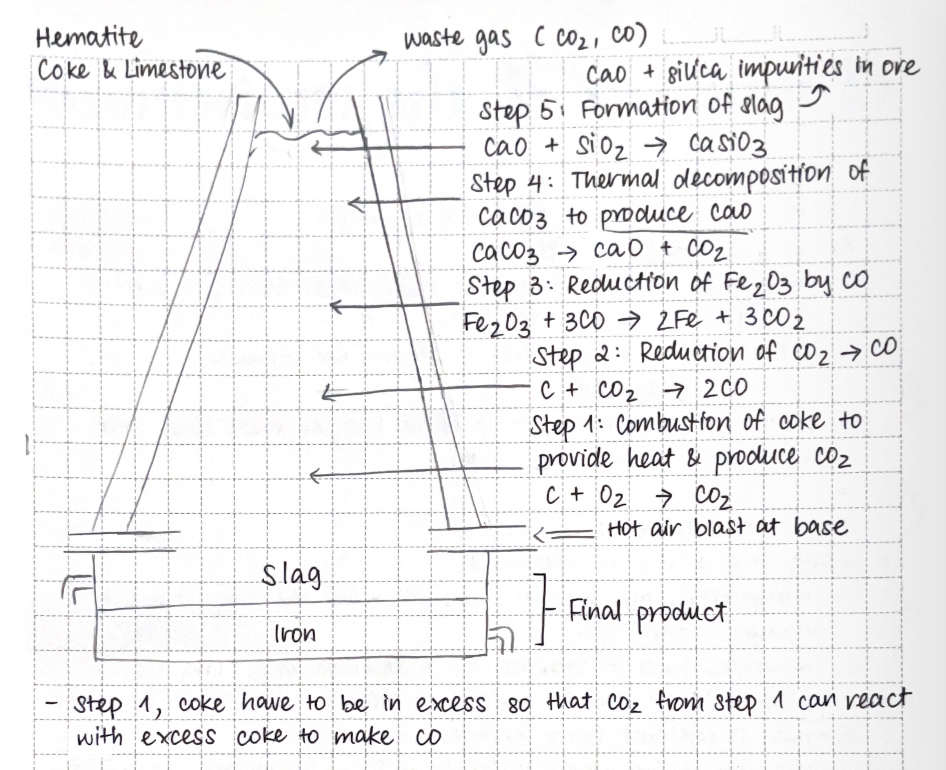
Extraction of iron:
hematite: ore containing iron oxide Fe2O3
iron is extracted from hematite by reduction of iron oxide in the blast furnace
raw materials:
hematite
coke (carbon): fuel and reducing agent
limestone (CaCO3): removes impurities as slag
hot air: supplies oxygen for combustion
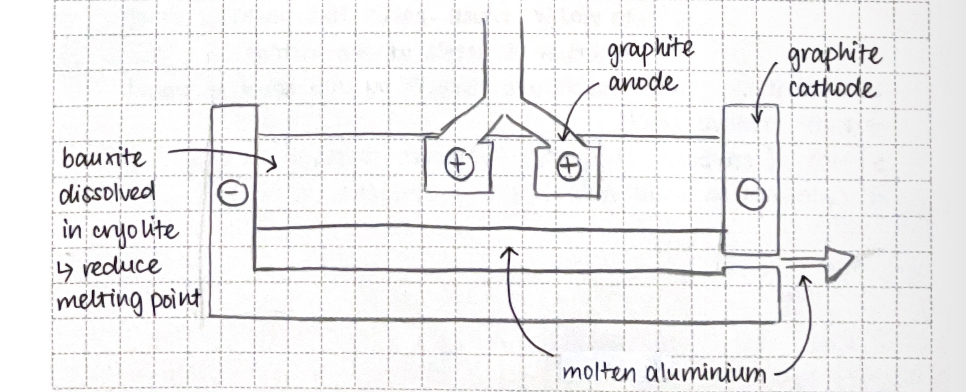
Fe2O3 dissolves in molten cryolite to reduce melting point
metals higher up in reactivity series are harder to extract from ore due to its stronger bond with its oxide
Extraction of aluminum:
bauxite
extracted by electrolysis
→ aluminum is higher in the reactivity series than carbon (cannot be extracted by the reduction of carbon )