11. IR spectroscopy
Infrared spectroscopy is a technique that exploits the vibration of the covalent bonds that join 2 atoms together to reveal how atoms within a molecule are connected. Vibrations are split into 2 categories; stretching and bending
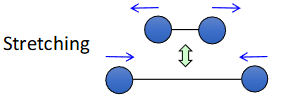
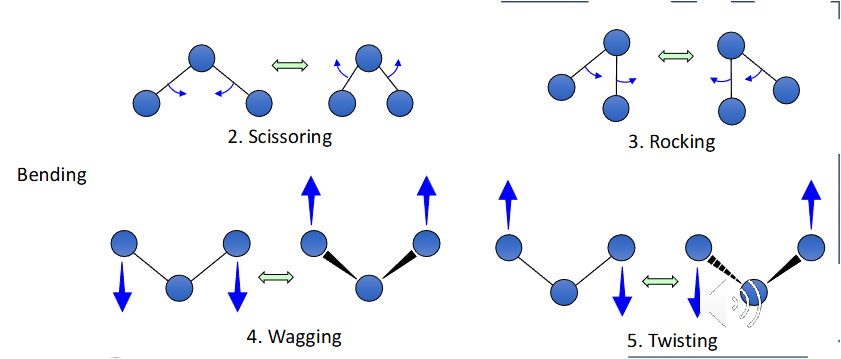
Factors affecting the speed of bond vibration
The strength of the bond joining the 2 atoms
Covalent bonds vibrate with a frequency proportional to their strength
The combined mass of the joined atoms
Covalent bonds vibrate with a frequency inversely proportional to their combined mass
Interpreting an IR spectra
When a compound is irradiated with infrared radiation, the bonds absorb the IR, providing the energy for bond vibration. Low frequency bond vibration will absorb longer wavelengths while high frequency bond vibration will absorb shorter wavelengths. With the use of a detector the peaks in absorbance (troughs in transmittance) can be measured. On the x-axis is wavenumber (cm-1) which is the reciprocal of wavelength
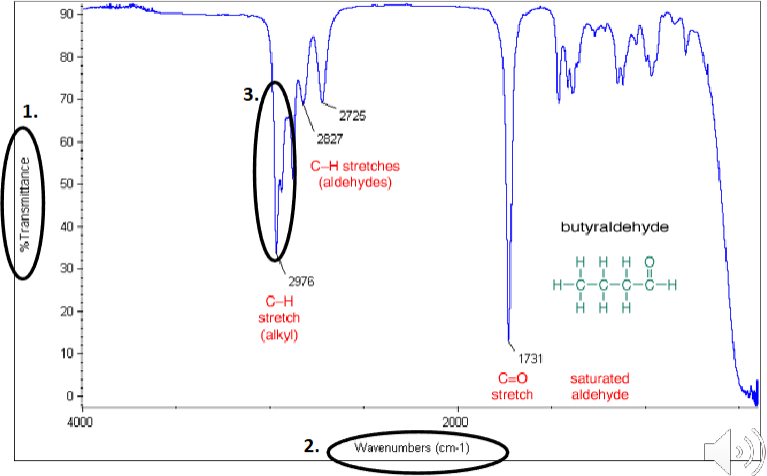
Different functional groups give rise to different peaks. However, bending and stretching vibrations occur at different frequencies. A bonding may exhibit both low energy bending and high energy stretching. Stretching peaks manifest as a peak at a slightly shorter wavelength. The wavelength at which a specific bond absorbs IR light is also dependent on its chemical environment. For example, the O-H bond in an alcohol absorbs different wavelengths than the O-H bond in a carboxylic acid