W12: Visual and non-Visual Melanopsin Photoreception
Melanopsin Photoreception Notes
Learning Outcomes
Understand the neurophysiological characteristics of melanopsin-expressing intrinsically photosensitive Retinal Ganglion Cells (ipRGCs).
Learn about ipRGC control of non-visual processes, including:
Isolation of melanopsin mediated pupil light responses.
Sleep and circadian rhythms.
the impact of ipRGC dysfunction in ophthalmic and neurodegenerative disease.
Explore Melanopsin as a 5th dimension of human vision.
Understand the functional impacts of melanopsin activation on behavioural responses to light.
History of Melanopsin Discovery
1927: A functionally blind mouse (due to retinal degeneration) was found to have robust pupil light responses, despite the absence of rods and cones (Keeler, 1927, Am J Physiol).
2000: A new pigment named melanopsin was discovered to be expressed in the human retina (Provencio et al., 2000, J Neurosci).
Melanopsin was detected on a subset of ganglion cell bodies, dendrites, and axons in mice and primates, leading to the name intrinsically photosensitive RGCs (ipRGCs) (Hatter et al., 2002, Science; Dacey et al., 2005, Nature).
A blind person (complete rod and cone degeneration) reported experiencing brightness in response to high-intensity blue/cyan light (Zaidi et al., 2007, Curr Biol).
The spectral sensitivity of the post-illumination pupil response (PIPR) matched melanopsin (Markwell, Feigl, & Zele, 2010, Clin Exp Opt).
In humans, melanopsin can see independent of rods and cones but cannot reconstruct image form (Nugent & Zele, 2024, Proc Natl Acad Sci USA).
ipRGCs
Normal Human Fundus: Displays melanopsin expressing intrinsically photosensitive Retinal Ganglion Cells (ipRGCs).
Cell Count:
>100 million rods (night vision).
- million L, M, S cones (day vision).
<5000 melanopsin ipRGCs per eye (<0.5% of all retinal ganglion cells).
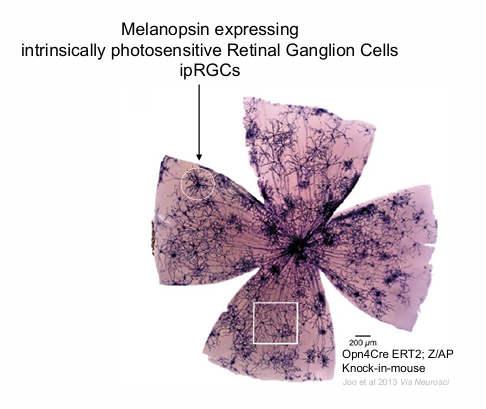
Diagram:
cells (circle) is a melanopsin cells
dark centre is the ON
the axons from the melanopsin cells goes to optic disc.
visual responses and pupil responses can be mediated by melanopsin at the blind stop.
Human ipRGCs form a photoreceptive net across the retina
no ipRGC at the fovea.
much larger than any other ganglion cells.
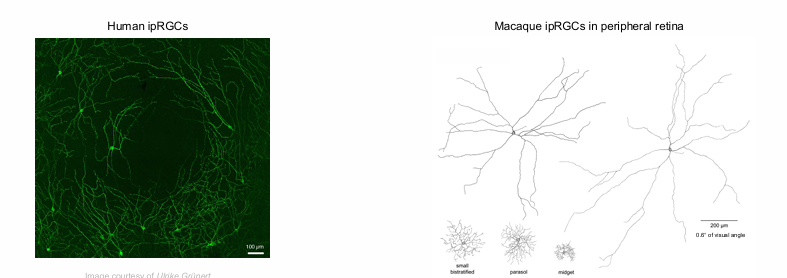
Macaque ipRGCs in peripheral retina cover of visual angle.
ipRGCs mediate extrinsic rod & cone photoreceptor signals.
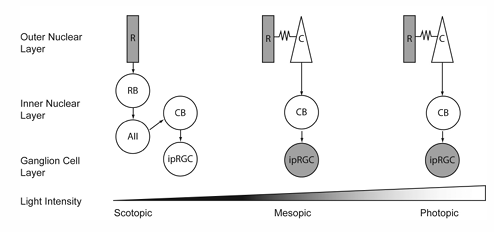
ipRGC Types and Function
ipRGCo (M1) - outer cell
ipRGCi (M2) - inner cells
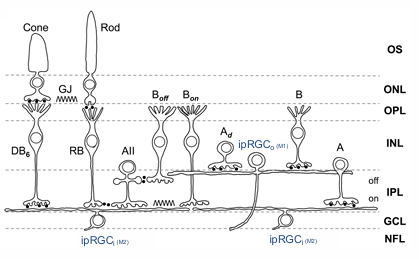
activation of the melanopsin cells causes feedback pathways to modulate the output of amacrine cells and dopamine amacrine cells and bipolar cells.
hence rods and cones are influenced by melanopsin
Rods and cones, through bipolar cells, AII amacrine and bipolar cells, input into Melanopsin
important for circadian rhythms to tell brain that it is nighttime.
melanopsin is important for ‘coding’ information for the brain.
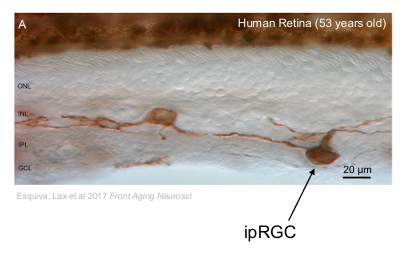
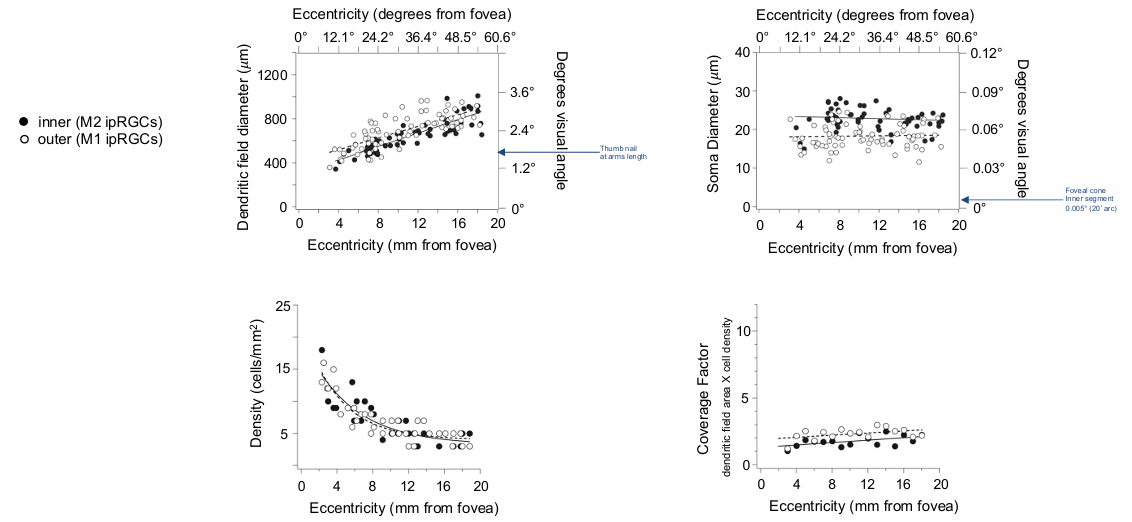
Morphological characteristic of ipRGCs in human retina
inner (M2 ipRGCs)
outer (M1 ipRGCs)
Density:
Density varies with eccentricity from the fovea.
Maximum density near the fovea, decreasing with distance.
eccentricity:
no melanopsin cells in the central 2 degrees.
Dendritic Field Diameter:
Increases with eccentricity.
Soma (cell body) Diameter:
Ranges from .
doesn’t really change with eccentricity
Coverage Factor:
Calculated as dendritic field area x cell density.
pretty constant - as diameter goes up, but density goes down = roughly CF of 2 melanopsin in an area.
density is highest outside of the fovea where the dendritic fields are smallest
the density decreases (fewer ipRGs) when you hit 60 deg in the peripheral retina.
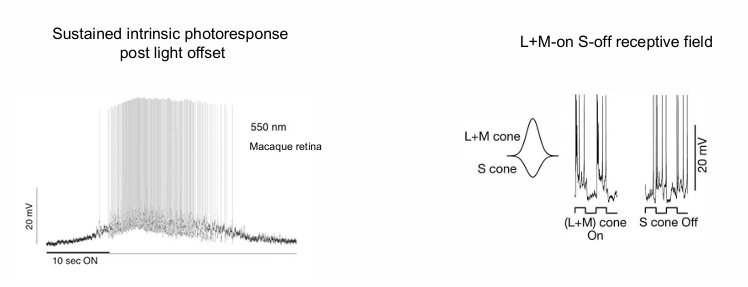
Physiological Response Properties of ipRGCs
Sustained intrinsic photoresponse post light offset.
when light is turned off (10 sec point), it continues to fires.
the major response is seen after the light is turned off.
linear relationship between number of quanta of light and cell spikes after the light turns off.
this is the signal that tells the brain it is morning or afternoon. No spikes = nighttime.
L+M-on S-off receptive field
receptive field has inputs on L+M cone (on) and S cones (off)
role in colour constancy.
Observed in Macaque retina.
Functions Mediated by ipRGCs
ipRGCs mediate the effects of light on circadian rhythms, pupil control, vision & mood.
ipRGCs inputs to brain areas for visual and non-visual processing
These are deep areas of the brain → implies that melanopsin has been around for a long time.
AH (anterior hypothalamic nucleus)
BST (bed nucleus of the stria terminalis)
IGL (intergeniculate leaflet)
LGN (lateral geniculate nucleus)
LH (lateral hypothalamus)
LHb (lateral habenula)
MA (medial amygdala)
OPN (olivary pretectal nucleus)
PAG (periaqueductal gray)
PHb (perihabenular nucleus)
PGN (pregeniculate nucleus (primates))
pSON (peri-supraoptic nucleus)
SC (superior colliculus)
SCN (suprachiasmatic nucleus)
SPZ (subparaventricular zone)
VLPO (ventrolateral preoptic area)
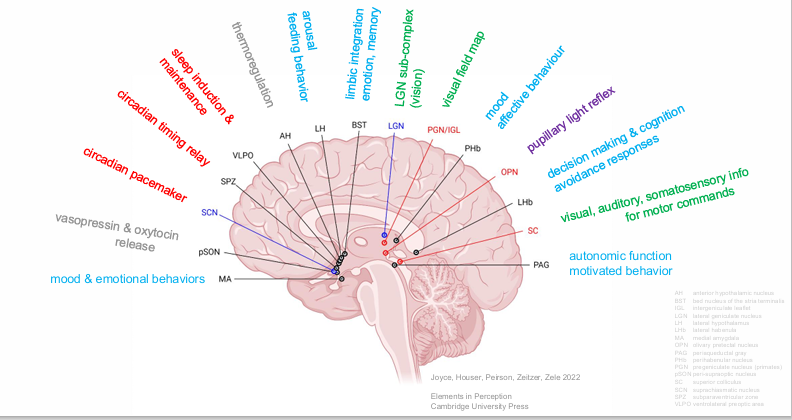
mood & emotional behaviors
autonomic function
motivated behavior
Pupil Light Response
ipRGCs control the pupil light response via brain projections to the olivary pretectal nucleus (OPN).
The sustained pupil constriction after light offset (PIPR) matches the spectral sensitivity of melanopsin (opn4)
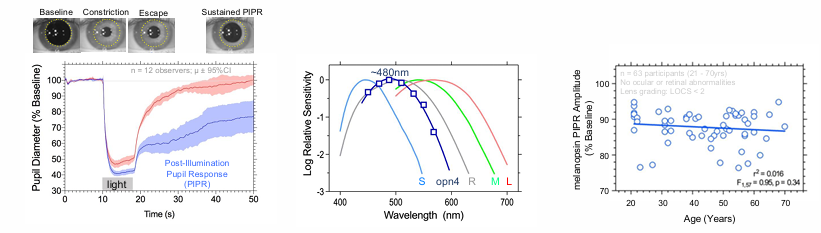
ipRGCs are more resilient to ageing than rod and cone pathways.
rods and cone degenerate over time - every years more ganglion cells are lost
melanopsin cells remain stable until at least the 7th decade.
Melanopsin and cone pupil responses combine additively in daylight
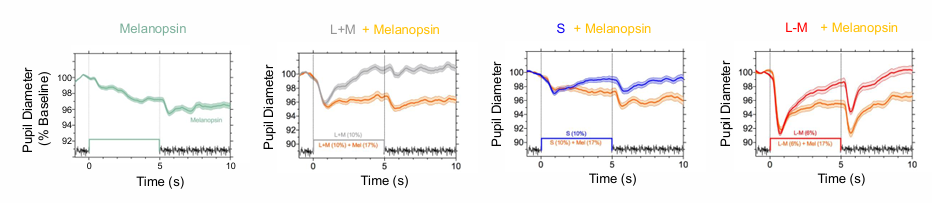
Melanopsin sets the pupil diameter during extended light exposure.
pupil constriction is very very slow when only activating melanopsin
after lights turn off, pupil size is sustained.
Cone-initiated responses are transient to contrast changes.
cones quickly constrict then immediately return back to baseline.
melanopsin pupil responses provide a marker of their brain projections
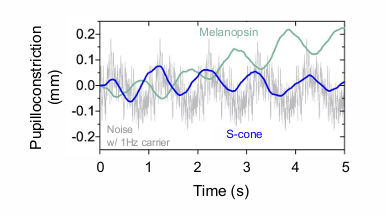
Melanopsin pupil flicker responses are opponent to the paradoxical S-cone response that dilates with increasing irradiance, reflecting the neurophysiological properties of the receptive field
when the light gets brighter - the pupil gets bigger for s-cone.
if the lights gets darker - it will get smaller for s-cone
PARADOXICAL S-CONE RESPONSE
melanopsin is in counter phase to the S-cones.
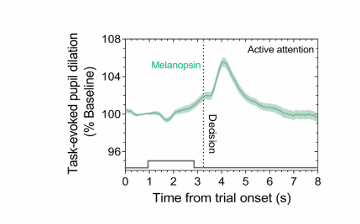
Task-evoked pupil dilations are linked to the global cortical arousal state via ipRGCs inputs to the amygdala, pulvinar & superior colliculus
pupil dilates on decision making → can activate deep brain through melanopsin pathway
marker of someone actively making a cortical decision.
Sleep and Circadian Rhythms
ipRGC control of sleep and circadian rhythms via brain projections to the suprachiasmatic nucleus (SCN) & ventrolateral preoptic area (VLPO).
circadian rhythm requires IPRGs and melanopsin cells, but cones don’t make a major contribution.
High prevalence of circadian disruption in eye disease: due to change in melanopsin function in the eye
Blind or partially blind patients
AMD (geographic atrophy & neovascular)
Glaucoma
Diabetes
Circadian disruption shows a causal relationship with:
Obstructive sleep apnoea
Medical and psychiatric illness
Restless leg syndrome
Autonomic dysfunction
how can we change the spectrums of light to account for dysfunctional responses.
Sleep & circadian rhythms quantified using objective (e.g., melatonin, polysomnography) & subjective makers (e.g., sleep diary, PSQI sleep quality assessment).
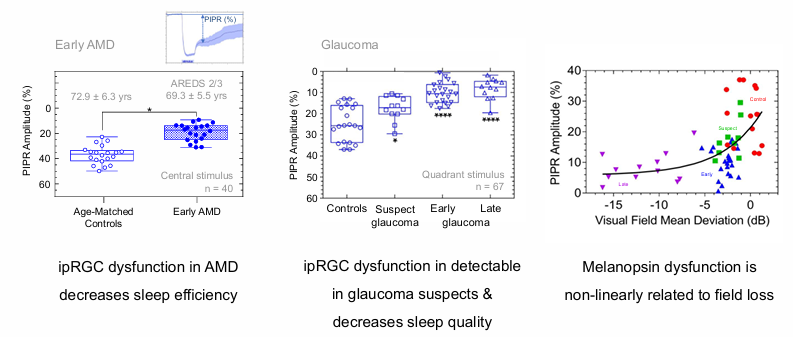
ipRGC Dysfunction in Retinal Disease
Glaucoma PIPR dysfunction is a robust biomarker maker of retinal disease
ipRGC dysfunction in AMD decreases sleep efficiency
Melanopsin dysfunction is non-linearly related to field loss
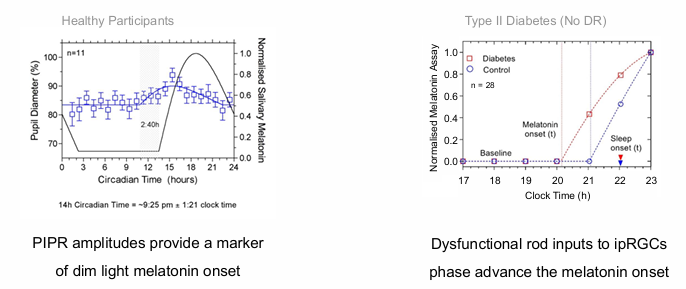
ipRGC signalling is phase locked to the dark light hormone (melatonin)
diabetics: Dysfunctional rod inputs to ipRGCs phase advance the melatonin onset.
in Diabetics - rods are primarily affected, leading to an early onset of melatonin (go to sleep early)
also leads to decreased sleep quality.
healthy participants: PIPR amplitudes provide a marker of dim light melatonin onset.
melanopsin function was suppressed through the retina by a higher order process in the brain.
Allows us to use melanopsin rather than directly measuring melatonin
useful application for shift-workers or jet lag.
ipRGCs and Parkinson's Disease
Altered ipRGC morphology is linked to sleep disruption in Parkinson’s disease
In PD, there are fewer ipRGCs & a smaller dendritic plexus.
Photoentrainment is advanced (phase advanced)
Melanopsin mediated PIPR amplitude is reduced.
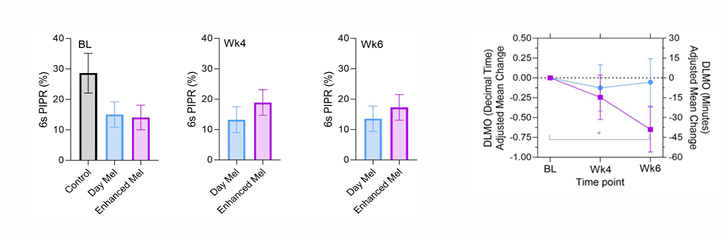
Photoreceptor Enhanced Light Therapy (PELT)
Recreating the biological effects of natural daylight with Photoreceptor Enhanced Light Therapy (PELT)
Light is a powerful modulator of sleep, mood & attention.
PELT targets the retinohypothalamic tract to synchronise circadian rhythms.
Daylight Melanopsin matches the biological effects of melanopsin, rods and cones as they occur natural daylight (3500K).
Enhanced Melanopsin lights cause a circadian equivalent increase in melanopsin excitation (5500K) to target melanopsin dysfunction.
Daylight & Enhanced Melanopsin PELT have the same colour and photometric luminance (i.e., are rod and cone metamers).
Daylight PELT significantly improves non-visual pupillary and circadian functions.
Phase II clinical trial evaluated PELT for 30 min/day over 4 weeks at a time pre-determined by chronotype
In patients with PD, stimulating the remaining retinal networks with daylight PELT compensates for the melanopsin degeneration to improve restorative deep sleep, wellbeing, pupil & motor function
purple line = enhanced melanopsin group → shift circadian rhythms by 36 minutes on average.
Melanopsin as the 5th Dimension of Vision
Melanopsin creates the 5th dimension of human vision via brain projections to the Lateral Geniculate Nucleus (LGN).
Isolating Melanopsin in humans:
melanopsin has lower sensitivity than rods and cones.
silent substitution using 5-primary lights (B C G A R) is needed to isolate melanopsin (i) function independent of the rhodopsin (R) & 3 cone opsins (S M L)
have to turn up the cyan and amber lights and town down a bit of red and blue, a lot of the green → activating melanopsin without seeing any changes from rods and cones.
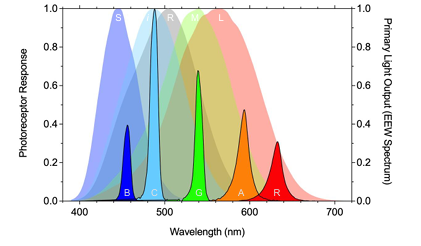
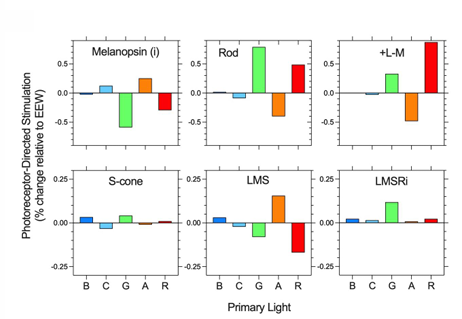
Observer calibrations minimise the effect of individual differences with reference to the CIE S026 2018 Standard Observer photoreceptor spectral sensitivities.
Melanopsin and Visual Perception
Melanopsin mediates conscious visual percepts
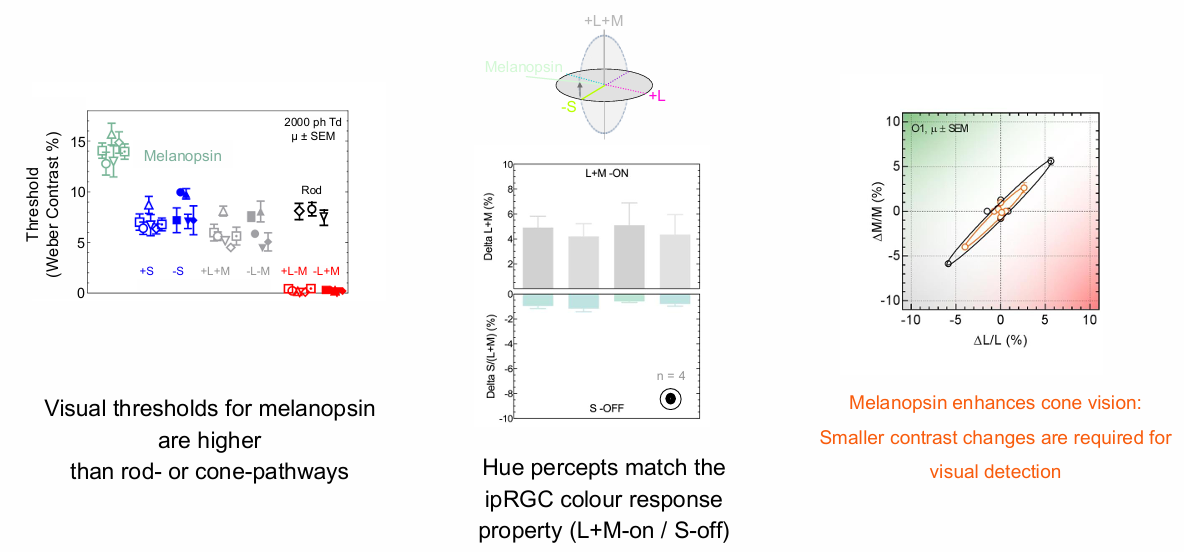
Hue percepts match the ipRGC colour response property (L+M-on / S-off).
Visualthresholds for melanopsin are higher than rod- or cone-pathways.
Melanopsin enhances cone vision: Smaller contrast changes are required for visual detection.
Melanopsin can detect low spatiotemporal patterns at high contrast.
Melanopsin’s unique detection tuning to low spatiotemporal elements (<0.4 cpd) is insufficient to create form for identifying image structure.
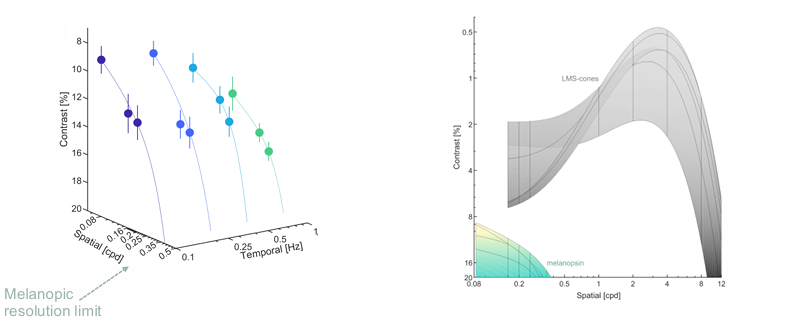
Melanopic spatial reconstruction using anatomical maps of human ipRGCs closely matches the dendritic field diameter of inner-stratifying M2 ipRGCs.
Visual percepts arising from melanopsin:
Melanopsin can see, but does not support form vision.
Melanopic gratings are perceived as spatially chaotic visual patterns.
Fixation losses lead to the disappearance of the melanopsin percept, consistent with sustained ipRGC signalling.
Percept described as fluorescent with a shared hue ranging from: sand-like faint yellow / orange with increased melanopsin activation, to a faint cyan (similar to a clear sky) with decreased melanopsin activation.
Melanopic hue may be used in colour appearance to ‘yellow-shift’ colour perception at cooler colour temperatures (e.g., bluish appearing daylight spectra) while ‘blue-shifting’ warmer colour temperatures (e.g., orangish appearing sunset spectra).
Melanopsin's Role in Daylight Visual Performance
Daylight visual performance is regulated by cones, rods & melanopsin
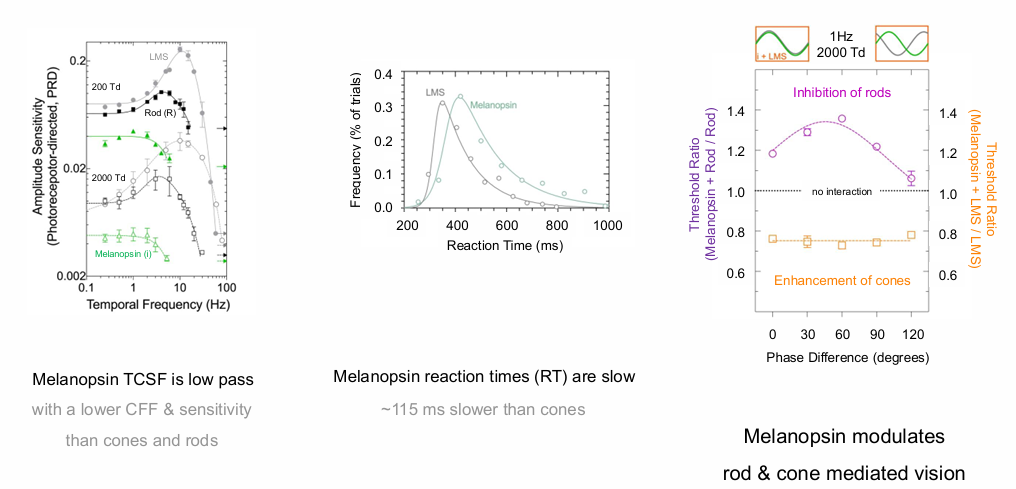
Melanopsin TCSF is low pass
Melanopsin reaction times (RT) are slow; ms slower than cones with a lower CFF & sensitivity than cones and rods.
Melanopsin modulates rod & cone mediated vision
Behavioural Responses to Light
Functional impacts of melanopsin activation on behavioural responses to light.
Melanopsin controls the coding efficiency of cone signals from retina to brain
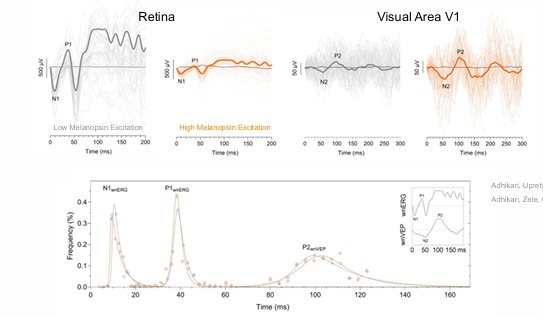
In the retina, melanopsin alters the gain of cone signals (i.e., adaptation).
In the brain (V1), melanopsin amplifies cone signals to increase sensitivity without altering transmission latencies.
Photophobia is driven by melanopsin & cone inputs to retino-thalamocortical pathways
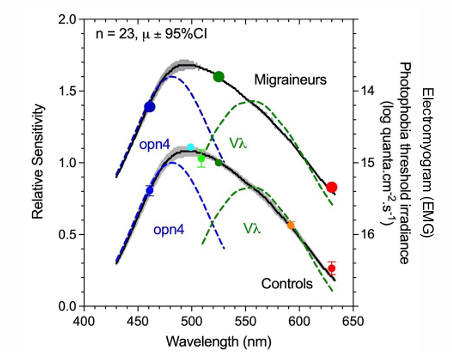
Migraineurs have supra-normal melanopsin responses (PIPR); Lower photophobia thresholds reflect hypersensitivity of ipRGC & cone luminance pathways.
Brightness perception requires melanopsin & cone pathways
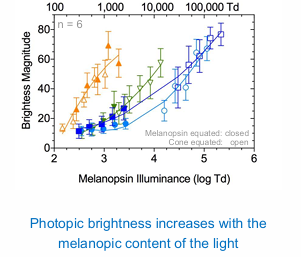
Melanopic content is an important consideration for artificial lighting design and energy efficiency in the built environment.
Melanopsin cells mediate both the visual and non-visual effects of light on behaviour and well-being.
Summary of Melanopsin's Roles
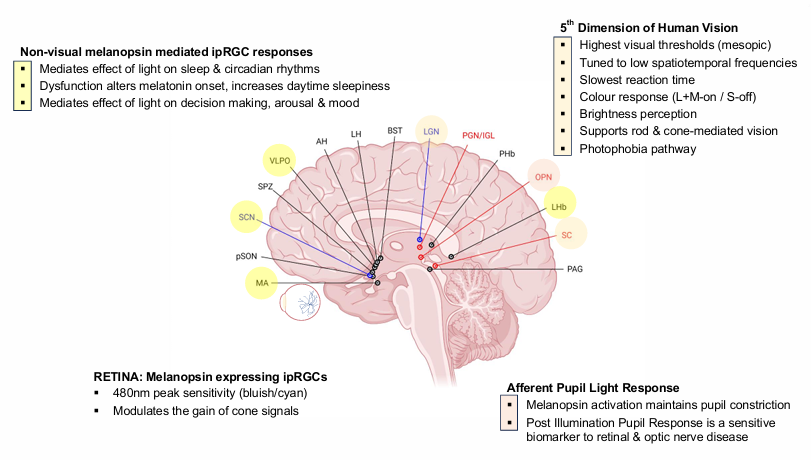
RETINA: Melanopsin expressing ipRGCs
nm peak sensitivity (bluish/cyan).
Modulates the gain of cone signals
Afferent Pupil Light Response
Melanopsin activation maintains pupil constriction.
Post Illumination Pupil Response is a sensitive biomarker to retinal & optic nerve disease
5th Dimension of Human Vision
Highest visual thresholds (mesopic).
Tuned to low spatiotemporal frequencies
Slowest reaction time
Colour response (L+M-on / S-off)
Brightness perception
Supports rod & cone-mediated vision
Photophobia pathway
Non-visual melanopsin mediated ipRGC responses
Mediates effect of light on sleep & circadian rhythms.
Dysfunction alters melatonin onset, increases daytime sleepiness
Mediates effect of light on decision making, arousal & mood
Learning outcomes: Questions to ask yourself
How are melanopsin cells (ipRGCs) anatomically and physiologically different from rods and cones?
Describe how the pupil light response and silent-substitution vision methods can be applied to quantify melanopsin function?
What effect does melanopsin have on cone signals in the retina (e.g., ERG), through the pupil control pathway, and in the visual cortex?
Melanopsin is a pigment discovered in 2000, expressed in the human retina within a subset of ganglion cell bodies, dendrites, and axons, known as intrinsically photosensitive RGCs (ipRGCs). These cells, numbering less than 5000 per eye, form a photoreceptive net across the retina, mediating both extrinsic rod & cone photoreceptor signals.
Key roles in the visual system:
Pupil Light Response: ipRGCs control the pupil light response via projections to the olivary pretectal nucleus (OPN), with melanopsin setting the pupil diameter during extended light exposure.
5th Dimension of Human Vision: Melanopsin creates a 5th dimension of human vision via projections to the Lateral Geniculate Nucleus (LGN), mediating conscious visual percepts and enhancing cone vision.
Behavioural Responses to Light: Melanopsin controls the coding efficiency of cone signals from the retina to the brain, alters the gain of cone signals in the retina, and amplifies cone signals in the brain to increase sensitivity.
Photophobia Pathway: Melanopsin, along with cone inputs, drives photophobia via retino-thalamocortical pathways.
Non-visual responses: Mediates effects of light on sleep, circadian rhythms, decision making, arousal and mood.
summary of the anatomical and physiological differences between melanopsin-expressing cells (ipRGCs) and rods/cones, based on the provided notes:
Cell Count:
Rods: >100 million per eye (night vision).
Cones: 4-8 million per eye (day vision).
Melanopsin ipRGCs: <5000 per eye (<0.5% of all retinal ganglion cells).
Location and Coverage:
ipRGCs form a photoreceptive net across the retina.
No ipRGCs at the fovea.
Size:
ipRGCs are much larger than other ganglion cells.
Dendritic Field Diameter:
ipRGC dendritic field diameter increases with eccentricity (distance from the fovea).
Soma (cell body) Diameter:
ipRGC soma diameter ranges from ~20-40 and doesn’t change significantly with eccentricity.
Physiological Response:
ipRGCs: Sustained intrinsic photoresponse post-light offset.
-Rods & Cones: Transient responses to contrast change.
Receptive Field:
ipRGCs: L+M-on S-off receptive field (involved in colour constancy).
Temporal Response:
Melanopsin: Slow reaction times; ~115 ms slower than cones with a lower CFF & sensitivity than cones and rods.
Peak Sensitivity:
Melanopsin: 480nm peak sensitivity (bluish/cyan).
Visual Thresholds:
Melanopsin: Highest visual thresholds (mesopic).
Function:
Rods: night vision.
Cones: day vision.
ipRGCs: mediate the effects of light on circadian rhythms, pupil control, vision & mood.