SCIENCE 2Q reviewer
SCIENCE REVIEWER
Atomic theory
- A scientific concept that explains the nature of matter and its behavior.
- Evolved over centuries with contributions by other/of many scientists.
Scientist’s atomic model:
- Dalton - solid sphere model
- Thomson - plum pudding/blueberry muffin model
- Rutherford - nuclear model
- Bohr - planetary model
- Schrodinger - quantum model
Atomism
- A theory that was first proposed by the Ancient Greek Philosopher, Democritus.
- All matter is made up of tiny, indivisible particles called “atoms” or “atomos”, which are in constant motion.
Solid Sphere model
- Made by Dalton
- Revived Democritus’ theory
- Believed that atoms of the same element are identical in their properties
- Atoms cannot be subdivided
3 fundamental laws:
- Law of Convervation of Mass
- States that in a chemical reaction, the total mass of the reactants is equal to the mass of the products.
- The numbers of atoms are conserved, but they are rearranged during a reaction and thus their total mass is the same before and after a reaction.
- Law of Definite Proportions
- States that a pure substance is composed of the same elements in the same proportions by mass regardless of the source of the substance.
- Law of Multiple Proportions
- States that when two elements combine to form more than one compound, the mass of one element that combines with a fixed mass of the other element is always whole-number ratios.
Plum Pudding model
- Made by Thomson
- Used the Cathode Ray Tube Experiment to discover the electrons
- Had the idea that the empty space is positively charged
- Also known as the “plum pudding” or “blueberry muffin” model
Nuclear model
- Made by Rutherford
- Positively charged nucleus w/ electrons orbiting it
- Also known as the “cherry with a pit” model
- Atom is mostly empty space
- Discovered the nucleus using the Alpha Particle/Gold Foil Experiment
James Chadwick
- Discovered the neutron of an atom
Planetary model
- Made by Bohr
- Electrons are held in their orbits (energy level) by electrostatic forces
- Each electron is confined to a specific orbit
Line Spectrum
- Consists of specific discrete line on a background
Quantum Mechanic model
- Made by Schrodinger
- Described the probability of finding electrons within given orbitals, or 3D regions of space, within an atom.
- Electrons are confined to a space in an orbit
- Electrons move at a high speed which makes an electron cloud
- Is also known as the “cotton ball” model
- Electrons within atoms could be explained mathematically by treating them as matter waves
Energy levels/electron shells
- Each shell can only accommodate a limited number of electrons.
NOTE! Each energy level contains sublevels or subshells designated by the letters S, P, D, and F. Each subshell is known as an “atomic orbitals”. |
|---|
Subatomic Particles:
- Protons
- Neutrons
- Electrons
Subatomic : Sub = under/inside
Atomic = atom
Electrons
- Are on energy shells
- Outside the nucleus
Protons and Neutrons
- Have basically the same mass
- Inside the nucleus
PARTICLE | CHARGE | MASS |
|---|---|---|
PROTON | +1 | 1 |
NEUTRON | 0 | 1 |
ELECTRON | -1 | 1/2000 or 0.0005 |
NOTE! The nucleus contains the mass of an atom. Protons and Neutrons are responsible for the mass of an atom since they are inside the nucleus. |
|---|
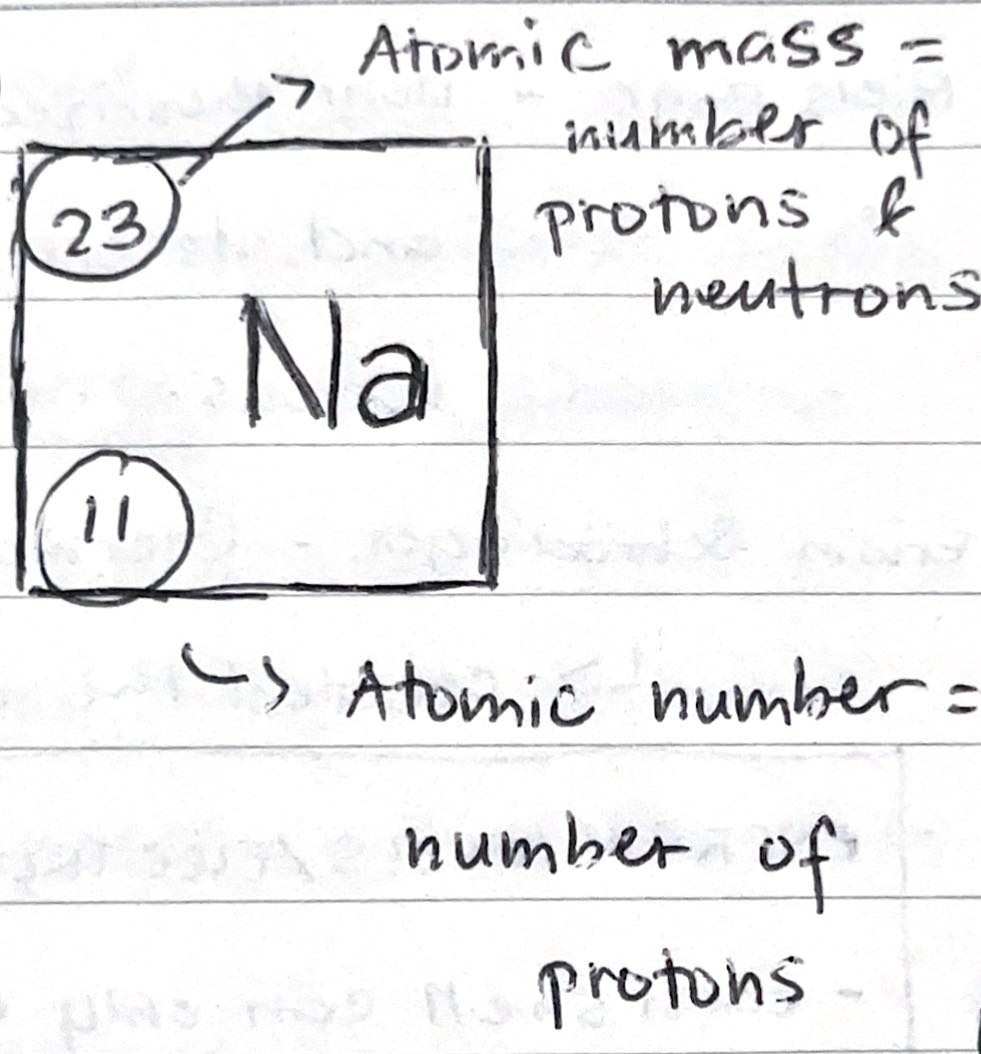
Atomic Number
- Number of protons
- Protons only
Atomic Mass
- Number of protons and neutrons
- Also known as “atomic weight” or “mass number”
- Protons + neutrons
Number of protons
- Dna of an atom
- Does not change
- Tells the so-called “identity” of an element
- Does not participate in chemical change
Neutral Atom
- Neutron is equal to the electron
- An element that has no charge
- Protons = electrons
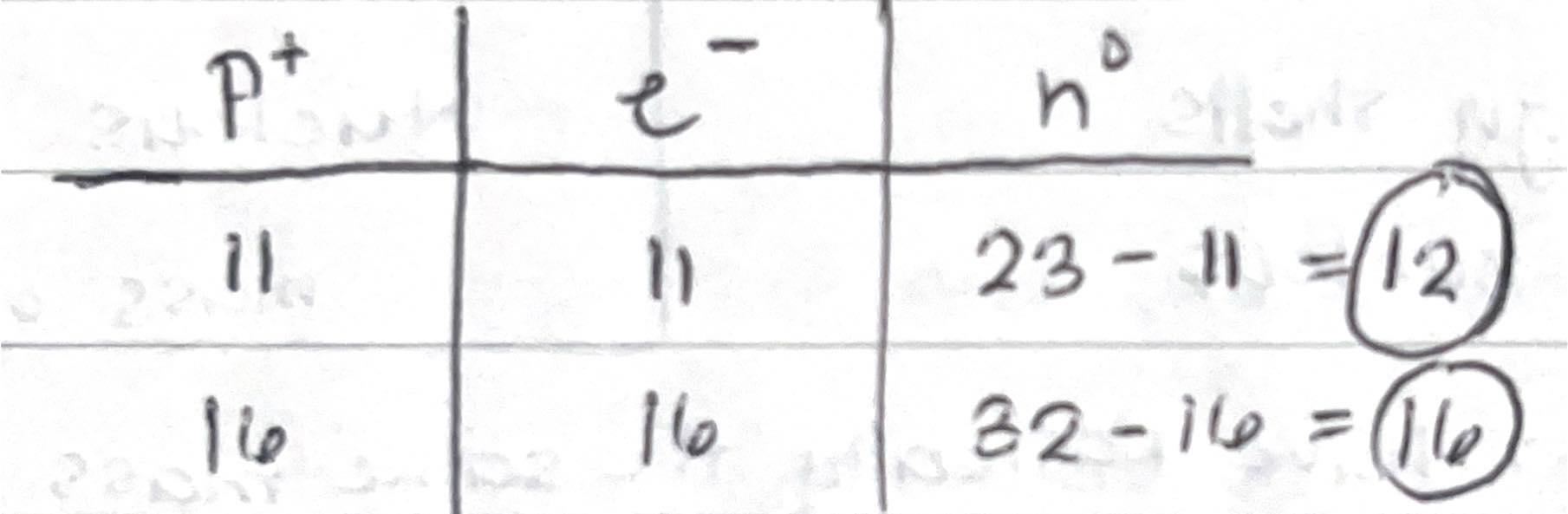
Ions
- Charged particles
- Have 2 types:
- positively charged ion = cation
- negatively charged ion = anion
Cation
- Donates electrons
Anion
- Accepts electrons
NOTE! Protons will never change as this is the identity of an element. The only subatomic particle to change is the electron. |
|---|
Periodic table
- Organizes the elements in a particular way
Johann Wolfgang Dobereiner
- The first developer of the periodic table
- Developed the law of triads
Law of Triads
- The atomic mass of the middle element was approximately equal to the average mass of the heaviest and the lightest ones
Dmitri Mendeleev
- The first to arrange the periodic table
- Developed the original periodic table
- Was the first to notice the relationship between the elements
- Arranged his periodic table by atomic mass
- Noticed that there were repeated properties that reoccur in a periodic pattern or “periodicity”
- Was known as the Father of the Periodic Table due to his prediction
John Newlands
- Made the “law of octaves”
- Noticed that every eighth element exhibited similar properties
Law of octaves
- Every eighth element exhibits similar properties in the periodic table
Julius Lothar Mayer
- Arranged the elements in increasing atomic mass
- Was the first to recognize the periodic trends in the elements
Henry Moseley
- Is responsible for the creation of the modern periodic table
- Elements should be arranged according to their atomic number
- Fixed the inconsistencies of Mendeleev’s periodic table
- Grouped the elements by their properties in a vertical column
- Added a new column called noble gases
NOTE! Elements can be classified into periods and groups/families. |
|---|
Periods
- Number of orbits in an atom
- Are horizontal rows on the periodic table
- Elements in the same period have consecutive atomic numbers
- Have the same number of energy shells/energy level
- Numbered from 1-7
Groups
- Are the vertical columns in the periodic table
- Elements of the same group usually have similar properties
- Elements of the same group have the same valance electrons
- Numbered from 1-18
- Not all of the groups follow the valance electrons
Valance electrons
- Are the electrons in the outermost energy level
Representative elements (A Group)
- Have predictable properties
- Groups 1&2 and 13-18
B Group
- The other elements
- The transition metals (the isolated elements are the inner transition metals)
- Have unpredictable properties

Special names of the representative elements:
- Group IA - alkali metals
- Group IIA - alkaline earth metals
- Group VIIA - Halogens
- Group VIIIA - noble gas
Alkali Metals
- Have a silvery appearance and are soft enough to cut with a knife
- Very reactive so they cannot be found in nature as free elements
Alkaline Earth Metals
- Harder, denser, and stronger than alkali metals
- Less reactive than alkali metals
Halogens
- Gases that are the most reactive nonmetals
- Reacts vigorously with most metals to form compounds
Noble Gas
- Do not react with other elements on the periodic table
- Do not need other elements to exist
- Chemically inert
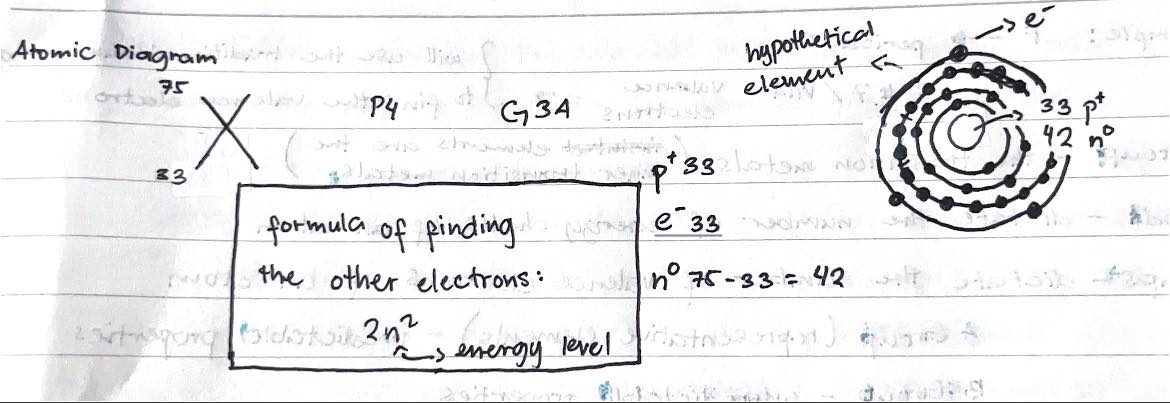
Atomic Diagram
Metals
- Are generally hard and lustrous
- The only nonsolid metal that is liquid at room temperature is mercury
- Are good conductors of electricity and heat
NOTE! Cg, Ag, and Au are chemically unreactive to other solids at room temperature and resistant to chemical corrosion and rusting. While Li, K, and Na are extremely reactive with air and water. |
|---|
Malleable
- Can be hammered or flattened into thin sheets
Ductile
- Can be drawn into thin wires without breaking
Nonmetals
- Generally exist as gases or solids in nature
- The only liquid nonmetal is bromine
- Because of their brittleness, they are neither malleable nor ductile
Metalloids
- Are elements that possess the properties of both metals and nonmetals
- A brittle solid that can conduct a limited amount of electricity is silicon
NOTE! Silicon is found in granite, quartz, clay, and sand. |
|---|