Estimating risk of inherited genetic disease
Lecture Summary: Alleles and selection. Calculating allele and genotype frequencies from population prevalence. Factors that change allele frequencies in a population. Heterozygote advantage and why harmful alleles are not eliminated from populations. Founder effects and importance of ethnicity in risk interpretation in some circumstances. Role of genetic testing. Examples to include sickle cell disease, cystic fibrosis and BRCA1 / 2. Ethnicity in interpretation of rare genetic variants.
Learning Outcomes: • Discuss the clinical relevance of population selection pressures and ethnic genetic variation
Alleles and Fitness
•Fitness” means the relative ability of organisms to survive (long enough) to pass on their genes.
•Alleles can affect fitness:
- not at all in most cases (neutral allele)
- sometimes decrease (deleterious allele)
- rarely increase (advantageous allele)
•In humans bringing up the child important too
–Role of grandparents / family / clan / society in food sourcing, defence against invaders, keeping child safe from harm and education
If selective pressures change, importance of different alleles may change

Source of genetic variation
•Mutation in recessive genes rarely affects carriers
–Few notable common recessive diseases
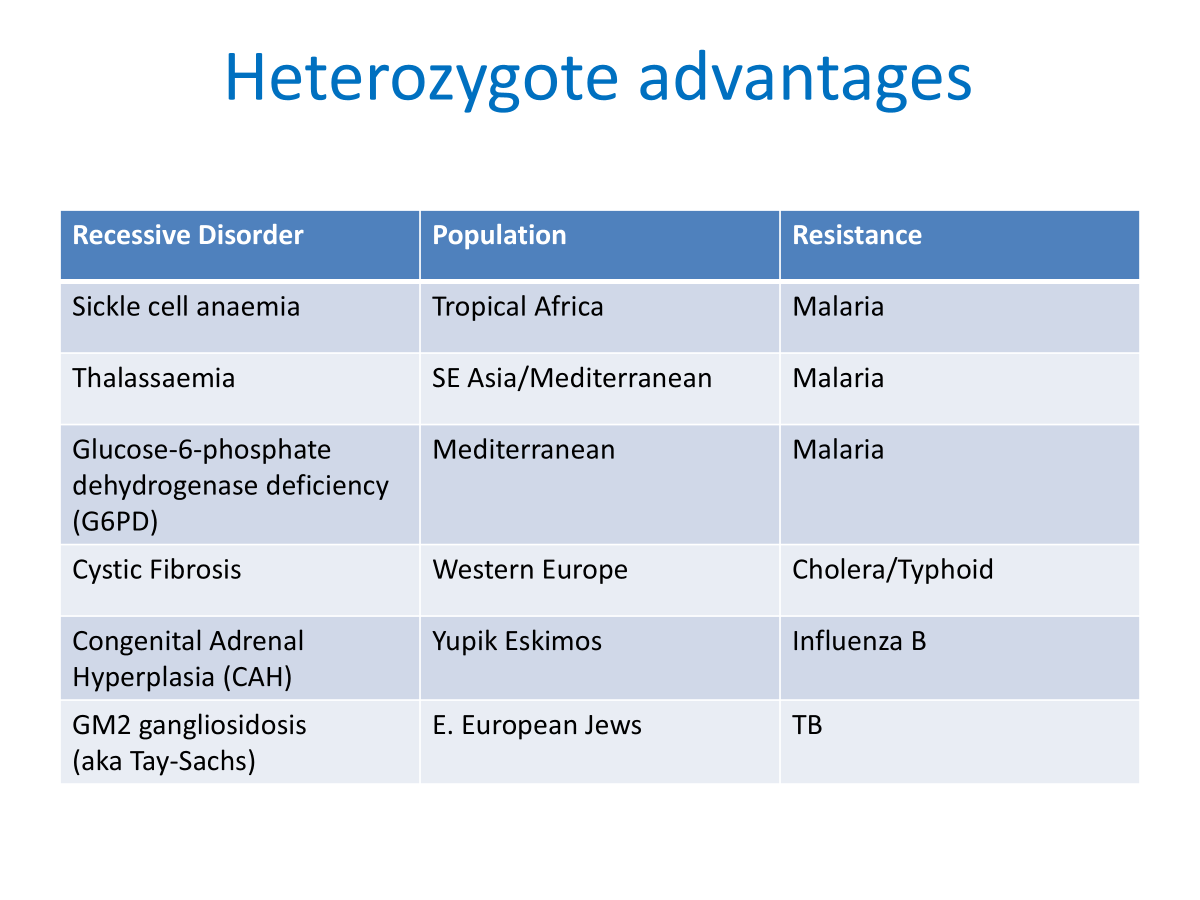
•Sickle cell disease
•Thalassaemia
–Where it does often associated with a selective pressure
•E.g. malaria resistance
•??plague or cholera resistance
–De novo recessive mutation uncommon as a cause of disease
•Mutation in dominant and X-linked genes can be inherited or de novo (anew or from the beginning)
Dominant & X-L mutation
•De novo mutation common in dominant disorders esp. where disease reduces reproductive fitness
–Recently recognised as a common cause of many severe learning disability syndromes
•Up to 1/3 of lethal X-L cases are due to a de novo mutation
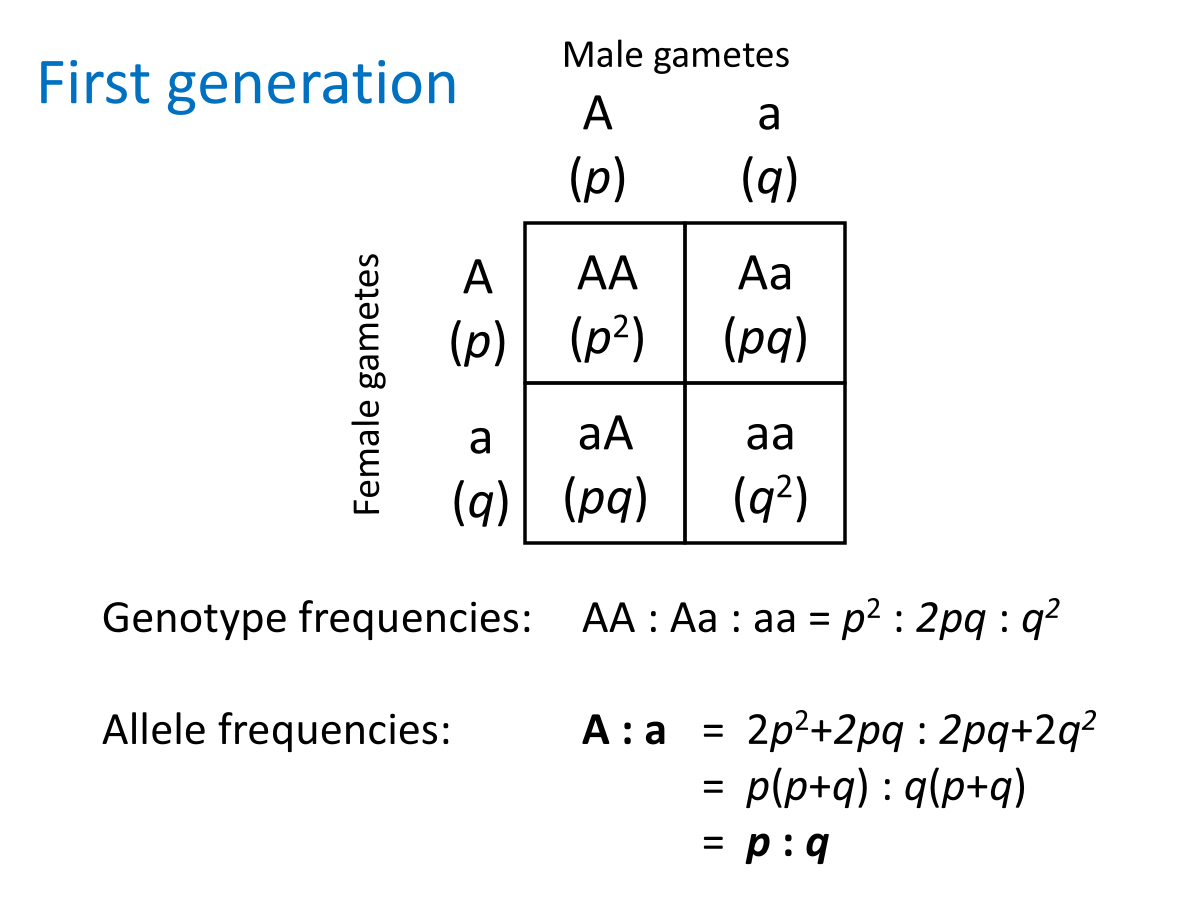
Genotype frequencies
A GENE with 2 alleles A and a
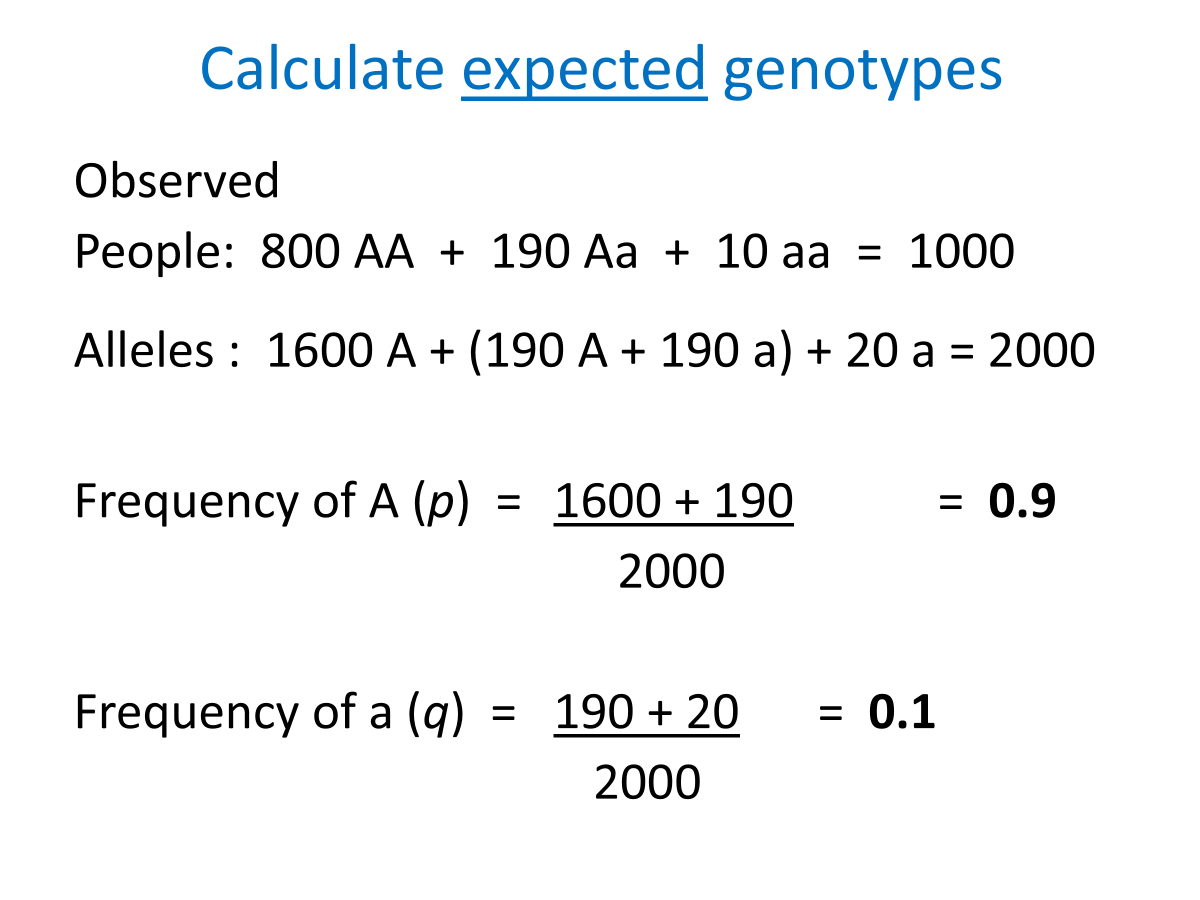
People : 800 AA + 190 Aa + 10 aa = 1000 total
Genotype frequencies:
AA = 800/1000 = 0.8
Aa = 190/1000 = 0.19
aa = 10/1000 = 0.01
Allele frequencies
People: 800 AA + 190 Aa + 10 aa = 1000
Alleles : 1600 A + (190 A + 190 a) + 20 a = 2000
Frequency of A (p) = 1600 + 190 = 0.9
2000
Frequency of a (q) = 190 + 20 = 0.1
2000
p + q = 1.0
P + Q, the two frequencies ALWAYS add up to 1
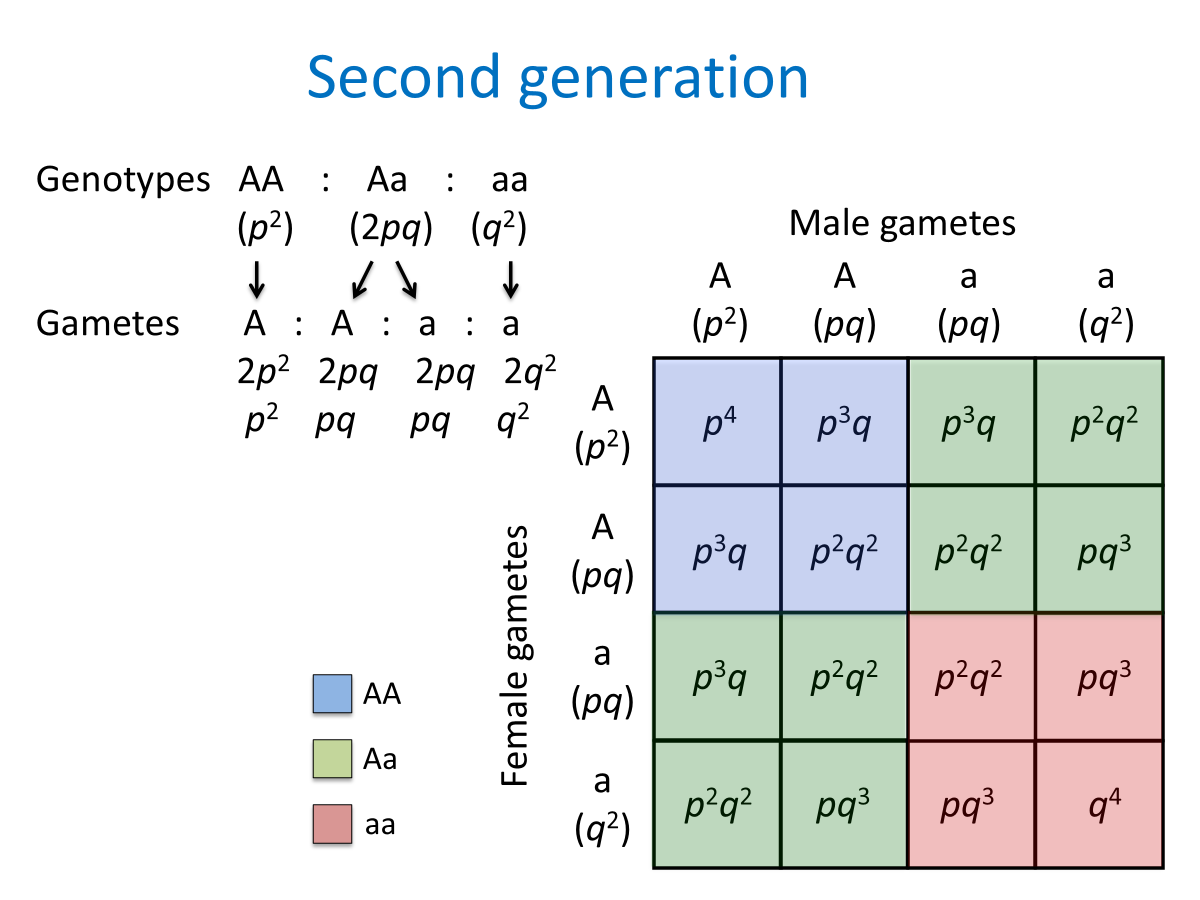

a recessive condition once in a population will stay the same frequency unless there is a selective pressure for the heterozygous

assumptions underlying HWE
Ideal population
-Mutation can be ignored
-Migration is negligible (no gene flow)
-Mating is random
-No selective pressure
-Population size is large
-Allele frequencies are equal in the sexes

Non-random mating
Non-random mating increases mutant alleles, thereby increasing proportion of affected homozygotes.
Assortative mating
- Choosing of partners due to shared characteristics
Deafness & sign language
Consanguinity
- Marriage between close blood relatives.
Cultural pressures for inter-marriage within clans / religions etc.
Natural selection
A gradual process by which biological traits become either more or less common in a population.
Negative selection
- Reduces reproductive fitness.
- decreases the prevalence of traits.
- leads to gradual reduction of mutant allele.
Positive selection
- Increases reproductive fitness.
- Increases the prevalence of adaptive traits.
-Heterozygote advantage.
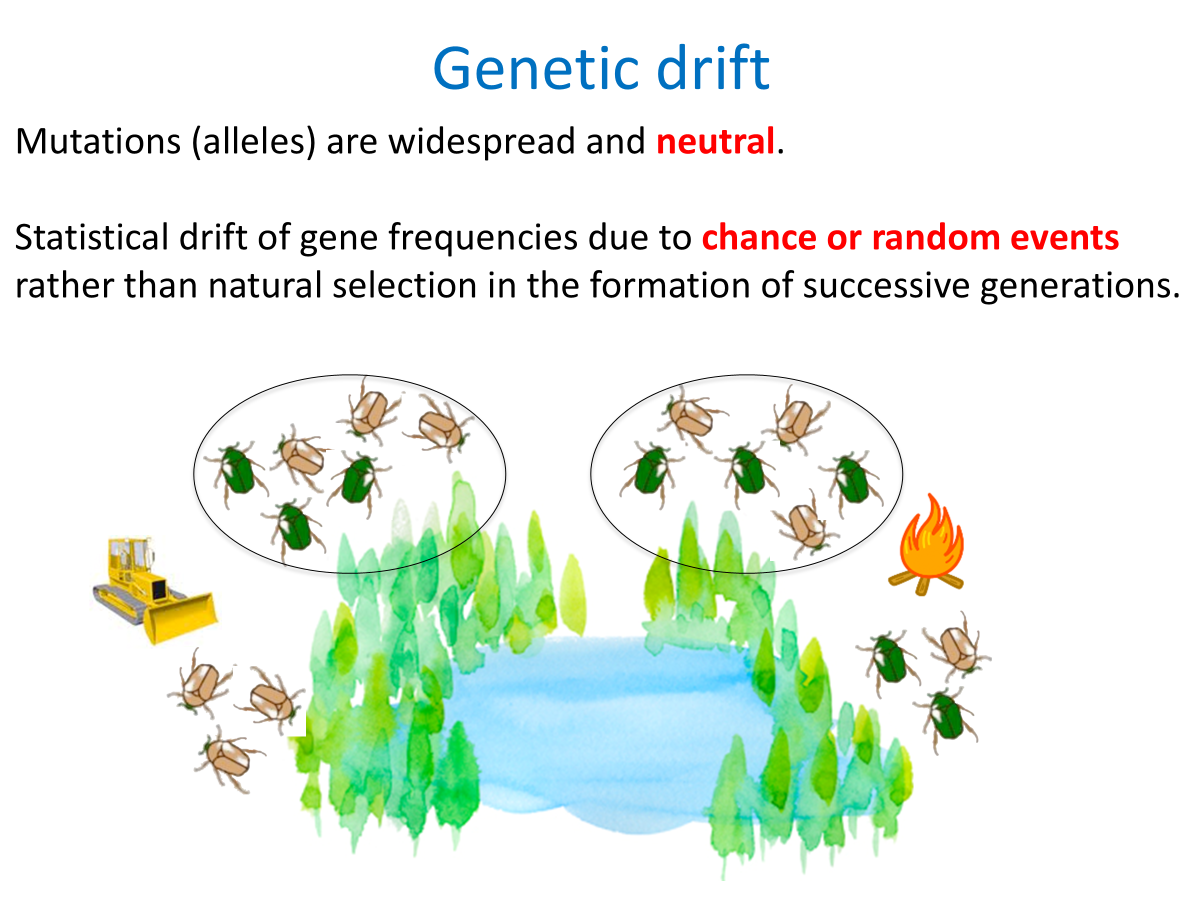
Small population size
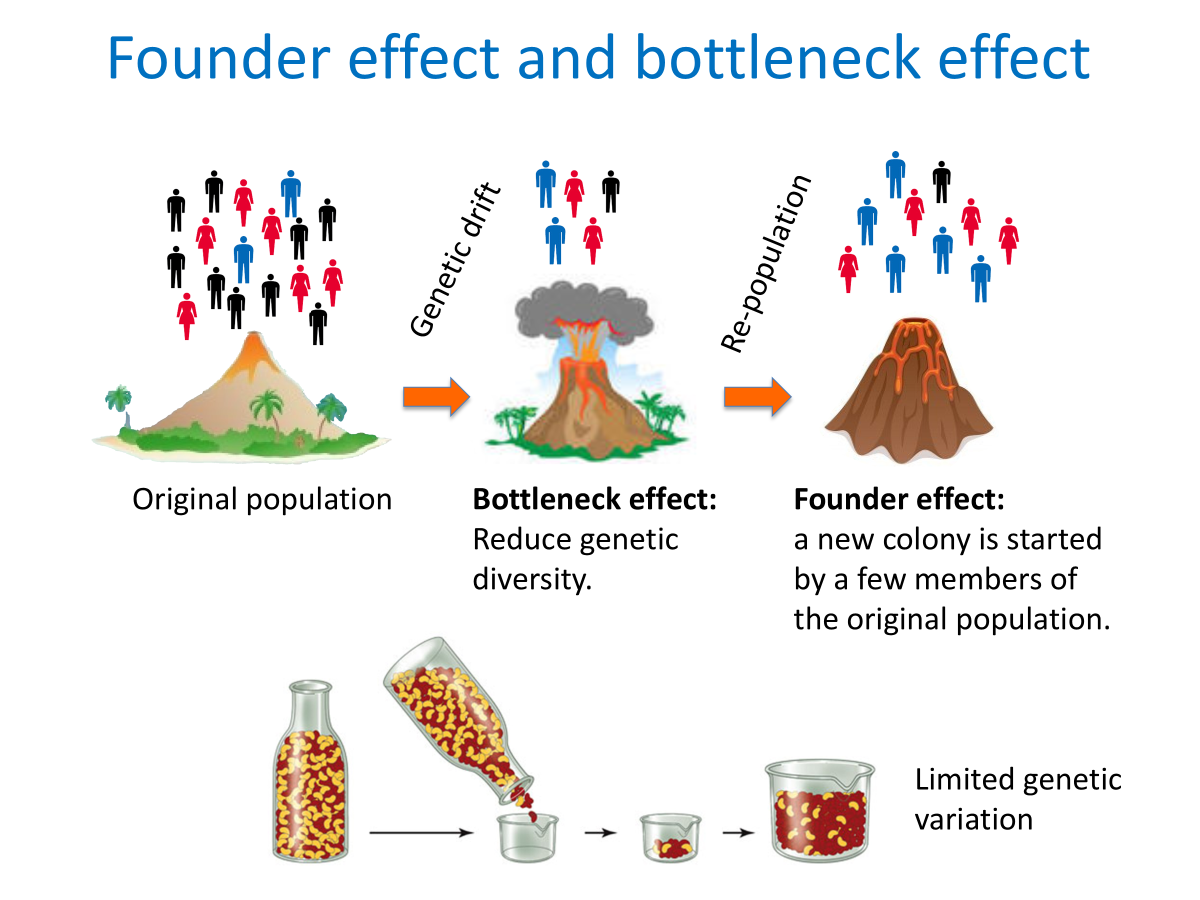
Large populations can balance out fluctuations, but small populations can exhibit “genetic drift” and cause “founder effect”.
Genetic drift:
Random fluctuation of one allele transmitted to high proportion of offspring by chance.
Founder effect:
The reduction in genetic variation that results when a small subset of a large population is used to establish a new colony.
Founder effect 2
•E.g. cystic fibrosis in the Faroes
–All one mutation
–CF More common than elsewhere in Europe
Cultural & geographic founder mutations
•Many examples
–Dominant
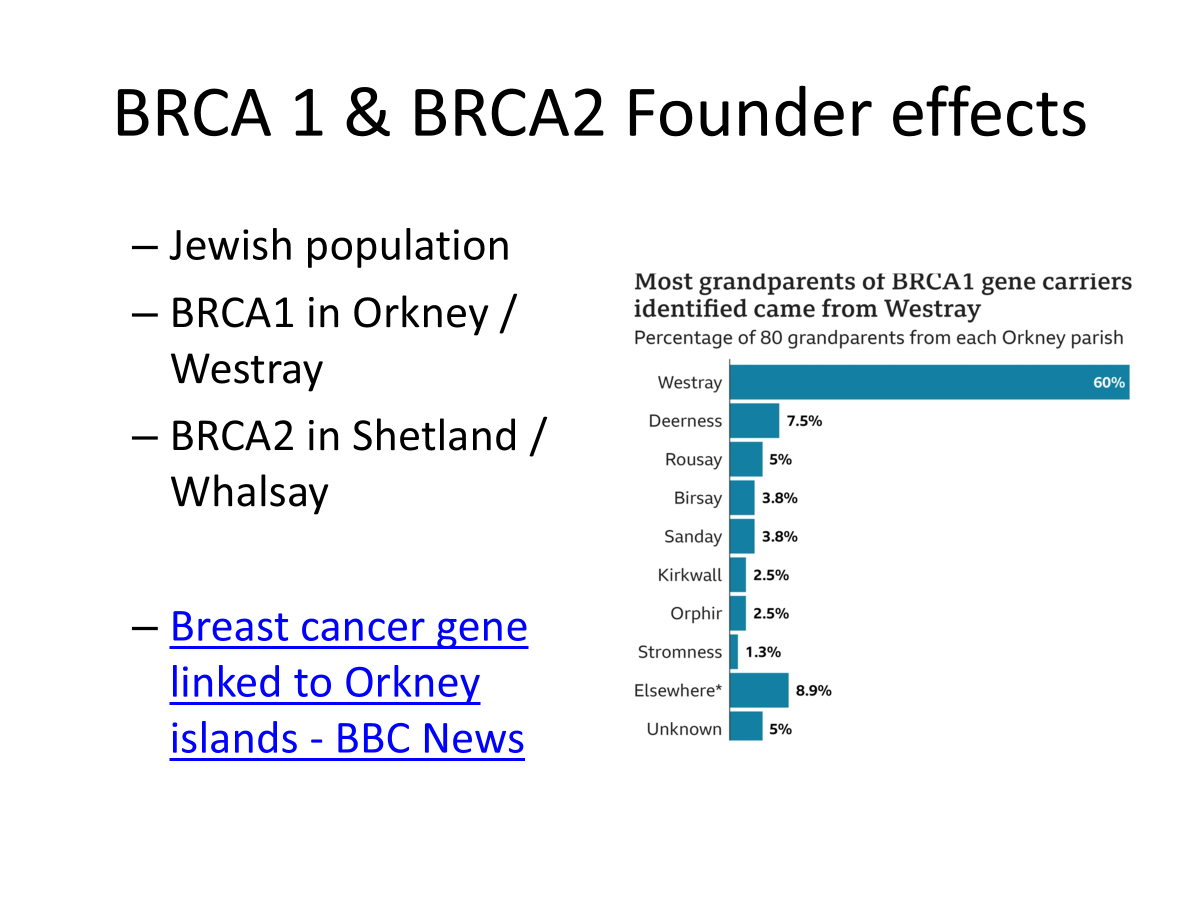
•Hereditary breast cancer gene mutations BRCA1/2
–Ashkenazi jewish
–Polish
–Lithuanian
–Scandinavian population isolates
–Orkney BRCA1 mutation
–Recessive
•Ashkenazi- Tay-Sachs etc.
•North-west European Celtic cystic fibrosis mutation

Applications of HWE
•Useful for calculating risk in genetic counselling
•Useful for planning population based carrier screening programmes


