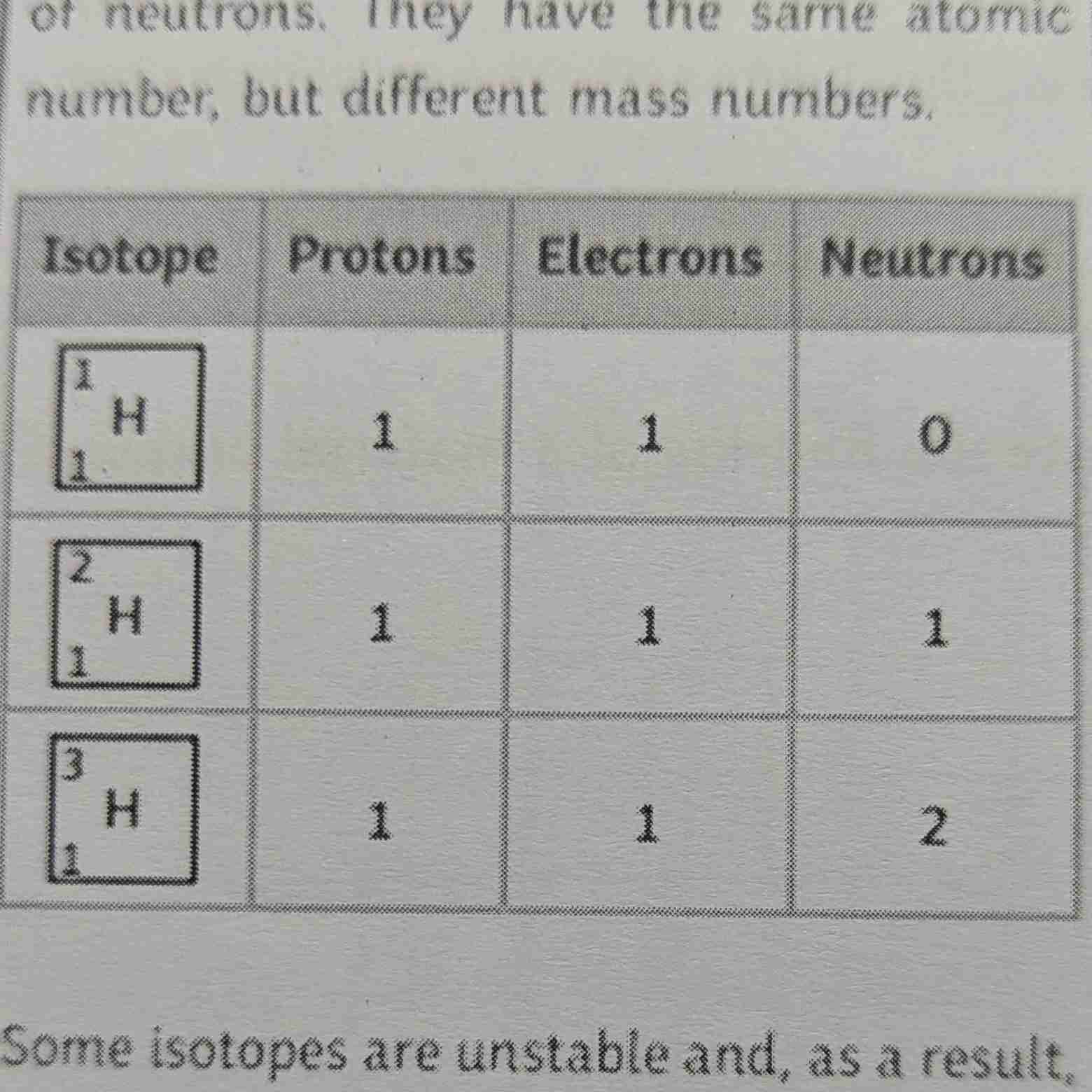
Isotopes
Isotopes: Isotopes are variants of a particular chemical element that have the same number of protons but different numbers of neutrons, resulting in different atomic masses.
Stable and Unstable Isotopes:
Stable isotopes do not undergo radioactive decay, whereas unstable isotopes (radioisotopes) emit radiation as they decay into other elements.
Applications:
Used in medical imaging (e.g., PET scans), carbon dating for archaeological finds, and as tracers in biochemical research.
Notable Examples:
Carbon-12 (stable) and Carbon-14 (unstable). The former is used in organic compounds, while the latter is used in dating ancient organic materials.