P3
Density
This is how compact an object is:

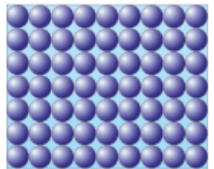
Solids: These have
Strong forces of attraction holding their particles close together
Arranges in a regular structure
Usually have the highest density due to their compact nature
Usually require the most energy to break apart solid forces of attraction
Can only vibrate around a fixed position when heated
Cannot fit the shape of their container
Draw them close together and compact like So:
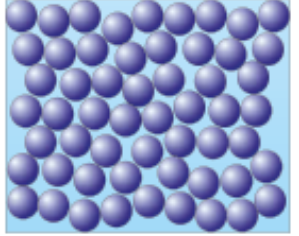
Liquids, These have:
Weaker forces of attractions between each particle
Particles are close together but can move past each other
Form irregular arrangements
Less dense than solids (Mid density)
Move about randomly at low speeds
Can fit the shape of their container
Draw all of them touching with gaps like So’s brother:
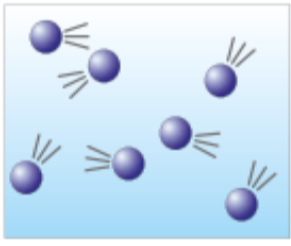
Gasses, These have:
Almost no forces of attraction
Have lots of energy
Move freely at high speeds
less dense than liquids (Lowest density)
Draw less than 4 with lines of motion to show that their fast moving like So’s cousin:
To find the density of an object you need to find its:
Mass and volume

Internal energy and changes of state
internal energy is the energy held inside the particles of on object:
E.g. The vibration of the particles as they get heated is their internal kinetic energy.
Or their GPE
If the internal kinetic energy is high enough then it can break the bonds between the particles changing its state
When an object is heated, its internal kinetic energy increases
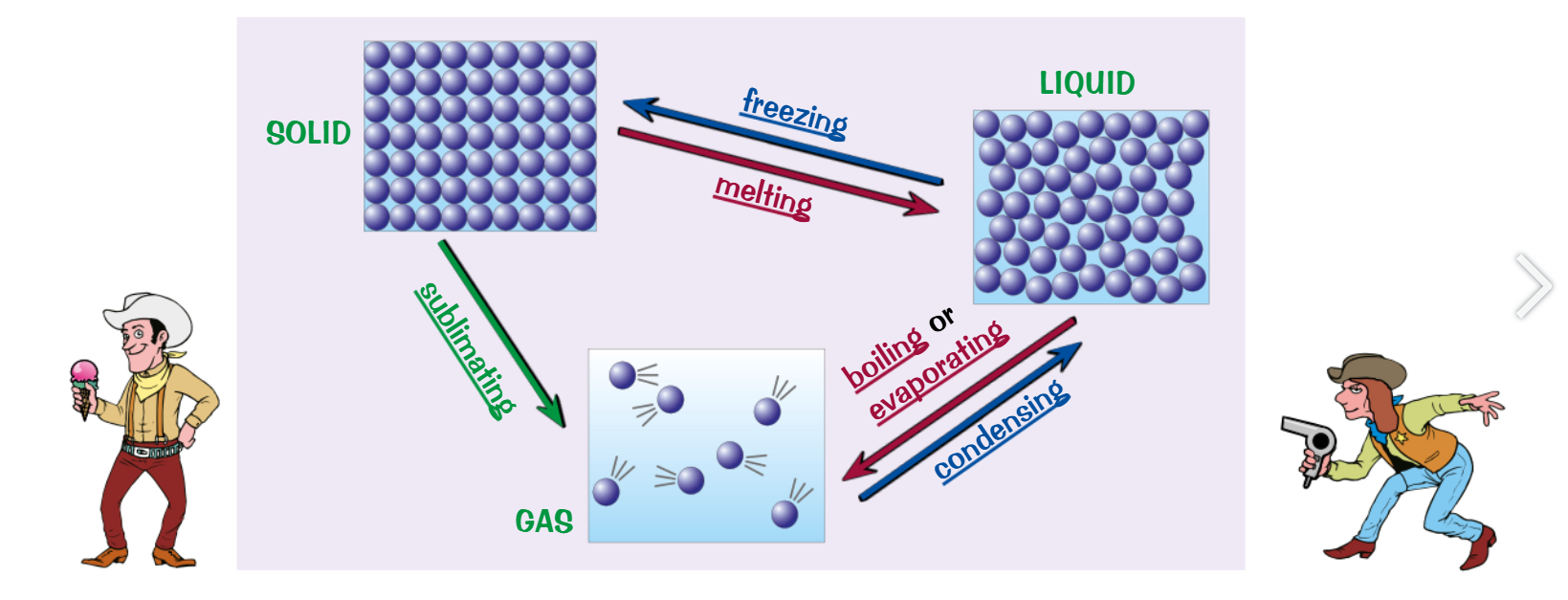
Specific latent heat
As a substant heats up the line goes up
When the line is flat that means that the energy is going towards breaking the bonds rather than increasing the temperature
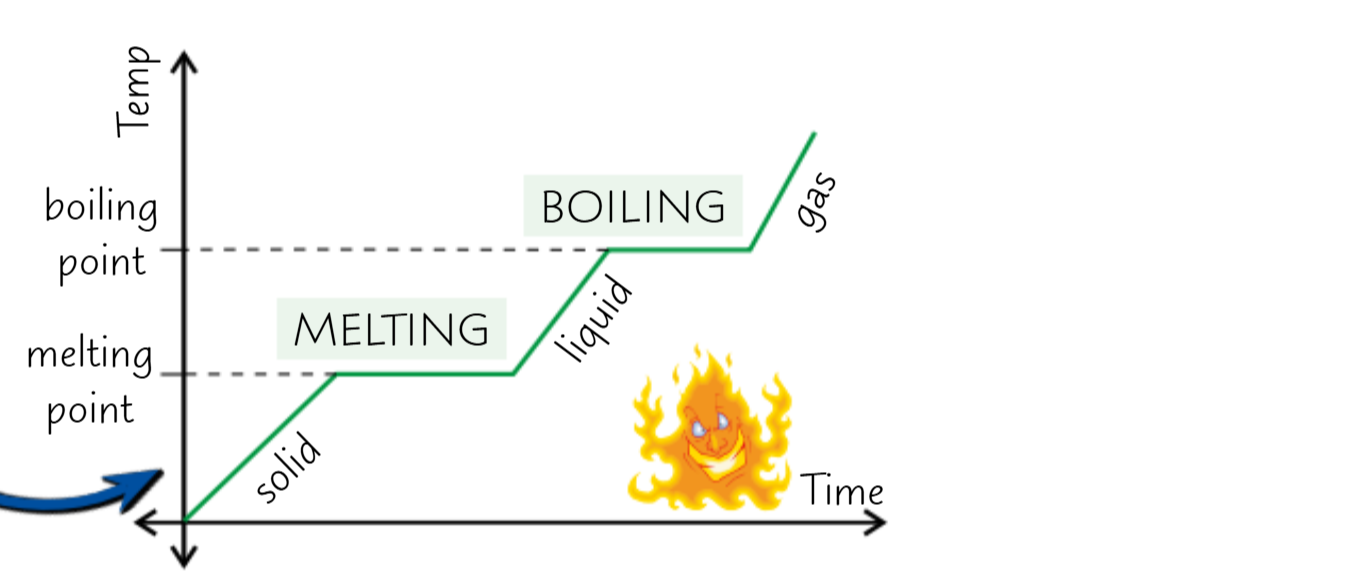
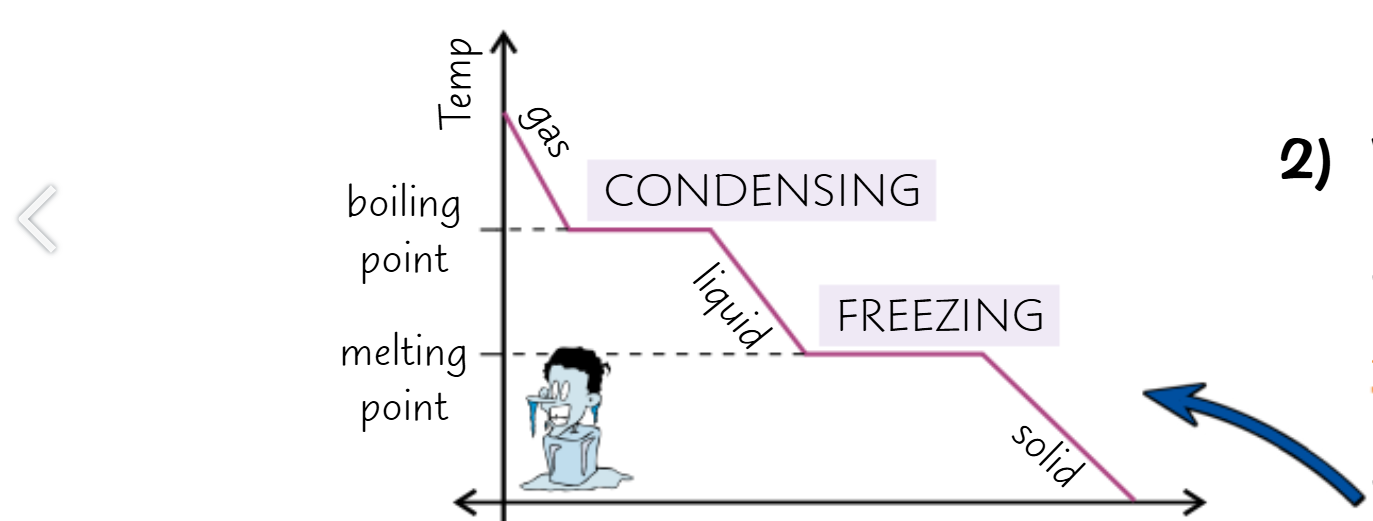
When the line goes down the energy is being lost to the surroundings
Which decreases the temperature
However when the line goes flat
Bonds are being made, which releases energy
Which stops the temperature decrease temporarily
Specific latent heat is the amount of energy needed to change the state of 1kg of substance without changing the temperature.
SLH of melting or freezing is called Specific latent heat of fusion
SLH of Boiling/ evaporation or condensation is Specific latent heat of vaporisation

Pressure
As you increase the temperature of a gas
you increase it’s kinetic energy
and you increase its speed
And it’s number of collisions
These collisions exert force on the inside of the gas’s container
Meaning that you increase the frequency of collisions when you increase its temp
And its force exerted
If you decrease the size of a gas’s container
Its pressure will rise
If you increase the temperature
Force will rise, increasing it’s pressure
Pressure = Force * Area
Pressure * volume = the constant

done…