BIOC*2580 - 5
Continued Discussion on DNA
Transitioning to the structure of DNA.
Review of DNA double helix geometry.
Discussion on base pair distances and helix dimensions.
Focus on Watson and Crick's contributions to DNA structure.
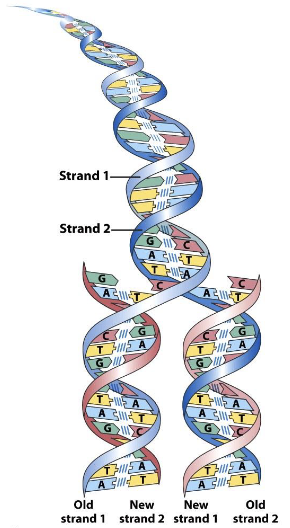
Key feature identified: Self-complementarity of DNA strands.
Explanation of how two strands with complementary sequences contribute to DNA functions.
Base Pairing Rules and DNA Replication
Significance of base pairing rules in DNA replication:
Strand one and strand two of the parental molecule serve as templates during replication.
Base pairing specifics:
Cytosine (C) pairs with Guanine (G).
Thymine (T) pairs with Adenine (A).
Implications for cell division and DNA repair mechanisms.
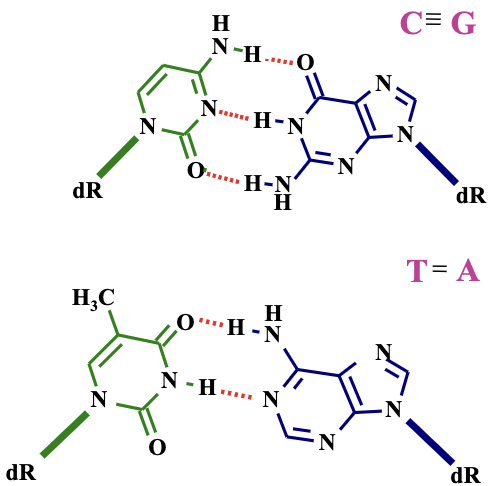
Hydrogen Bonds in DNA Structure
Discussion of hydrogen bonds' role in DNA stability.
Identification of hydrogen bond donors and acceptors among base pairs.
For instance, in C-G pairing:
C pairs with G through three hydrogen bonds.
Strength makes CG base pairs more stable than AT pairs (which utilize two hydrogen bonds).
Greater energy required to separate CG pairs in comparison to AT pairs.
Major and Minor Grooves in DNA
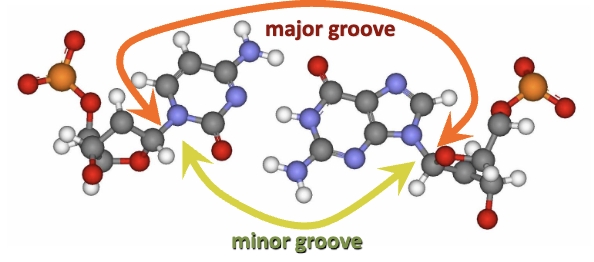
Examination of major and minor grooves resulting from DNA structure:
Grooves formed due to angled glycosidic bonds linking bases and sugars.
Implication of these grooves on accessibility of base pairs to other molecules (e.g., enzymes).
Importance in enzyme interaction and reaction facilitation.
Secondary Structure of Proteins vs. DNA
Contrasting secondary structures of proteins and DNA:
Proteins exhibit helices and strands influenced by amino acid sequences.
DNA's double helix structure largely constant regardless of base sequence.
Stabilization of DNA Double Helix
Discussion of forces stabilizing DNA structure:
Hydrogen bonds stabilize base pairing.
Hydrophobic effect due to sugar-phosphate backbone and base pair arrangement.
Van der Waals interactions among stacked bases.
Central Dogma of Molecular Biology
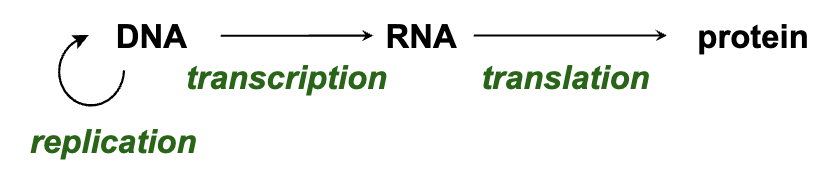
Explanation of the central dogma:
Information flow from DNA to RNA to proteins.
Clarified distinctions from chemical reactions.
Introduction to Metabolism
Segue into metabolism discussion.
Emphasis on ATP and its significance in metabolic processes.
Overview of pathways like glycolysis, TCA cycle, and beta-oxidation.
ATP Structure and Function
Description of ATP: Adenosine triphosphate and its components.
Contains adenine base, ribose sugar, and triphosphate units.
Discussed bonds in ATP:
Phosphoester bond between alpha phosphate and sugar.
Phosphoanhydride bonds between phosphates.
ATP as the energy currency in cells:
Analogous to money in society—energy required for cellular activities.
Importance of ATP Hydrolysis
Energy release during ATP hydrolysis.
Implications for driving endergonic reactions in cells.
Example calculation for ATP requirements in resting conditions.
Energy Cycle of ATP
ATP is not a store of chemical energy; rather it is a link between catabolism and anabolism
Cells breakdown nutrient molecules (catabolism), uses this energy to synthesize ATP from ADP in exergonic processes.
ATP donates energy to endergonic processes
ATP turns over (break down and synthesized) rapidly within the cell, allowing for continuous energy supply
ATP lifespan is seconds to minutes
Description of continuous ATP synthesis and usage cycle.
Highlights exergonic catabolic versus endergonic anabolic processes.
Importance of ATP being short-lived, necessitating constant regeneration.
High-Energy Bonds in ATP
Definition and significance of high-energy bonds in ATP.
Explanation of hydrolysis reaction and factors contributing to energy release.
Comparison of ATP's hydrolysis with other common bonds.
Mechanism of ATP Hydrolysis
Step-by-step explanation of ATP hydrolysis processes:
ATP —> ADP + PI (first mode): Single high-energy bond hydrolysis.
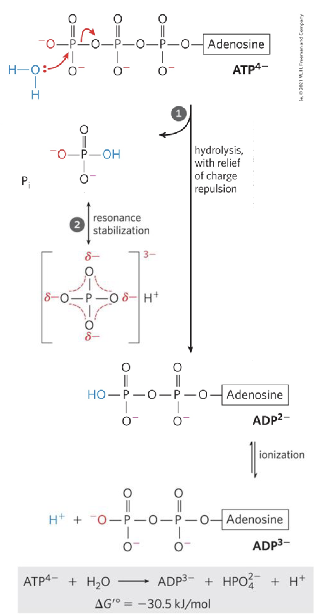
Hydrolysis releases the electrostatic repulsion
among the negative chargesThe product inorganic phosphate has greater
resonance stabilization than does ATP
ATP —> ADP + PI (first mode): Hydrolysis of both high-energy bonds
1. Hydrolysis of the link between gamma and beta phosphate
(nucleophilic attack of the γ phosphate)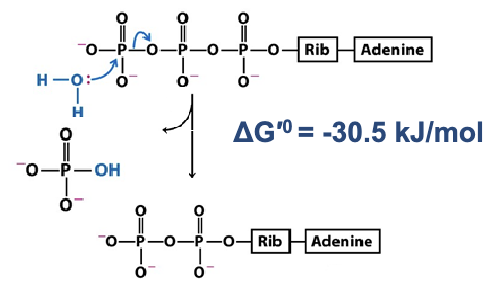
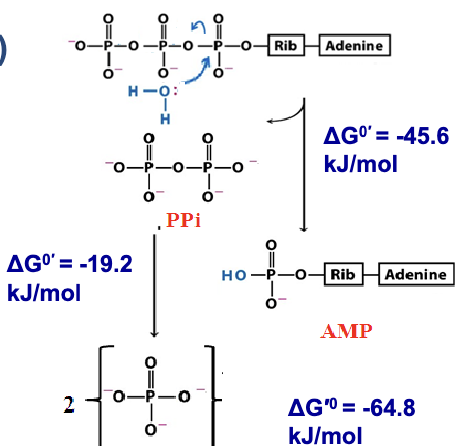
ATP —> AMP + 2 PI (second mode): Hydrolysis of both high-energy bonds via pyrophosphatase.
Group Transfer Mechanism
ATP as a participant in the group transfer mechanism:
Example with glutamate and glutamine synthesis.
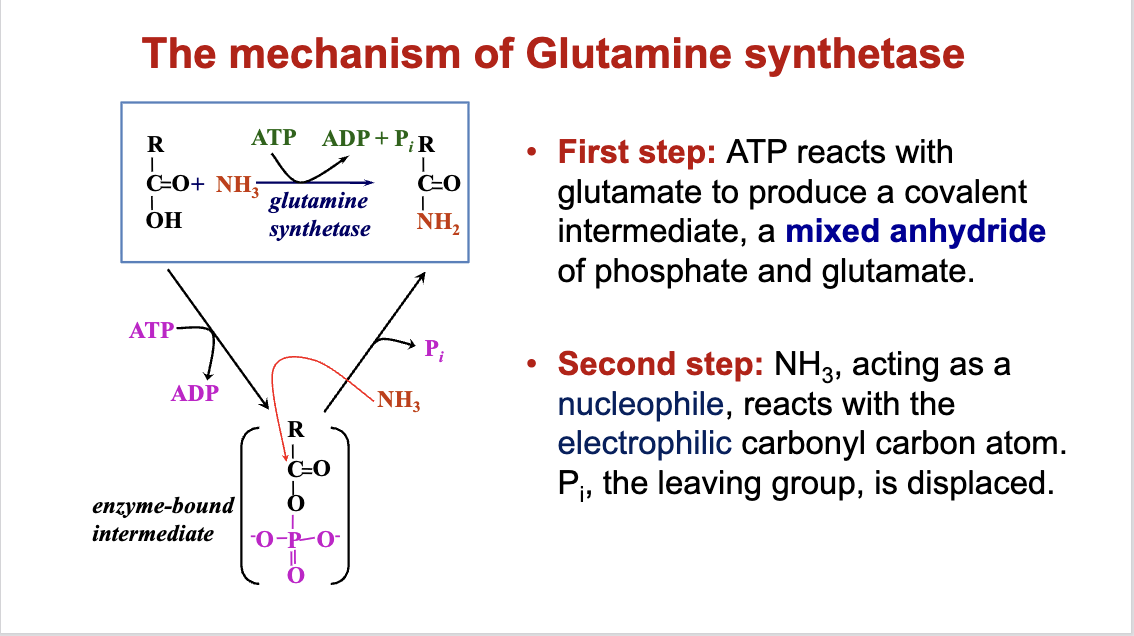
Highlights ATP's versatility in transferring various functional groups to influence reaction outcome.

Comparison of High-Energy Compounds
Overview of other high-energy compounds:
Creatine phosphate and its role in immediate energy supply for muscles.
Other phosphorylated molecules in biochemical reactions.
Acetyl-CoA as another high-energy molecule involved in metabolism.
Conclusion
Recap of ATP's role as the primary energy currency.
Emphasis on its significant investment in cellular energetic processes and the necessity for continuous synthesis.