Oxidative phosphorylation
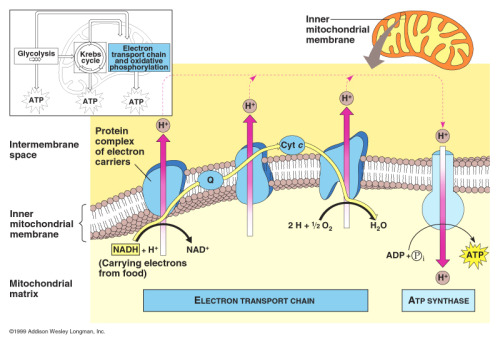
Takes place in inner mitochondrial membrane
Results in production of many molecules of ATP and production of water for oxygen
Chemiosmotic theory
Energy from electrons passed through a chain of proteins in the membrane (electron transport chain) via a series of redox reactions is used to pump protons (H+ ions) up their concentration gradient into the intermembrane space
H+ ions then flow by facilitated diffusion through a channel in ATP synthase into the matrix
Energy of the hydrogens flowing down concentration gradient harnessed which results in phosphorylation of ADP into ATP by ATP synthase
The electron transport chain
Made up of a series of membrane proteins/electron carriers
Positioned close together - allows electrons to pass from carrier to carrier
Inner membrane impermeable to H+ so electron carriers required to pump protons across the membrane to establish concentration gradient
Oxygen acts as final electron acceptor
Without oxygen accepting the electrons electrons won’t have anywhere to go
NADH and FADH2 cannot be oxidised to regenerate NAD and FAD, so they can’t be used in further hydrogen transport
Theoretical yield = 38 ATP per glucose molecule but rarely achieved due to inner mitochondrial memrane being leaky to H+ therefore not all H