Effect of Molecular Shape on Polarity
Molecules are formed in various shapes depending on the bonds formed between atoms, and the presence or absence of lone pairs. The differing shapes of a molecule affects its polarity, due to symmetry.
Symmetrical Molecules
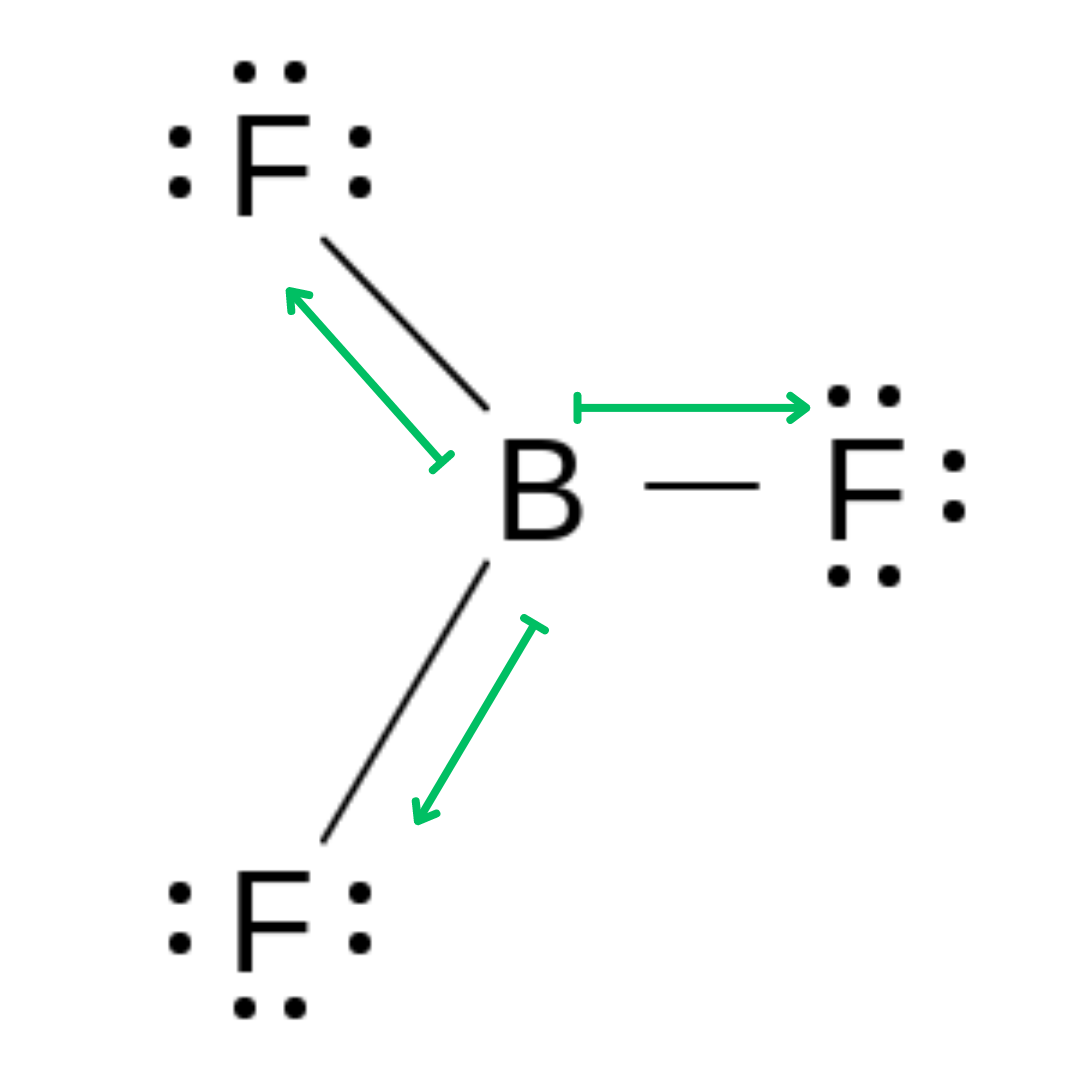
In a symmetrical molecule, equal but opposite bond polarities can cancel each other out, and result in a non-polar molecule that contains polar bonds.
Equal but opposite bond polarities are bond polarities within a molecule that have exactly the same ΔΣN as each other, so that each terminal atom exerts the same pull on the central atom, and forms a symmetrical molecule.
Symmetrical molecules can be linear, trigonal planar, or tetrahedral, but these molecule shapes are not always symmetrical
Examples:
Carbon Dioxide (CO2) →Linear Molecule
Boron Trifluoride →Trigonal Planar Molecule
Carbon Tetrachloride (CCl4) → Tetrahedral Molecule
The polarity arrows in all of these molecules point to the more electronegative atom in a bond from the less electronegative one. They also denote the electronegativity values with their size, so in each of these symmetrical molecules, all of the arrows in the diagram are the same size.
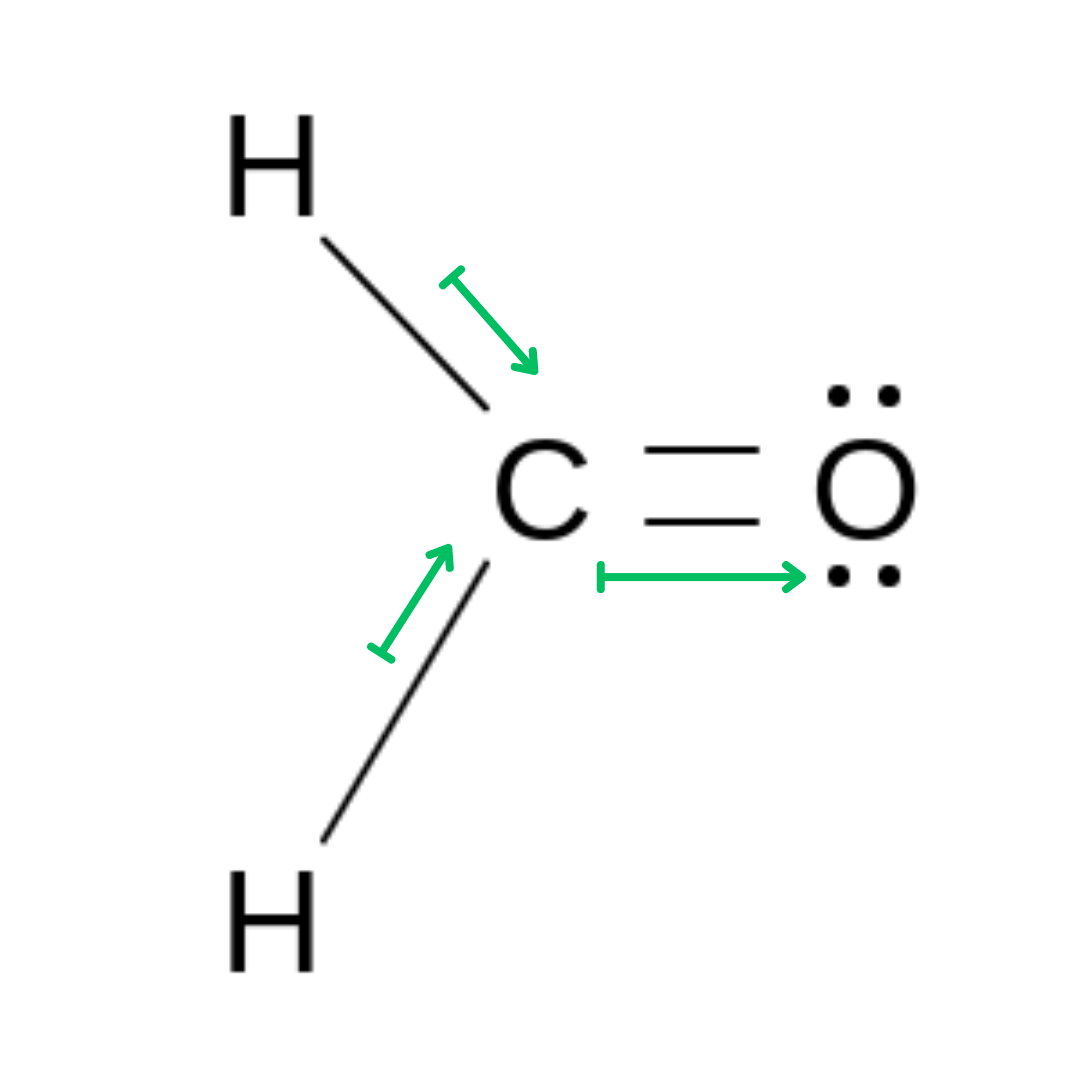
Non-Symmetrical Molecules
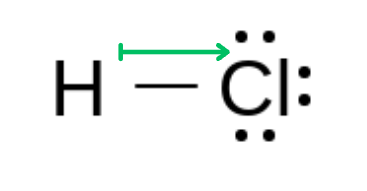
In a non-symmetrical molecule, the bonds do not cancel each other out, as they are differing values. This results in a polar molecule.
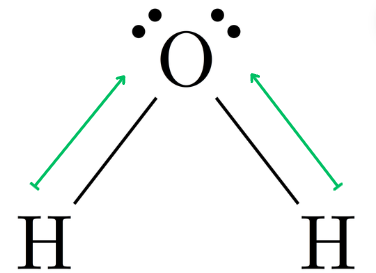
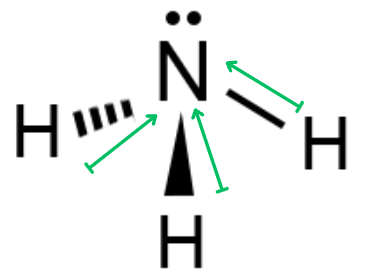
Non-symmetrical molecules are bent and pyramidal as well as the shapes that can be symmetrical. Bent and pyramidal shaped molecules’ bond polarities will never cancel out to make a non-polar molecule.
Examples:
Hydrogen Chloride (HCl) →Linear Molecule
Dihydrogen Monoxide (H2O) →Bent Molecule
Ammonia (NH3) →Pyramidal Molecule
Formaldehyde (H2CO) →Trigonal Planar Molecule
Chloroform (CHCl3) →Tetrahedral Molecule
The polarity arrows on these molecules donate the same as symmetrical molecules, but the use of length to indicate polarity is more apparent.