[FIN. 01] ANALYSIS II LABORATORY NOTES
[01] IPQC HOMOGENEITY TESTING IN SUSPENSIONS
What is Sedimentation Volume?
Definition: It is the ratio of the ultimate (final) volume of the sediment (Vu) to the original total volume of the suspension (Vo) before settling.
The Goal: To determine how much of the liquid remains as a “uniform mix” versus how much has settled as a concentrated layer.
Importance: A high sedimentation volume indicates a stable, flocculated suspension that is easy to re-disperse.
Sedimentation always correlates to suspension.
The volume when they are being measured they plays a lot of roles in terms of bioavailability, etc.
When there are different layers in a suspension, we can conclude that it is already unstable.
To calculate the sedimentation volume (F), we use the following equation:
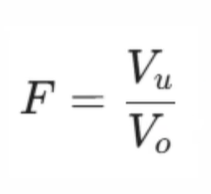
(a) Vu: Final settle volume of the sediment.
(b) Vo: Original total volume of the suspension.
The numerical value of (F) tells the story of the product’s homogeneity:
F = 1.0 (Ideal): No visible settling. The suspension is perfectly homogenous.
F = 0.5: The solid particles have settled to occupy 50% of the total volume.
F < 0.1: Significant settling; the drug is concentrated in a tiny layer at the bottom.
F > 1.0: Occurs when the network of particles (floccules) is so loose it occupies more space than the original liquid (rare but possible in highly flocculated systems).
The higher the sedimentation volume, it is most likely stable.
IPQC PROCEDURE:
Sampling: Take a known volume (Vo) of the suspension (e.g., 100 mL).
Transfer: Pour it into a graduated cylinder.
Settling: Allow the cylinder to stand undisturbed for set period (e.g., 24 hours or 7 days).
Measurement: Record the height/volume of the sediment layer (Vu).
Calculation: Apply the (F) formula.
Redispersibility: Invert the cylinder to see how many “shakes” it takes to return to a homogenous state.
FACTORS AFFECTING SEDIMENTATION:
To maintain a high (F) value and homogeneity, IPQC monitors:
Particle Size: Smaller particles settle slower (Stoke’s Law).
Viscosity: Thicker liquids (using agents like Xanthan Gum) slow down settling.
Zeta Potential: Electrical charges that keep particles from clumping into a hard cake.
CONCLUSION:
High (F) Value = Better Homogeneity + Easier re-dispersion.
Low (F) Value = Risk of “Caking” and inaccurate dosing.
Action: If (F) drops below the validated limit during IPQC, the formulation (viscosity or wetting agents) must be adjusted.
[02] IN-PROCESS QUALITY CONTROL (IPQC)
IPQC is the series of checks performed during the manufacturing process to ensure that every batch meets its specified requirements before it becomes a finished product.
Think of it as a “continuous physical exam” for a drug while it’s still on the assembly line. If a problem is detected during IPQC, the process can be adjusted or stopped before thousands of sub-standard tablets are produced.
THE THREE PILLARS OF IPQC:
Monitoring: Tracking environment conditions (humidity/temperature) and machine parameters (compression speed, mixing time).
Testing: Taking random samples from the production line at set intervals (e.g., every 30 minutes) to check for weight, hardness, or thickness.
Correction: If a sample fails a test, the “In-Process” nature allows operators to adjust the machinery immediately to bring the product back into specification.
Common IPQC Tests by Dosage Form
Dosage Form
Key IPQC Tests
Tablets
Weight variation, Hardness, Friability (Brittleness), Disintegration Time
Capsules
Shell moisture content, Fill weight, Locking length
Liquids/Suspensions
pH value, Viscosity, Sedimentation volume (F), Specific gravity
Semisolids (Creams)
Homogeneity, Particle size, Viscosity, Leakage test for tubes
FINISHED VS. IN-PROCESS:
While Finished Product Quality Control (FPQC) happens at the very end (the “final exam”), IPQC is “preventative maintenance”.
IPQC: Catches errors during the run (e.g., “The tablets are getting too soft, increase the pressure”).
FPQC: Confirms the batch is safe after it’s done (e.g., “The batch is perfect/failed, release/reject it”).