Organic Lecture 4/11
Enol and Keto Forms
Equilibrium Direction:
The equilibrium between the enol and keto form lies to the left, meaning there are more carbonyl compounds present.
Acidity:
pKa values for alpha hydrogens in carbonyls range from 19 to 20.
Hydroxide ions can abstract these protons, but not efficiently, leaving a significant amount of carbonyl intact.
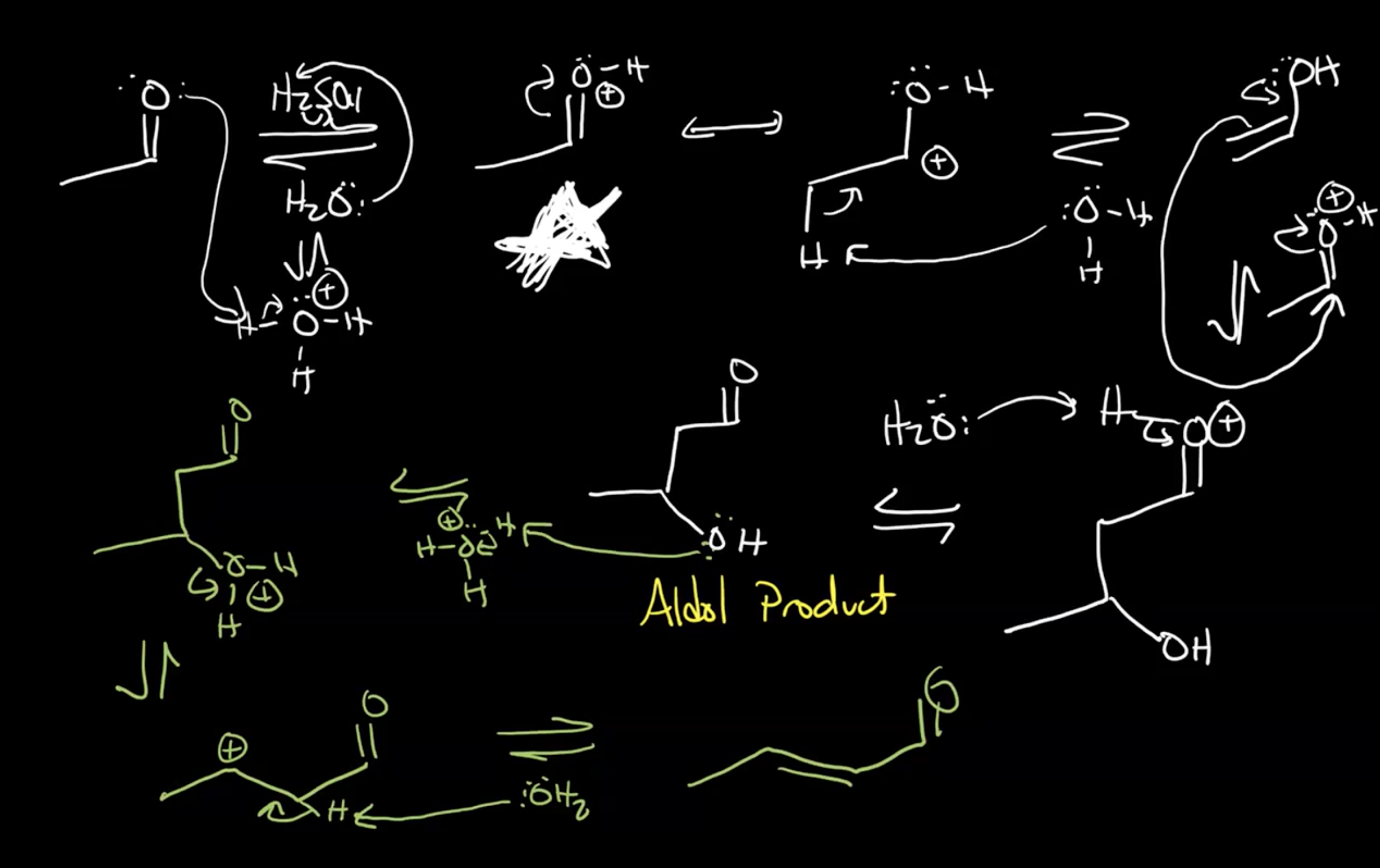
Mechanism Overview
Reaction Conditions:
The reaction can be conducted under basic or acidic conditions, with considerations for dehydration.
Temperature Effect:
Heating is necessary in basic conditions to promote dehydration since hydroxide is a poor leaving group.
In acidic conditions, the reaction proceeds to dehydration almost inevitably.
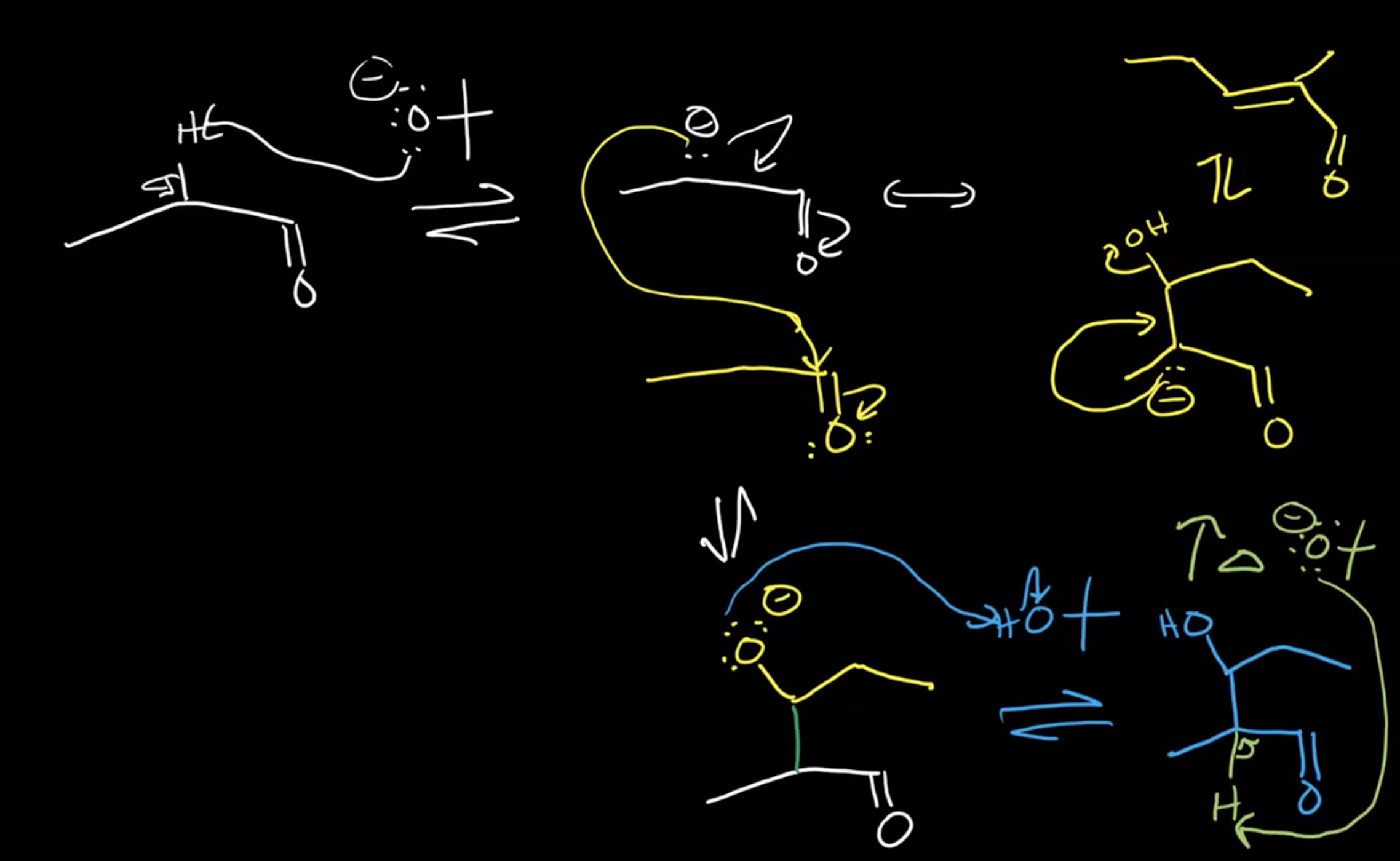
Aldol Reaction: Doing reaction with alpha hydrogen (next to carbonyl)
Formation of Larger Molecules:
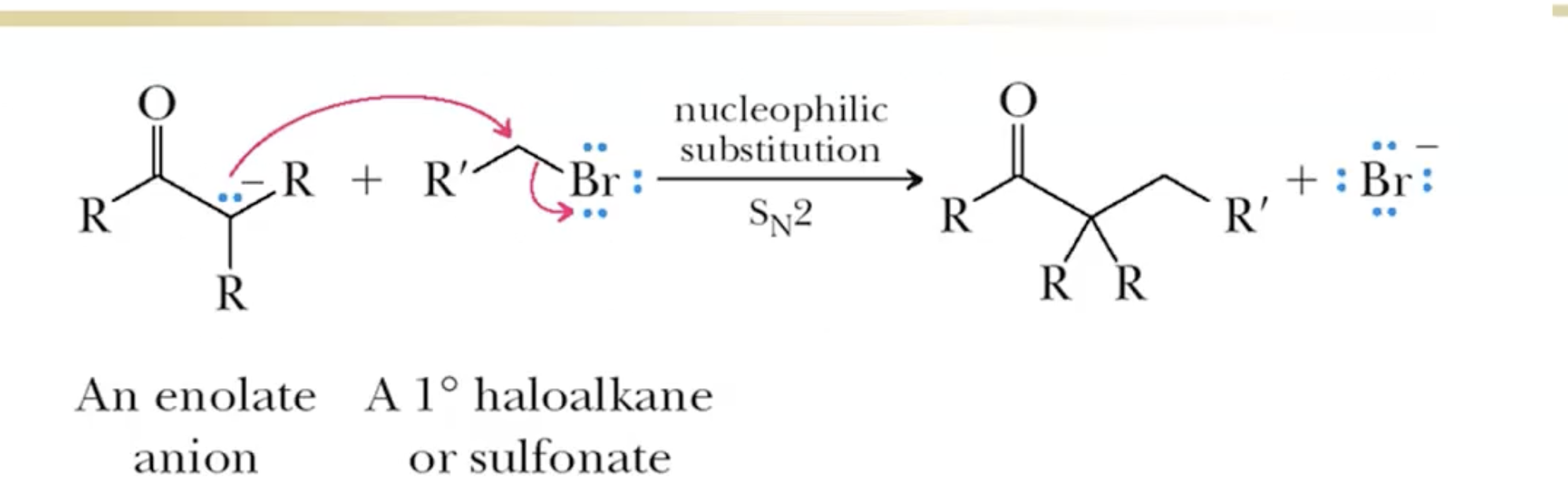
Removal of alpha hydrogen creates an enolate ion (anion of an enol)
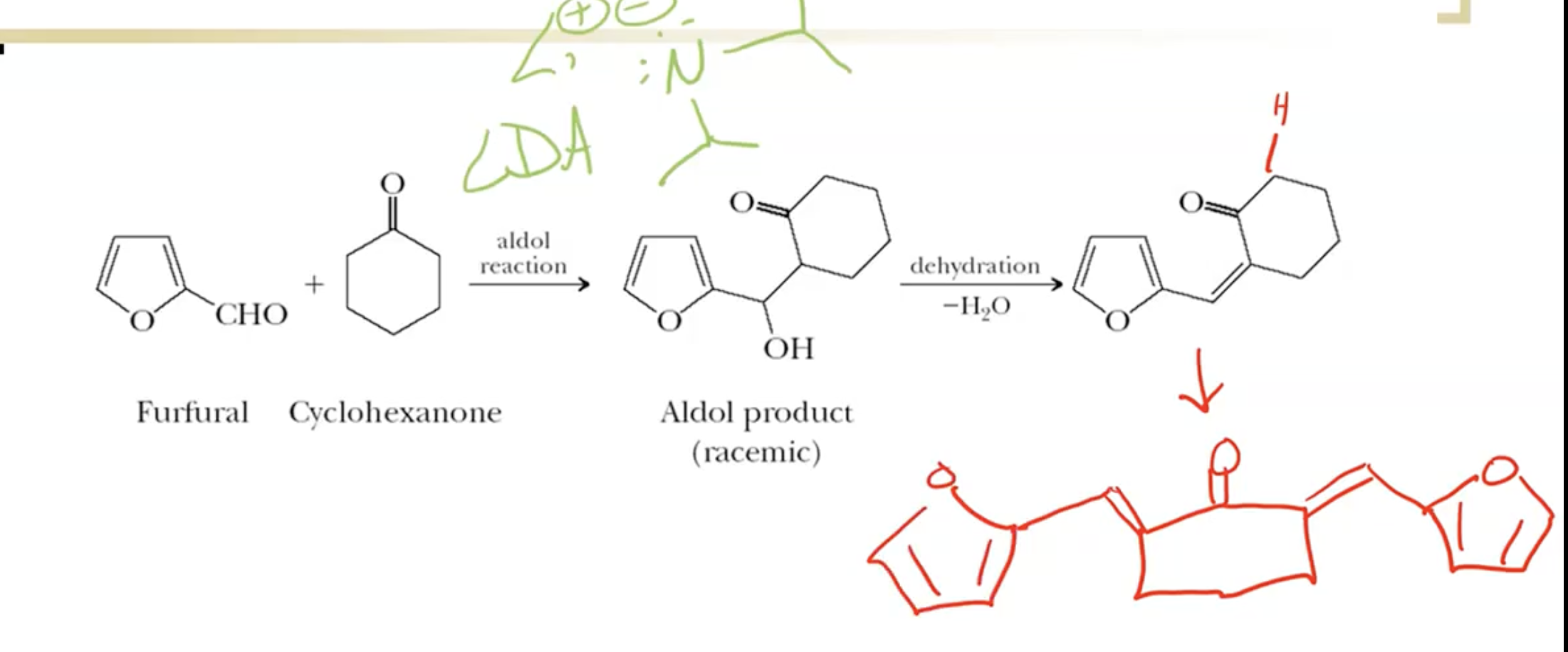
Enolates can act as nucleophiles to form larger molecules through aldol reactions, especially in five- to six-membered rings.
Enolate stabilized by resonance and formation is an equilibrium process
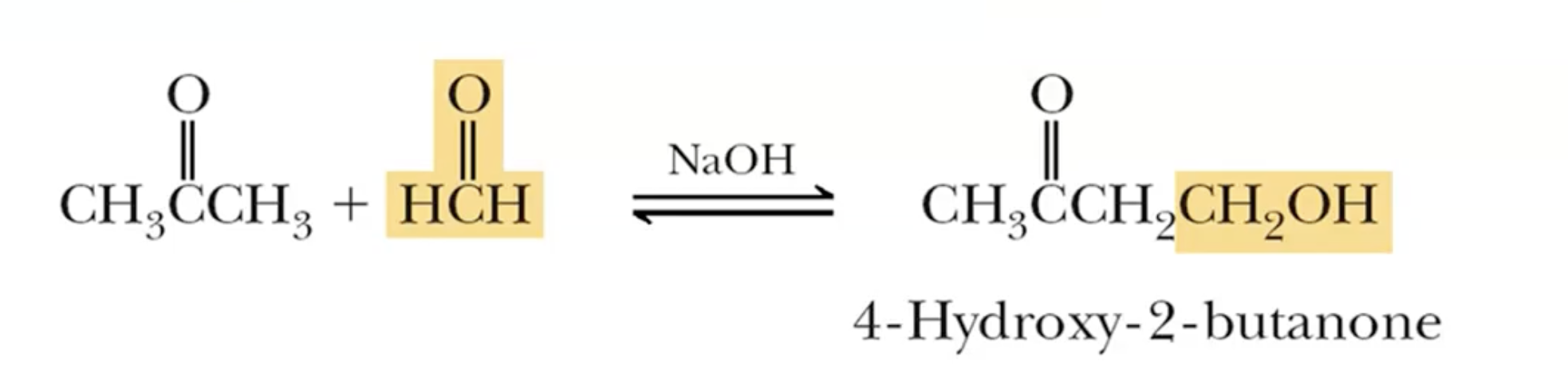
Cross Aldol Reactions:
Focus on minimizing self-aldol reactions by choosing reactants without alpha hydrogens, e.g., acetone for a formaldehyde reaction.
Works best if the carbonyl being attacked is an aldehyde
Using Strong Bases:
LDA (Lithium diisopropylamide) is a super strong base (pKa ~36) often chosen to promote enolate formation without competing self-aldol reactions.
If excess of the agent we’re attacking, we can do a double reaction (adding to each side). Minimize by using LDA to use 1:1 ratio, only getting single addition.
Enolate Reactions
Sn2
Carbonyl Addition (Aldol)
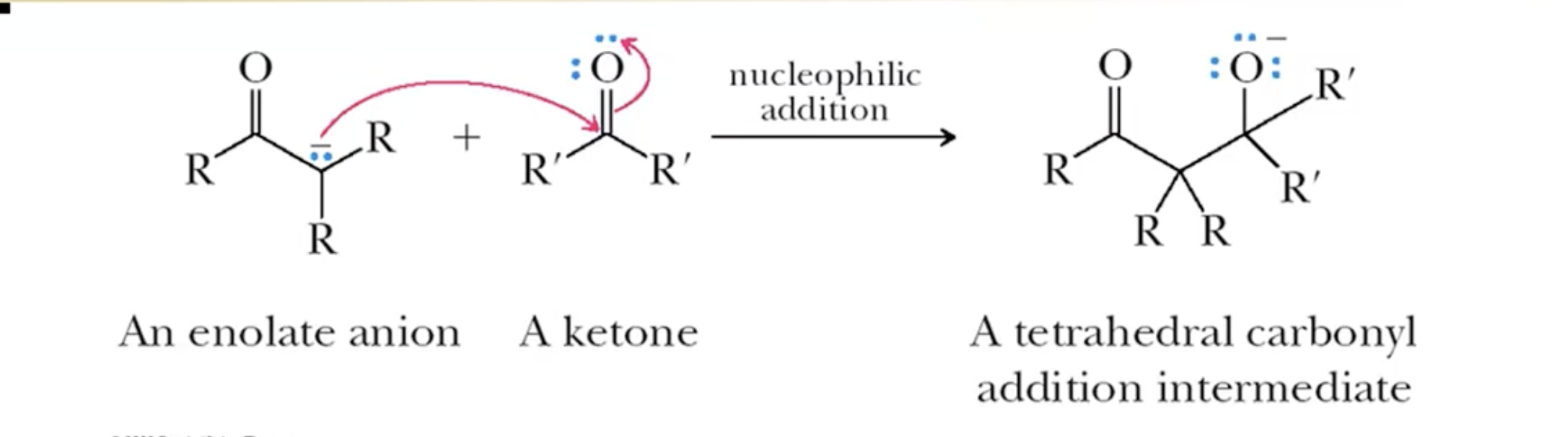
Basic conditions= require heat to get dehydration
Acidic conditions= hard not to do the dehydration
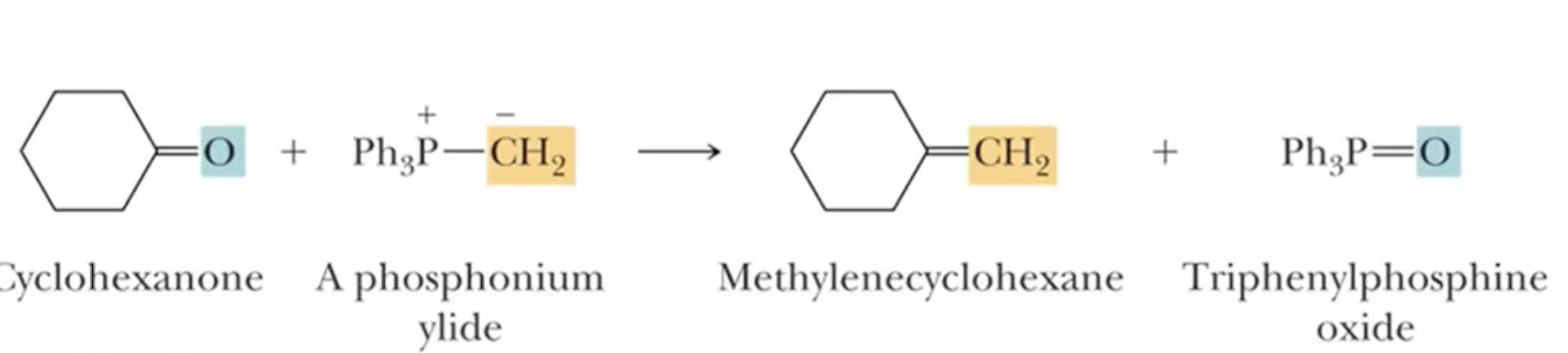
Wittig Reaction: Converts aldehydes and ketones to alkenes
Phosphonium Ylids:
The reaction involves a phosphonium ylide reacting with aldehydes or ketones to form alkenes.
The process replaces oxygen in the carbonyl with a double bond to the alkyl component from the ylide.
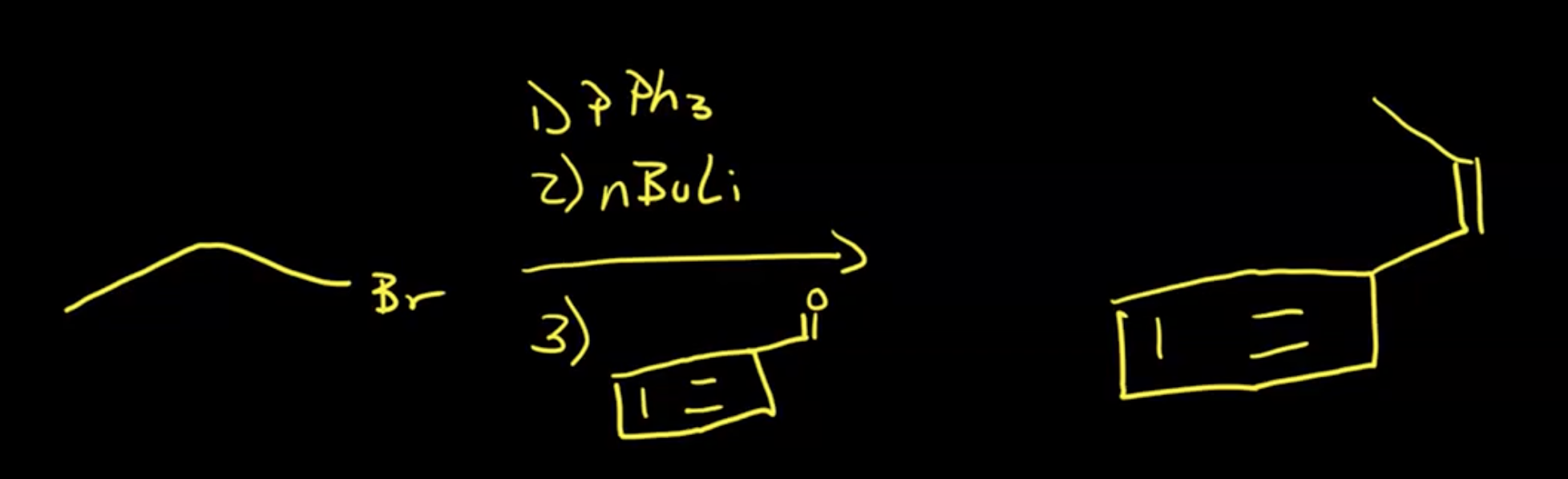
Written as CH3I —> 1. P(Ph3), 2. n-BuLi
Mechanistic Steps:
Start with triphenylphosphine for ylide creation through nucleophilic attack (Sn2) on primary/methyl/allyl alkyl halides.
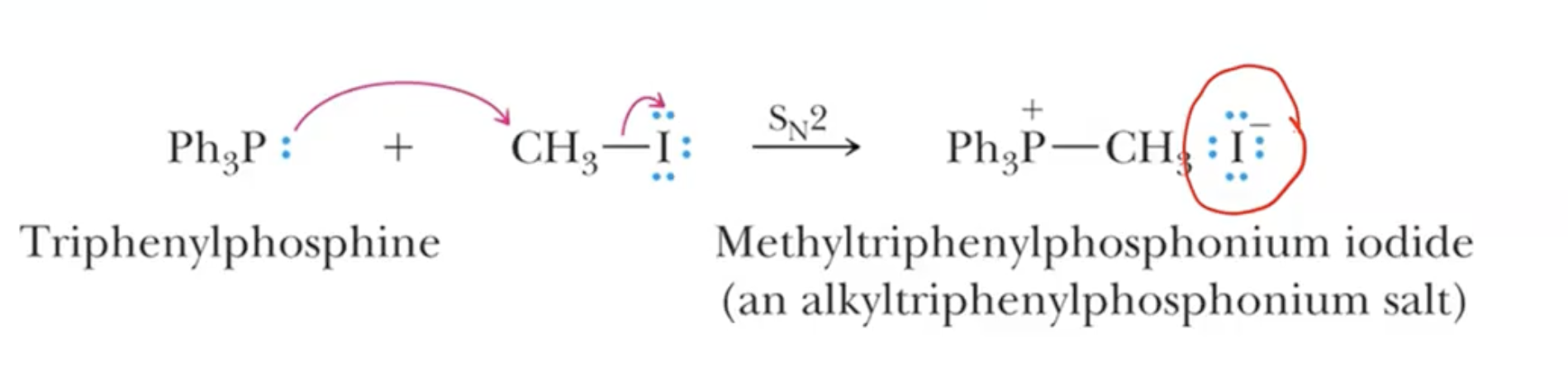
A strong base abstracts a proton, leading to the formation of the phosphonium ylide, which is resonance-stabilized.
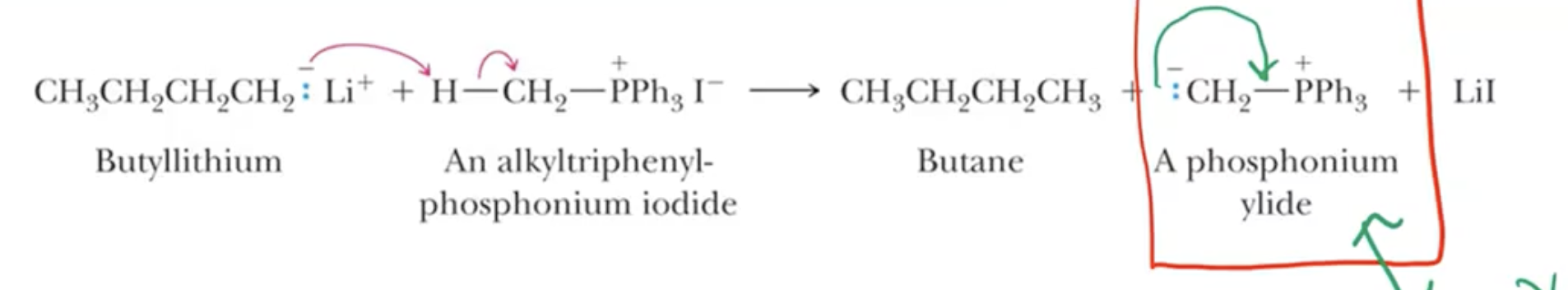
Reaction Dynamics and Intermediates
Intermediate Formation:
The proposed beta-aimine intermediates are highly controversial, as actual isolation has not been achieved.
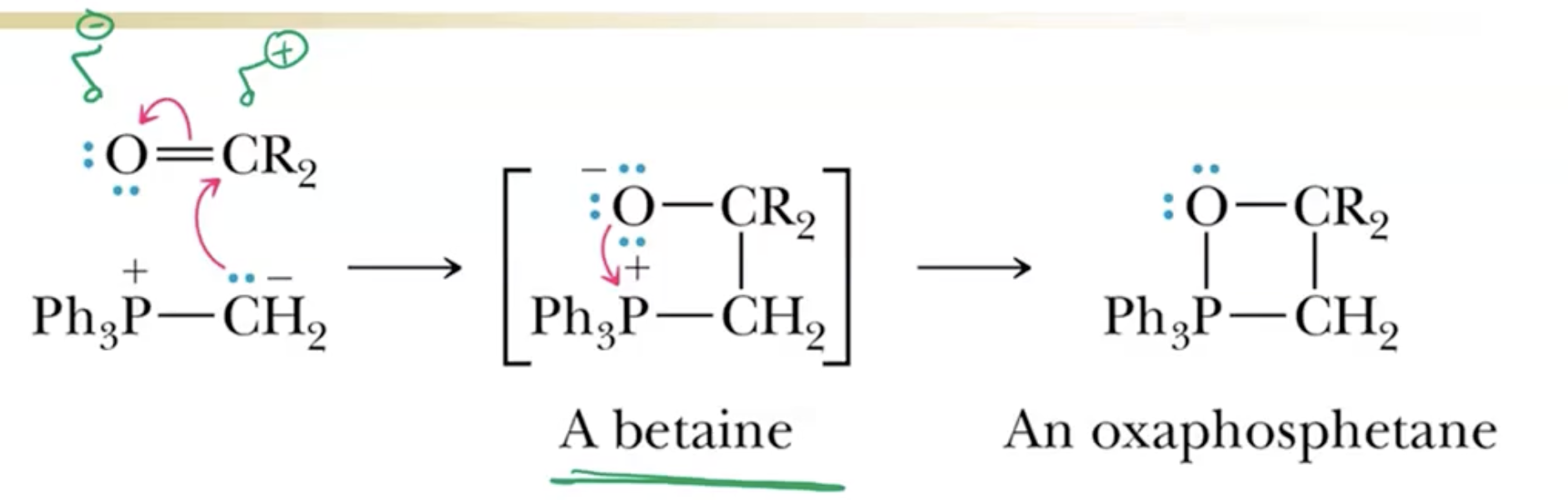
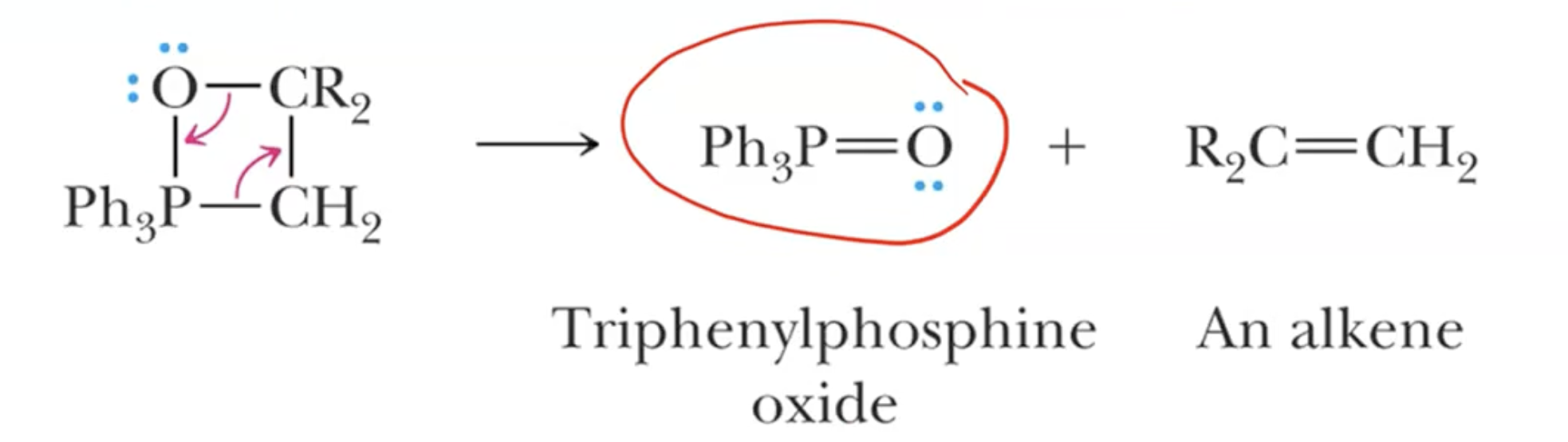
Mechanism Alternative Views:
Some chemists advocate for a direct pathway to the product rather than a two-plus-two cycloaddition scenario.
Mechanically, this reaction does not follow typical two-plus-two cycloaddition rules due to its lack of light requirement.
Low temperatures and don’t have electron withdrawing group on ylide= z product
Heat up and ylide has a electron withdrawing product= e product
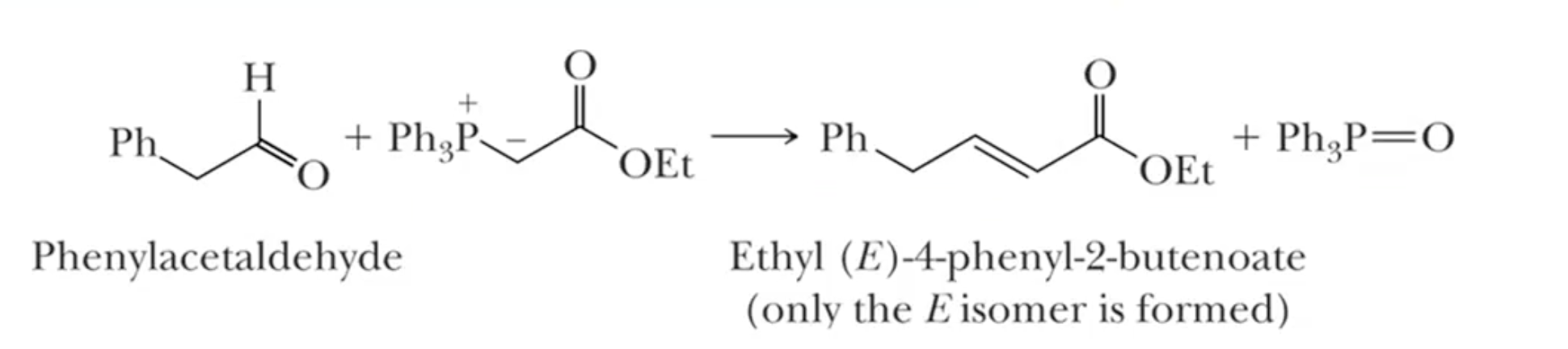
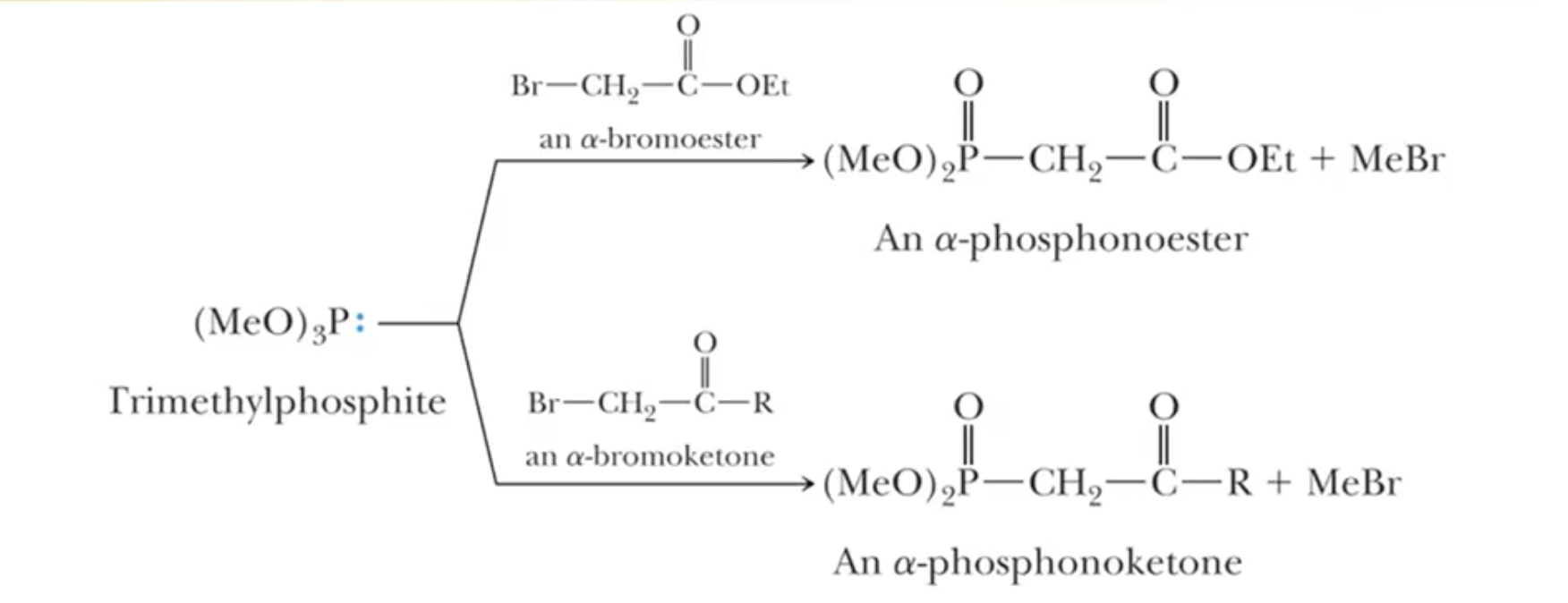
Horner-Emmons-Wadsworth Modification: similar to Wittig, but uses phosphonate ester instead of ylide
different solubility to make soluble in more polar substances
Almost exclusive to E selectivity due to EWG
Enolate Stability and Product Outcomes
Temperature Influence on Product:
Low temperatures yield the Z product, while higher temperatures favor E product formation unless an electron-withdrawing group is present.
Historical Context:
The Wittig reaction was discovered in the 1950s, with a Nobel Prize awarded in 1979, highlighting its prominence in organic chemistry synthesis.
Ongoing Developments:
The field of organic synthesis is continually evolving, with thousands of new reactions being discovered yearly.