Specific Heat Capacity
The thermal capacity of an object is the energy that must be supplied to raise it temperature by 1 degree Celsius
Heat received= mass (kg) x temperature change (degrees Celsius= 0C) x specific heat capacity (J/kg0C)
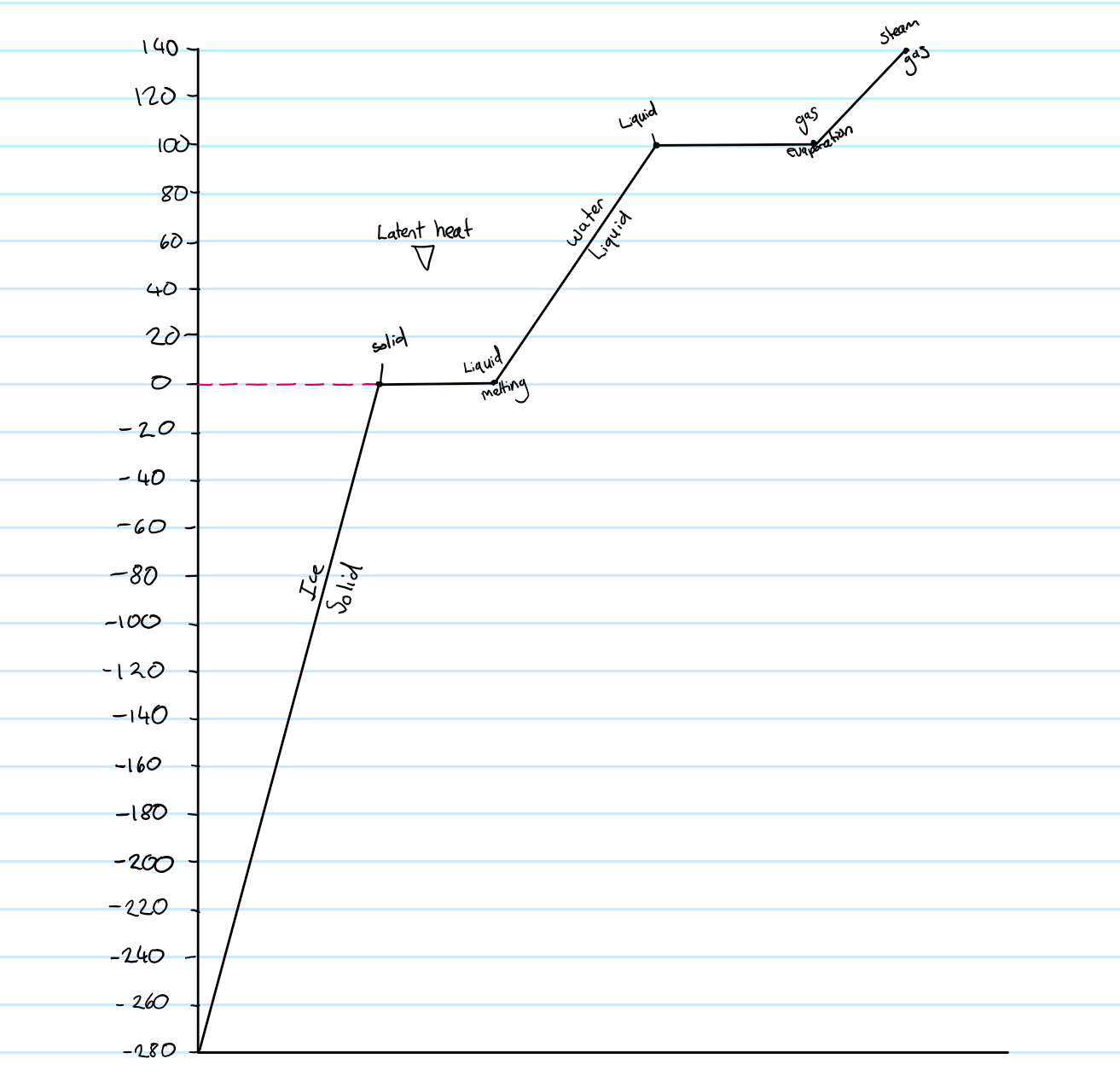
Specific Heat Capacity= Energy required to raise temperature of 1kg of substance by 1 degree Celsius