Lecture 12 - Nitrogen Metabolism
Learning Outcomes:
Quantify the inputs and outputs that represent daily nitrogen balance
Recognise the physiological periods in which amino acid processing is highest and lowest
Describe the basic principles of amino acid processing
Understand the significance of amino acid degradation enzymes having a high Km
Describe the central role of transaminases in the shuffling of amino groups during amino acid processing
Summarise the key features of the urea cycle
Conceptualise the relationship between the urea cycle and other central metabolic pathways
Understand why some amino acids are ketogenic and some are glucogenic
Appreciate the reasons why most amino acids need to be in the human diet
Predict the effects of purine synthesis inhibitors on cell health
Deduce the reasons for uric acid accumulation in cells experiencing chronic energy charge crisis
Most nitrogen in our body is wrapped up in out amino acids
Many in nucleic acids aswell
Amino Acids:
Twenty in proteins
More involved in metabolism
Lots of different side chains
Pathways of formation and disposal are varied and complex
Synthesis and degradation of proteins
Each under different control
hard to generalise
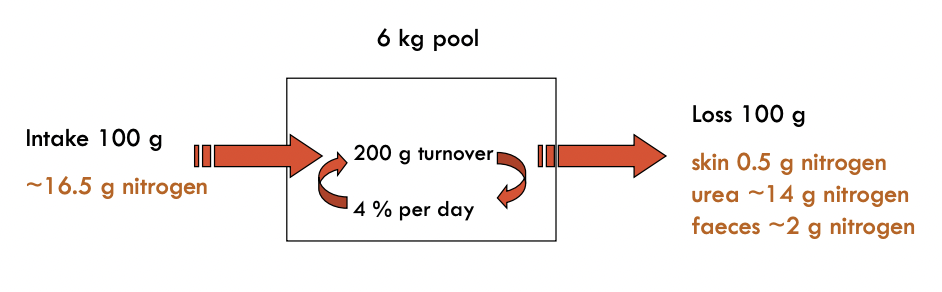
Daily Flux:
70 kg of male has 6kg of protein
Most of nitrogen is being lost to urea
Turnover = breaking polypeptide chain into amino acids and using amino acids to make another protein
Turnover varied massively between tissues: muscles 2%, intestine 15%
But muscle is greatest bulk of protein
When do we need to process Amino Acids?
During normal protein turnover
Released amino acids reassembled into protein
But many escape resynthesised
Oxidised in tissue or bloodstream
During starvation
When diet is rick in protein
How to deal with amine groups (ammonia is toxic)?
What to do with carbon skeletons?
Might get oxidised to CO2 for ATP
Or go to gluconeogenisis
Principles:
Liver is very important
First place amino acids go from the intestine (through hepatic portal vein)
Processing of amine groups
Urea synthesis
Conversion of amino acid backbones
Gluconeogenisis, lipogenesis, oxidation (we can not stor prroteins, so any excess amino acids MUST be degraded)
Not inter-conversion of backbone
Degradation enzymes have very high Km – not the rate limiting step
Not controlled
Only affected by [amino acids]
So excess amino acids are degraded
After Feeding:
Portal vein (into liver)
Amino acid mixture reflects protein composition of food we consumed
3/20 are branched chain amino acids (20%)
Hepatic vein (out of liver)
70% branched chain amino acids
Liver has preferentially kept the non-branched chain amino acids
Arterial circulation
Same as what went out of liver (previous point)
Vein from muscles
Branched chain amino acids removed
Muscles uses a lot of branched chain amino acids for energy
During Starvation:
Hypoinsulinemia stimulates proteolysis
Muscle released all types of amino acids
But disproportionately high amounts of alanine and glutamine
Branched chain amino acids used as fuel (leucine, isoleucine, and valine)
Alanine pick up amine group from branch chain amino acids, and others, and drop it back at the liver into the urea cycle, a pyruvate is left to enter gluconeogenesis during starvation
Prevents build up of amine n the muscles
Transamination
Shuffling of amino groups
Moving amine group to:
Pyruvate – alanine
2 α-ketoglutarate – glutamate
Oxaloacetate – asparate
To form amino acids
Amino acids can drop amino acids off at the urea cycle and allow amine group to be detoxified
Left over carbon skeletons can be used during starvation to fuel gluconeogenesis or to build fat in non-starvation periods
Urea Cycle:
Costs a lot of ATP to detoxify ammonia into urea
Similar to Krebs cycle
Not efficient
Original source of amine groups
Ammonia releasing reaction
Ammonia fixing reaction
Route of the carrier ornithine
Processing Skeletons:
Some feed into Krebs Cycle:
Anaplerotic – replenish
Can be diverted to gluconeogenesis
If liver transamination reaction in the liver
Some can only be made into acetyl CoA
Can only be used for FA synthesis or ketone bodies (ketogenic)
Acetyl CoA in the Krebs cycle can reduce our carriers and ultimately give ATP
Amino Acid Synthesis:
Very complex
Human can’t do most so we rely on bacteria for the amino acids we require
We can make:
Alanin
glutamate
Aspartate
Asparagine
Serine
Bacteria is required for the other which will steal from a central pathway in metabolism to produce amino acids
Protein Malnutrition:
If you do not have all 20 amino acids, you can not build a single protein
All the other amino acids then ‘in excess’
When a ribosome translates the mRNA, if theres no tRNA, it will stall, release and breakdown the half made protein
Other Nitrogenous ProductsL
Creatine
Non-peptide hormones (adrenalin)
Nucleotides
A single purine rings requires many amino acids, requires a lot of ATP
Cells do not want to synthesis too and break down too many as they require a lot of energy.
Inhibitor of Purine Synthesis:n
Cancer cell growing rapidly have a high demand for purine and pyrimidine synthesis
Needs billions of nucleotides to build one more cell – it will try and salvage most purines
Needs first enzyme PRPP to build purine
Drugs can inhibit PRPP and block de novo purine synthesis
Looks like a purine but the cell can’t use it
Doesn’t just kill cancer cells, but any cell trying to replicate – lots or terrible side effects
Purine Degradation
Cells dying release lots of purine and lots of energy
Puts the in an energy crisis – energy charge is very low
As a last resort, it can oxidise and breakdown the AMP
Force equilibrium to the right and have one final production of ATP
Lots of urate waste products

Lots of urate (waste products) (uric acid in blood stream) in chemo patients as cells are dying
Urate crystallises out in joints and causes gout
Inflammation occurs
Allopurinol (drug) replicated intermediates and lowers uric acid concentration
Seen in alcoholics as well