chemistry
Matter and its properties
What is matter?
matter is anything that has a mass and takes up space(volume)
all matter has two things in common: mass and volume.
mass: the amount of matter in an object or sample
units: kilogram(k), gram(g), miligram(mg)
volume: the amount of space that a material takes up
units: solid - cubic metres(m³), cubic centimetres(cm³)
gas or liquid - litres(L), mililitres(mL)
What are some ways to describe matter?(properties of matter)
matter can be described based on its physical properties and chemical properties
physical properties: characteristics of matter that can be observed or measured without changing its chemical identity
some examples include:
melting point: the temperature at which a substance changes from solid to liquid state
boiling point: the temperature at which a substance changes from liquid to gaseous state
malleability: describes how easily a substance can be hammered or bent into different shapes
hardness: describes the resistance of a solid to being scratched or dented
solubility: describes the ability of a substance to dissolve in a solvent
viscosity: describes how quickly a substance flows in liquid form
density: quantity of mass in a certain volume of material
chemical properties: ability of matter to react with another substance to form one or more new substances
can only be observed when a substance chemically interacts or reacts with another substance
some examples include:
reactivity with acids: some substances react vigorously with acids and others do not. Ex. baking soda and vinegar produce a gas
reactivity with oxygen: substances in some objects react with oxygen when exposed to air. Ex. avocadoes turning brown, rusting nails
combustibility: ability of material to catch fire and burn in the air. Ex. burning wood
lack of reactivity: substances that do not react with other substances are “inert”. Ex. helium in balloons
Matter also undergoes changes: physical change vs. chemical change
physical change: change of matter that does not change its chemical identity or composition
some examples include:
freezing of water(liquid) to form ice(solid)
Liquid water(h2o) → solid water(h2o)
cutting a paper into pieces
chemical change: change of matter that produces one or more products. When a new chemical substance is created, it is a chemical change
some examples include:
2h + o → h2o (formation of water)
toasting bread(evidence of new substances forming: colour, smell of bread change when you toast it)
how do we know a new chemical has formed?
Here are some clues:
a new substance or material appears
emission, sound or light are given off
bubbles of gas are formed(not boiling water)
a solid material appears when two liquids are mixed(a precipitate)
Two different types of matter data: qualitative vs. quantitative data
qualitative data: can be observed and compared using words(subjective)
some examples include:
sight
odour
texture
state
etc.
quantitative data: can be measured and recorded by using numbers(objective)
some examples include:
there are 8 dolphins in this pod
the boiling point of water is 108C
the dolphin is 1.45m in length
Classifications of matter
matter can be classified as:
mixture: can be separated into parts by physical changes
eg. trail mix, salt water
pure substances:
elements: cannot be separated or broken down by physical or by chemical changes
compounds: can be broken down into 2 or more elements by chemical changes but not by physical changes
eg. h2o → 2h + o
Measuring volume and density
volume: the amount of space taken up by a substance or object
volume of solid measured in cubic centimetre(cm³) or cubic metre(m³)
volume of liquid + gases measured in litres(L) or millilitres(mL)
volume is determined by:
viewing the measurement of a liquid in a container(mL)
volume of a rectangular prism(cm³) = length(cm) * width(cm) * height(cm)
using a displacement method in a container to measure and irregular shaped object(mL)
volume of solid + (volume of water + solid) - (volume of water)
measuring volume:
we use volumetric/graduated flasks to measure volume precisely
when we read the reading, we look at the lowest point of the meniscus
metric system:
an international system of measuring(based on multiples of 10):
m,mm,km for length
grams for mass
secs for time
mL, cm³ for volume
g/mL, g/cm³ for density
celsius for temperature
fluids and density
a fluid is any form of matter that can flow
liquids and gases are fluids since they do not have a fixed shape or volume
solids are not fluids as they have a fixed shape
density of matter(solids, liquids, and gases)
density is the amount of material(mass) for each unit of space it occupies(volume)
density and layers of fluids
fluids that do not mix separate themselves according to their density
less dense fluids settle on top of more dense fluids
density of water is 1g/mL
units for measuring density:
fluids: g/mL
solids: g/cm³
calculating density
density = mass/volume
practice question:
Q. What is the density of a 4cm³ rock that has a mass of 24g?
A. D = (mass/volume) = 24g/4cm³ = 6g/cm³
Q. a 5mL sample of motor oil has a mass of 4.5 g. What is the density of the motor oil?
A. D = mass/volume = 4.5g/mL = 0.9g/mL
Buoyancy
buoyancy is a physical property that is directly proportional to the density of the fluid in which an object immersed(in a fluid)
it is the tendency to rise or float in a fluid
you can think of it as the “upward thrust” or “upward force”
Viscosity
viscosity is a physical property that can be used to describe liquids
it is the measure of resistance to deformation at a given rate
you can think of it as the “thickness” of a liquid
states of matter
matter is made up of tiny particles in atoms and molecules
matter can be found in 4 states: solids, liquids, gas, and plasma
solid | liquid | gas | plasma | |
example |
|
|
|
|
properties |
|
|
|
|
Kinetic molecular theory
a model is a verbal, mathematical or visual representation of a scientific structure or process
theory is a scientific explanation that has been supported by consistent repeated experimental results and is therefore accepted by most scientists
the scientific explanation of the behavior of matter based on all matter being made of particles that possess kinetic energy is called Kinetic Molecular Theory of Matter(KMT)
kinetic energy is the energy of motion
even though we cannot see with out naked eyes, all particles are always moving and therefore possess kinetic energy
energy makes particles move. the more energy the particles have, the faster they can move and the further apart they can get
particles stop moving at zero Kelvin(K), which is -273C
states of matter | KMT states… |
particles in solid | particles are very close together particles vibrate but cannot move around particles attract one another strongly in a rigid structure |
particles in liquid | particles are also very close together but can move around particles slip and slide past and revolve around each other but stay close together they attract one another less strongly than in solids |
particles in gas | particles are very far apart compared to their size move around randomly and quickly in straight lines attraction to one another is effectively zero |
particles are constantly moving
energy makes particles move
the more kinetic energy the particles have, the faster they move and the farther apart they get
more energy → faster movement → particles move farther apart
gas > liquid > solid
more kinetic energy → less kinetic energy
The effect of temperature on matter

changes of state occur when matter transforms from one state to another
example: liquid(water) to solid(ice)
when temperature increases, particles gain energy and move faster and further apart
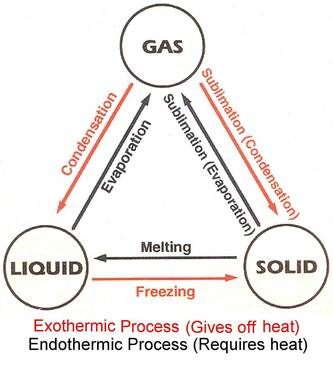
liquid to gas - evaporation/vaporization
gas to liquid - condensation
liquid to solid - freezing
solid to liquid - melting
gas to solid - deposition
solid to gas - sublimation
changes of state and temperature
temperature is the measure of the average kinetic energy of particles in a substance
increasing the temperature of matter means the particles of the matter are gaining energy
once the matter reaches a certain temperature, the particles have gained enough energy to change state
the temperature at which a substance melts is called its melting point(solid to liquid)
the temperature at which a substance boils is called its boiling point(liquid to gas)

KMT explains diffusion and thermal expansion/contraction
diffusion is the movement of particles from an area of high concentration to low concentration
solids liquids and gases expand when heated and contract when cooled
thermal expansion is the increase in volume of a substance when its temperature is increased
heating increases kinetic energy of particles
thermal contraction is the decrease in volume of a substance when its temperature is lowered
cooling decreases kinetic energy of particles
The atom
the smallest particle of an element is an atom
the atom is made up of
the nucleus which is made up of protons and neutrons
electrons
The atomic theory: all matter is made up of particles called atoms
what is a nucleus?
the center region of an atom is the nucleus
the nucleus of most hydrogen atoms contain only 1 proton
the nucleus of all other atoms contain both protons and neutrons
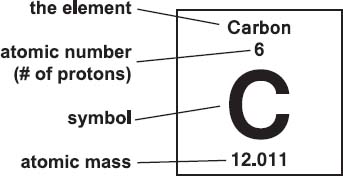
what is the mass number?
the atomic mass is the total number of protons and neutrons
what is a proton and an electron?
a proton is a positively charged subatomic particle while an electron is a negatively charged subatomic particle
the number of protons in a nucleus determine the atomic number of the nucleus and the identity of an atom
protons are also called “elementary particles”
the region that electrons occupy is called energy shells, which surround the nucleus
an electron is like a spread out cloud of high energy that exists in the whole region. it is also called an electron cloud
electrons do not have a mass → not matter
protons, electrons and neutrons are classified as subatomic particles
for a neutral(not charged) atom, the number of protons is equal to the number of electrons.
in this case, the number of electrons in a neutral atom is 6
what is a neutron?
a neutral subatomic particle found in the nucleus
summary:
name | symbol | electric charge | location in the atom |
proton | p+ | positive(+) | nucleus |
neutron | n0 | neutral/none | nucleus |
electron | e- | negative(-) | energy shells around the nucleus |
atomic number = no. of protons
atomic mass = no. of protons + no. of neutrons
neutral atom = no. of protons = no. of electrons
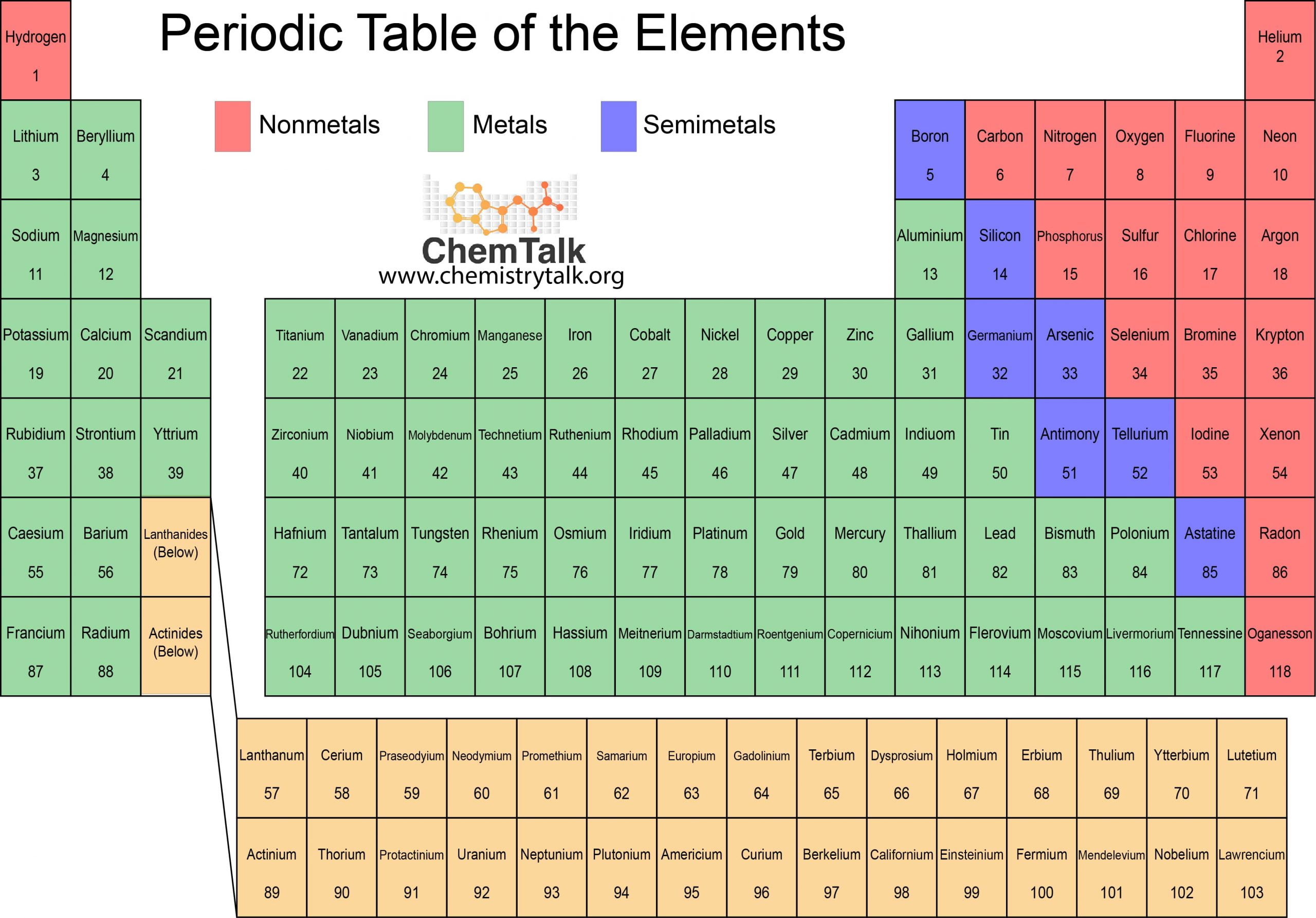
periodic table
as chemists discovered more elements, they saw the need of organizing all the elements in a consistent way, hence the periodic table
development of the periodic table
by arranging elements in order of increasing atomic mass into columns, Dmitri Mendeleev created the first periodic table
as more and better technology became available, chemists made a significant change to Mendeleev’s method of organizing the elements. the modern periodic table is organized according to the atomic number rather than the atomic mass
why is the periodic table important?
the table acknowledges the existence and properties of all elements
what are elements, compounds and mixtures?
elements are composed of:
only one type of atom
atoms can exist individually or in compounds(molecules)
cannot be separated or broken down by physical or chemical changes
compounds: can be broken down into two or more elements by chemical changes but not physical changes
mixtures: can be separated into parts by physical changes
the major divisions within the periodic table
the set of all elements in a given row going across the table is called a period
the row shows the number of energy shells the element has
the set of all the elements in a given column going down the table is called a group/family
they have the same number of electrons on the outermost shell, hence they have similar chemical properties
group 1: alkali metals
group 2: alkaline earth metals
group 17: halogen
group 18: noble gas - they are the most stable because they have full energy shells
elements on the periodic table are divided into three kinds
example of metals: group 1(sodium), group 2(calcium) and transition metal
example of non-metals: noble gases, halogens and oxygen
example of metalloids: baron, arsenic and silicon(which means they have properties that are a mix between those of metals and solid non-metals)