Effect of Surface Area on Rate
Collision Theory Recap
Reactions only happen when particles collide with enough energy.
Rate of reaction depends on the frequency of successful collisions.
Frequency = number of successful collisions per second.
Effect of Surface Area
Only the particles on the surface of a solid can collide with particles in solution (or gas).
Larger solid blocks → fewer surface particles exposed → slower reaction.
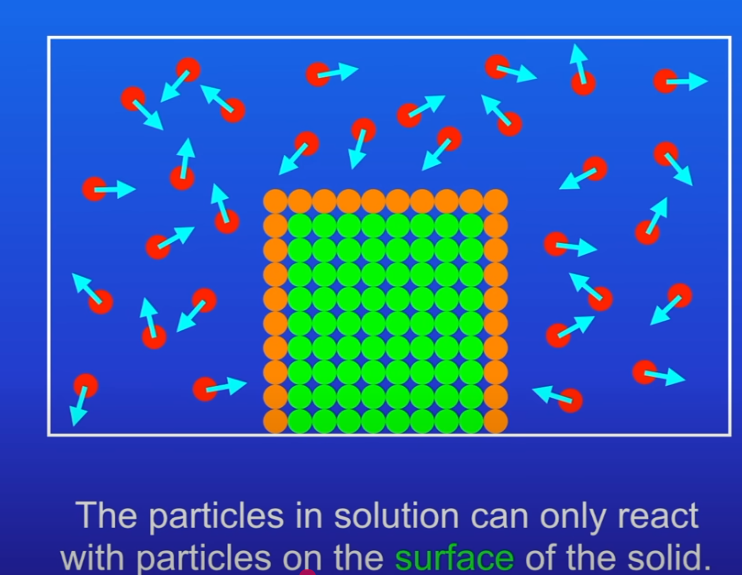
Smaller pieces / powdered solids → greater surface area-to-volume ratio (SA:V) → more exposed particles → more collisions per second → faster reaction.

Practical Example: Marble Chips + Hydrochloric Acid
Reaction:
Calcium carbonate (s) + Hydrochloric acid (aq) → Carbon dioxide (g) + Calcium chloride (aq) + Water
Method 1: Measuring Gas Volume
Conical flask with marble chips + HCl.
Delivery tube into water trough + measuring cylinder (or gas syringe).
Record volume of CO₂ produced over time.
Repeat with different sizes of marble chips (large vs. powdered).
⚠ Issue: Measuring cylinder can be inaccurate if bubbles form too quickly. → Gas syringe is more precise.
Method 2: Mass Loss
Conical flask on a balance.
Reactants: marble chips + HCl.
As CO₂ gas escapes, mass decreases.
Plot mass lost over time.
Use cotton wool plug in flask:
Allows gas to escape.
Stops acid splashing out (which would give anomalous results).
Key Points for Exams
Increasing surface area = faster reaction (more frequent collisions).
Smaller particles → higher SA:V ratio.
Graphs:
Steeper curve = faster reaction.
Reaction with powdered chips finishes quicker (but both produce the same final amount of product if the same mass of CaCO₃ is used).
✅ Conclusion:
The rate of reaction increases as the surface area of solid reactants increases, due to more collisions per second.