R3.4 Electron-pair sharing electrons (PART 2)
Bronsted-Lowry Theory Recap
Acid - proton DONOR
loses hydrogen
Base - proton ACCEPTOR
gains hydrogen
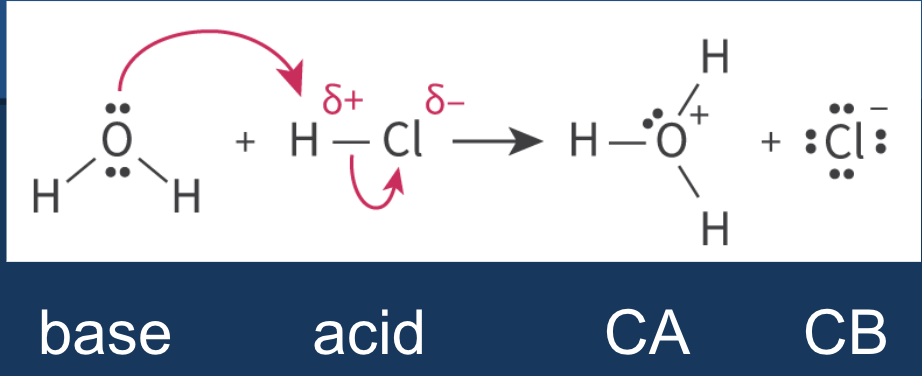
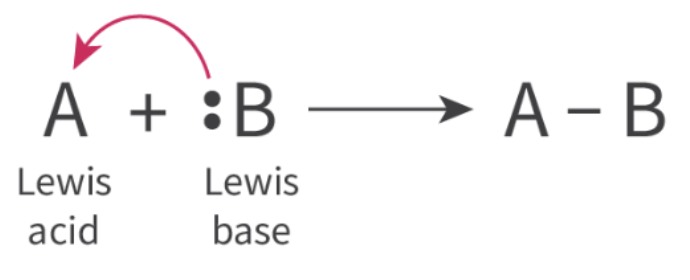
Lewis acid base theory - defines acids/bases in terms of electron pair transfer
Lewis acid - electron pair ACCEPTOR
gains electrons
Lewis base - electron pair DONOR
loses electrons
applies to both organic + inorganic reactions
Inorganic
metal cations are lewis acids
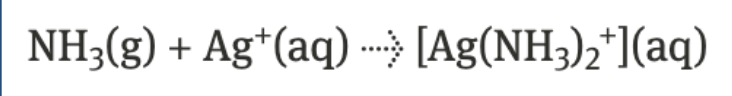
organic
electrophiles (lewis acid) - gains electrons
electrophile - electron deficient + attracted to electron rich
H+
Br+
NO2+
nucleophiles (lewis base) - loses electrons
nucleophile - electron rich + attracted to electron deficient
H2O
OH-
NH3
X-
CN-

Lewis theory is broader than bronsted lowry theory
can be applied to wider range of chem reactions
All BL acids are Lewis Acids
BUT not all lewis acids are BL acids
all BL bases are lewis bases
BUT not all lewis bases are BL bases
lewis acid reacts w lewis base → coordinate covalent bond
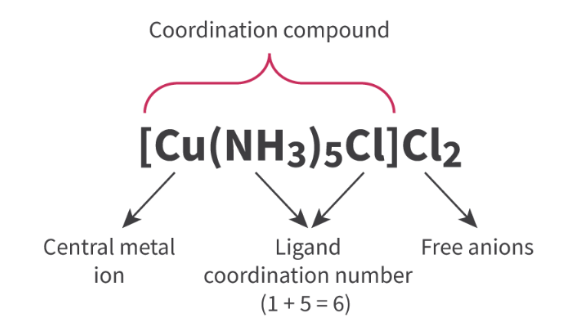
complex ions
transition metal + ligand
formed through lewis acid-base reactions
Coordination # - number of coordinate covalent bonds on central metal ion
charge - overall charge of complex ion
sum of cation + ligand charges
Alkenes - double bonded molecules
unsaturated - double bond
double bond = nucleophile
high electron density
attracted to electrophiles
sigma bond - the first bond formed btwn 2 atoms
pi bond - the double/triple bonds but not the sigma bond
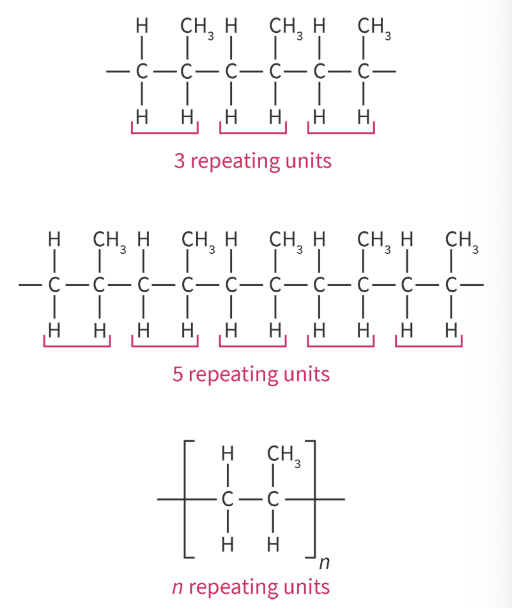
Polymers - large molecules (macromolecules) made from monomers
monomers - repeating subunits
classified into 2 main categories
natural - found in nature
sugars, proteins, DNA, silk, cellulose
play crucial role in biological processes
synthetic - human made
plastics, clothing
created through polymerization
addition polymers → plasics
condensation polymers → clothing
properties of plastics
flexible
durable
moldable
lightweight
electrical/thermal insulators
physical properties of polymers
determined by nature of monomer + way they are linked together
intermolecular forces
Addition polymerization - breaking a double bond in each monomer to create new bonds linking the monomer units together
monomer must be an alkene
pi bond broken in each monomer → single bond forms btwn monomers
represent the repeating unit of an addition polymer when given the monomer structure
structural formula of a specific number of repeating units
OR repeating units in brackets with subscript of n
deduce monomer for addition polymers when given polymer structure
monomer is ALWAYS an alkene
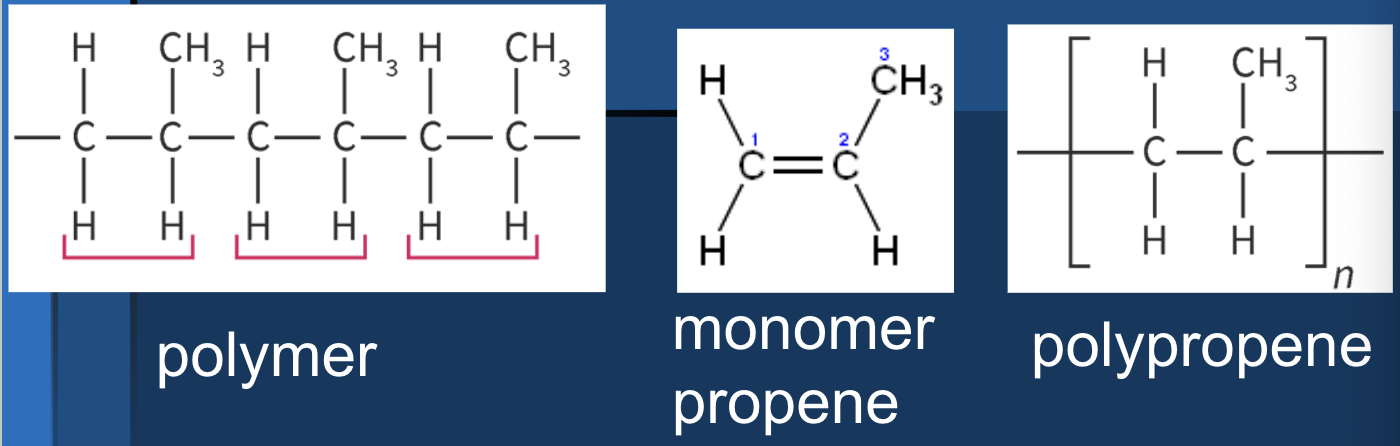
naming polymers
determine polymer structure (alkene)
name the monomer via IUPAC
put poly in front
electrophilic addition to alkenes
double bond in alkene breaks
2 additional atoms bond with the molecule
1 atom bonds to each carbon in the double bond
product = saturated compound
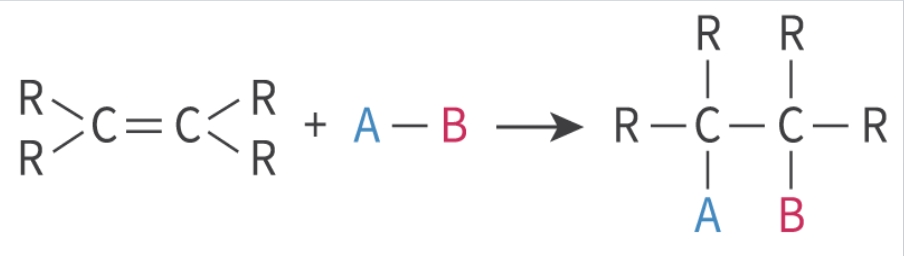
types of electrophilic addition to alkenes
halogenation - addition of halogens (X2)
forms di-halogenoalkane
hydrohalogenation - addition of HX
forms halogenoalkane
hydration - addition of water
forms an alcohol
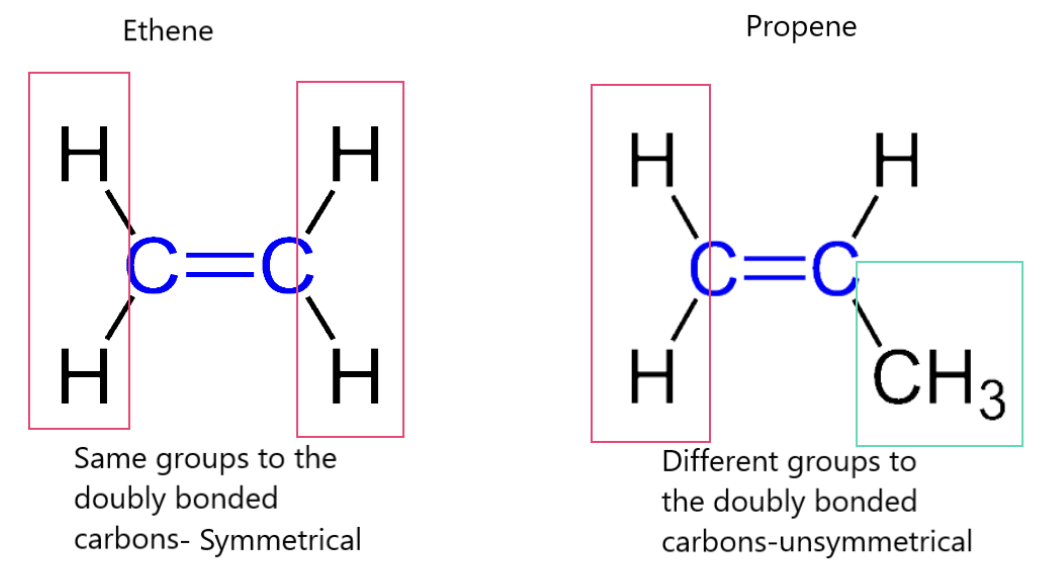
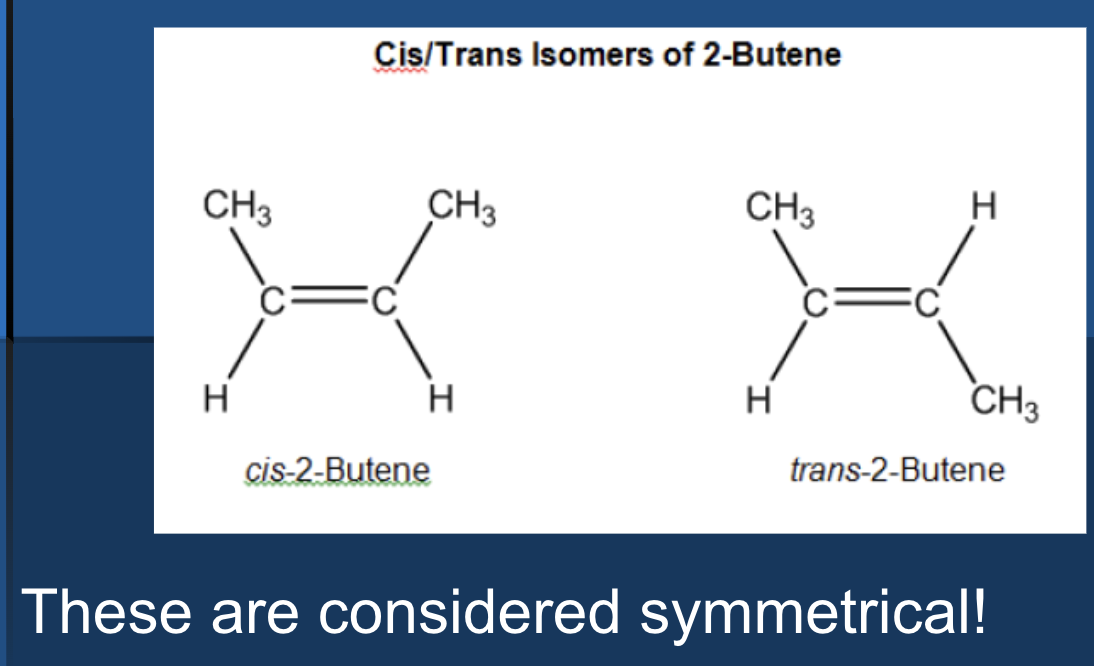
symmetrical alkenes
identical substituent groups attached to carbons
double bond = line of symmetry
mechanisms are drawn
asymmetrical alkenes
different substituent groups attached to carbons
no line of symmetry
products are predicted
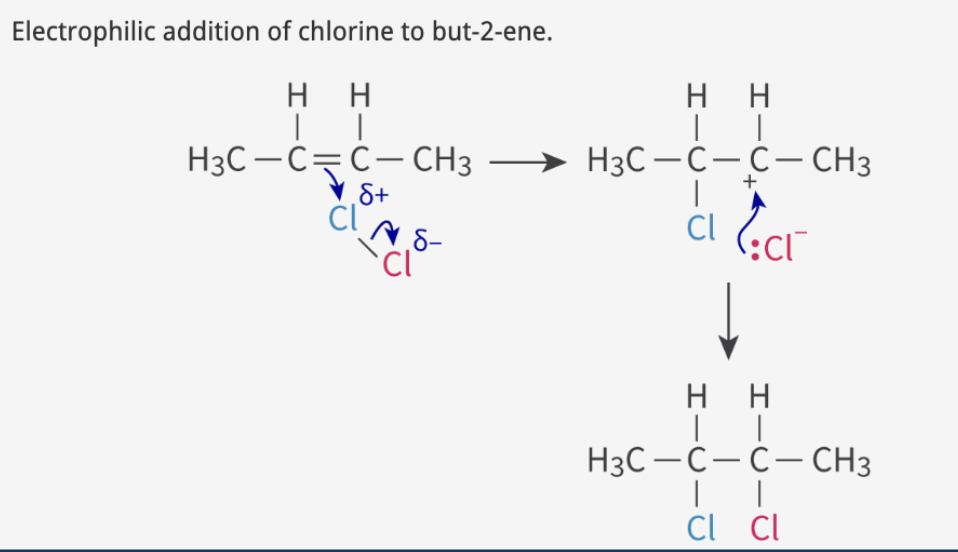
Halogenation: alkene + H2
Step 1
double bond e- induces dipole on X2 + attack electrophilic end
electrophilic end = partially positive end
forms carbocation intermediate + negative halide ion
Step 2
halide ion attacks carbocation
If 2 different halogens (ex. ClF) → less electronegative halogen binds to carbon w most hydrogen
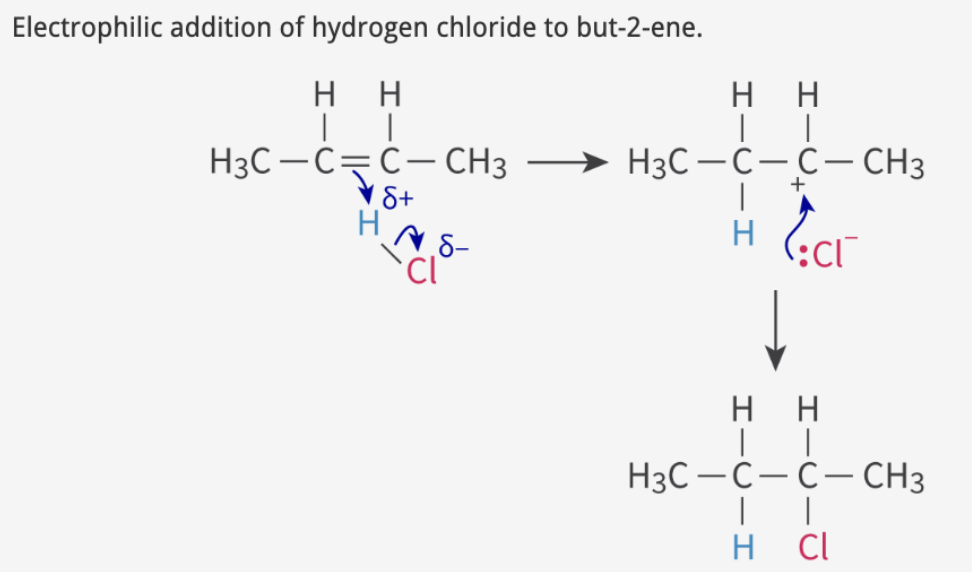
hydrohalogenation: alkene + HX
Step 1
double bond e- attacks H
polar bond where H is partially positive
forms carbocation intermediate + negative halide ion
Step 2
halide ion attacks carbocation
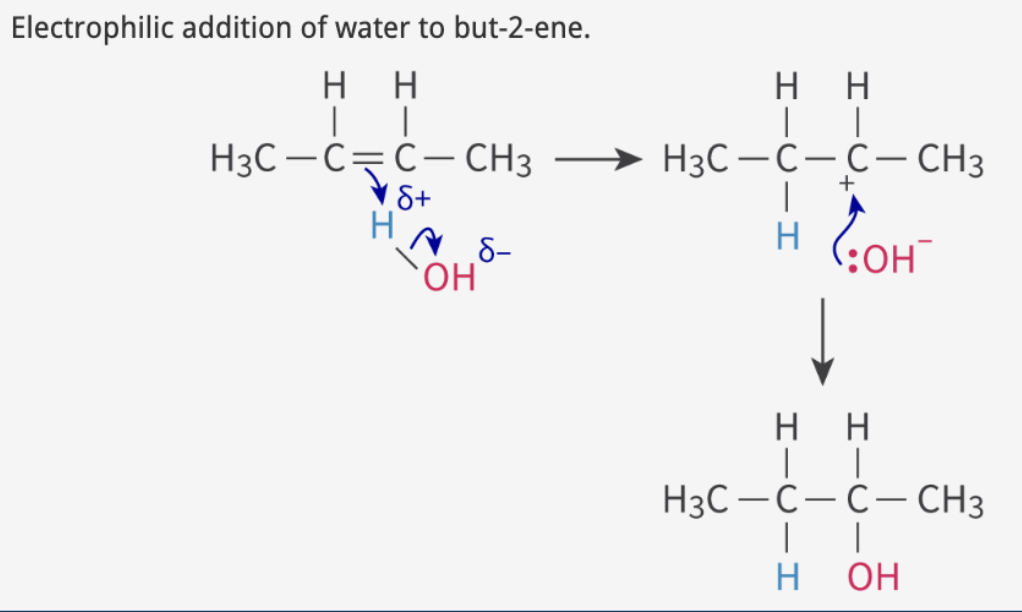
hydration: alkene + H2O (water)
Step 1
double bond e- attacks H
polar bond where H is partially positive
forms carbocation intermediate + hydroxide ion
Step 2
hydroxide attacks carbocation
Unsymmetrical alkenes
unsymmetrical alkenes react with HX OR H2O → 2 products can form
how to determine major product
stability of carbocation intermediate
more stable carbocation intermediate is favored → most stable is major product
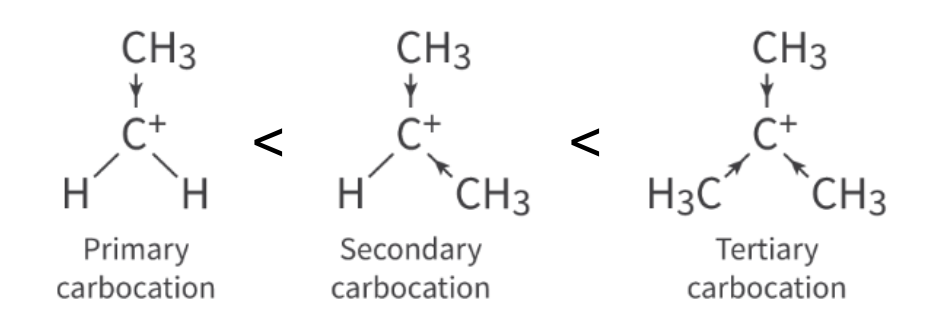
major product can be predicted using Markovnikov’s Rule
Markovnikov’s rule - hydrogen atom will attach to the carbon w more hydrogens
Benzene C6H6 - most common aromatic compound
cyclic
planar
3 double bonds in resonance
high electron density
acts as nucleophile
attracted to electrophiles
extremely stable
delocalized electrons = resonance = stability
double bonds do not want to break
Favors substitution over addition reactions
electrophilic substitution
high activaction energy for reaction
unstable carbocation intermediate
final product is stable
aromaticity + resonance is re-established when H is lost
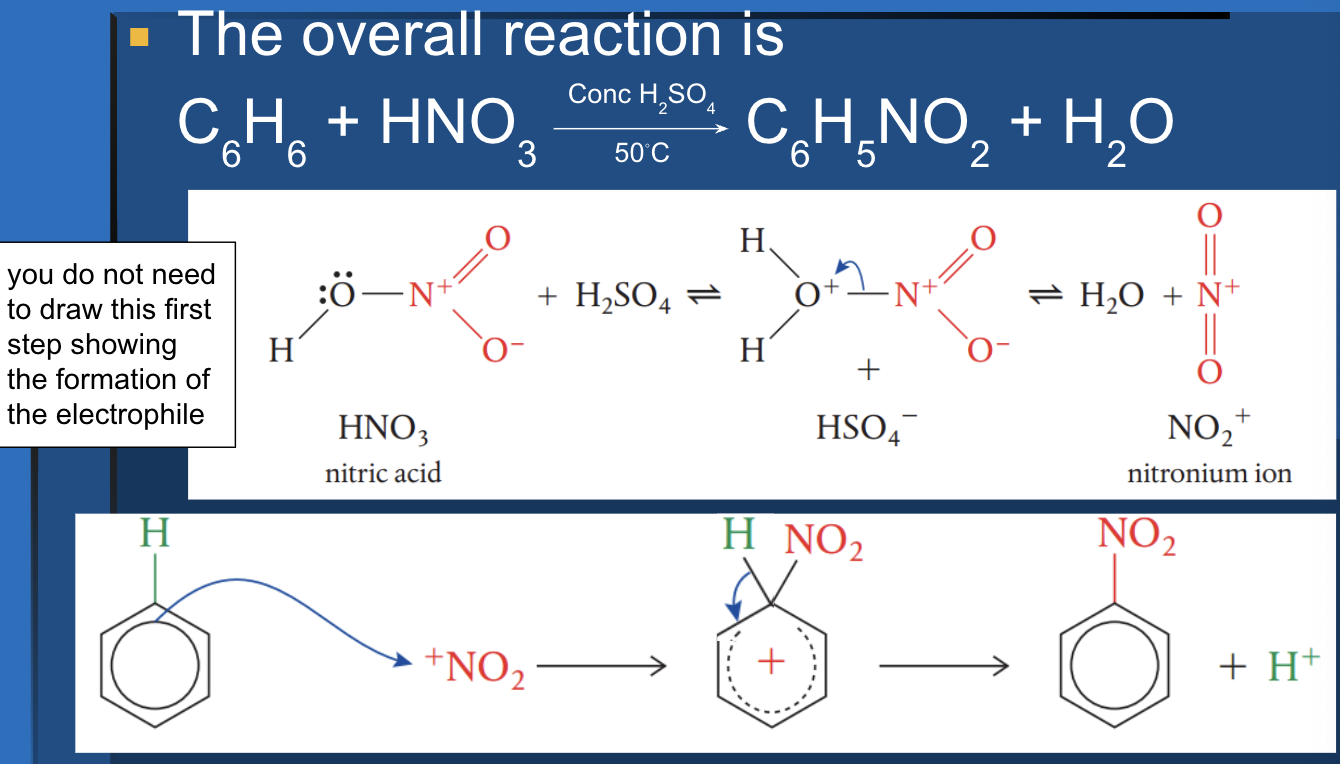
electrophilic aromatic substitution mechanism
Step 1
E+ added to ring → loss of aromaticity
Step 2
removal of H → reconstitutes aromatic ring
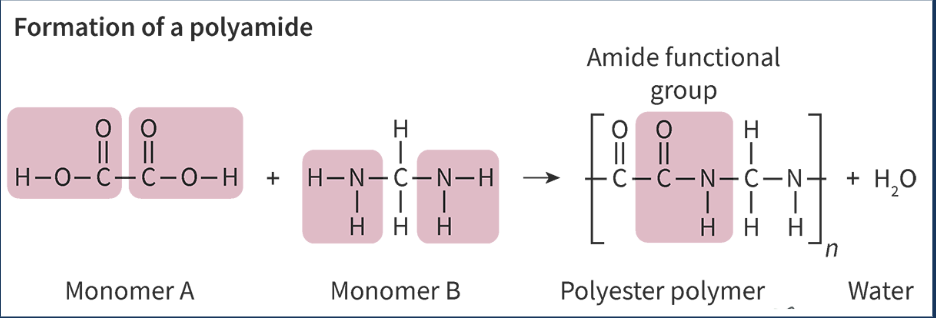
condensation polymer - polymer formed by reaction btwn 2 functional groups with the release of a small molecule
small molecule is usually water
categorized according to the functional group present on repeating unit
IB requires to draw the repeating unit of polyesters/polyamides when given the monomer structures
polyesters and polyamides are formed form the combo of 2 different monomers
polyesters - most commonly used in fabrics/fibers
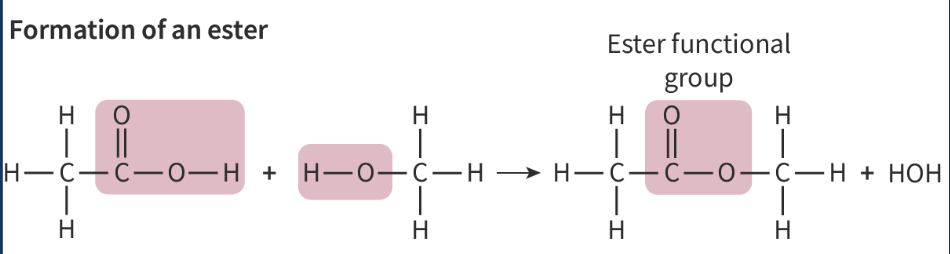
Ester functional group produced from alcohol + carboxylic acid
be mindful which atoms are lost to form the water molecules
H - from alcohol
single bonded O ends the alcohol side
OH - from carboxylic acid
C=O at the carboxylic acid end
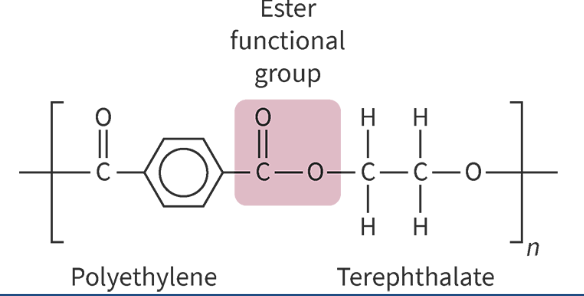
polyesters formed from combos of monomers that each have 2 functional groups
diol (2 hydroxyls) + dioic acid (2 carboxyls)
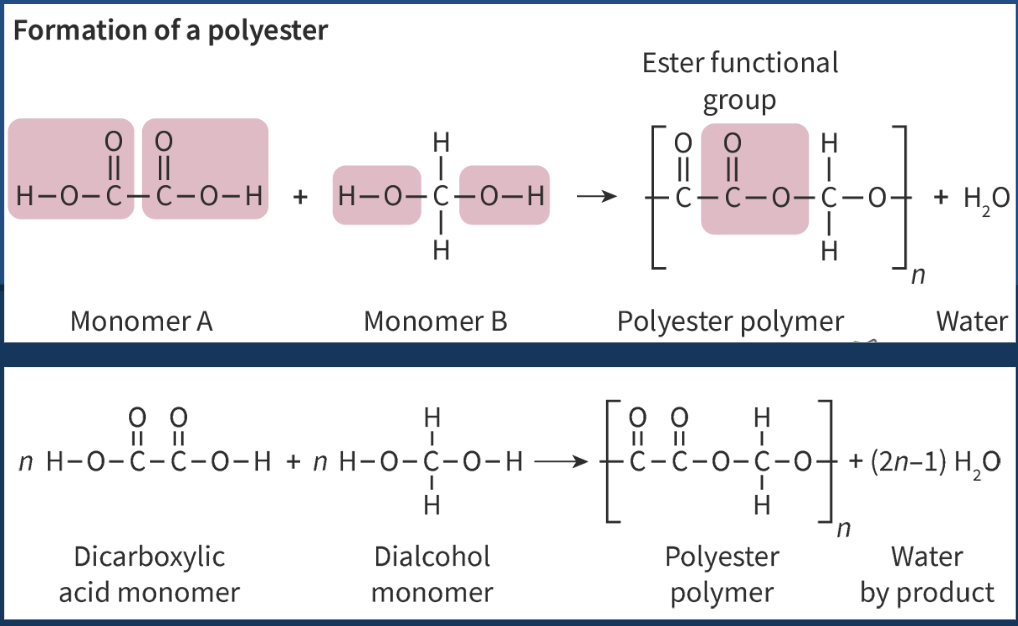
polyamides - used in ropes, seatbelts, and dental floss (most common = nylon)
amide functional group produced from amine + carboxylic acid
be mindful of which atoms are lost to form the water molecules
H - from amine
single bonded -NH ends the amine side
OH - from carboxylic acid
C=O at the carboxylic end
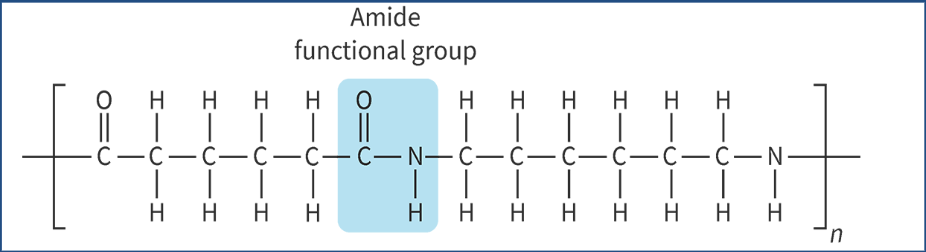
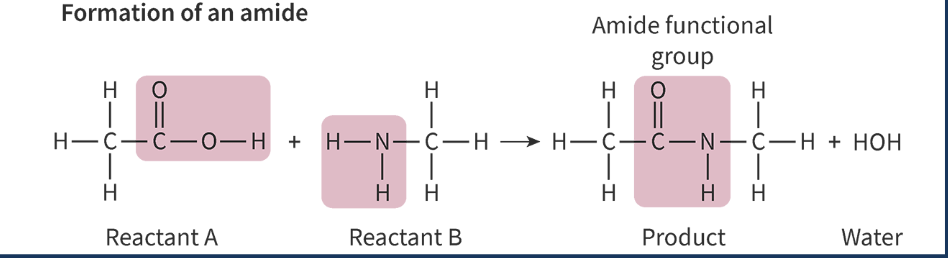
polyamides produced from combos of monomers that each have 2 functional groups
diamine (2 aminos) + dioic acids (2 carboxyls)