The Challenge of Extreme Environments I: Altitude
Introduction
This section explores the physiological challenges posed by extreme environmental conditions, with a primary focus on high altitude. It will cover:
The impact of altitude on oxygen availability and ambient temperature.
Acute physiological responses of lowland populations to sudden exposure to high altitude.
Longer-term acclimatization processes, particularly renal and hematological adaptations.
Adaptations observed in populations native to high-altitude environments, including variations among different groups (e.g., Andean, Tibetan, Ethiopian populations).
The role of genetic factors, such as Hypoxia-Inducible Factors (HIFs) and ACE gene variants, in high-altitude adaptation.
The relevance of high-altitude research to clinical medicine.
The Challenges of Altitude
High altitude presents significant physiological challenges, primarily due to:
Reduced Oxygen Availability:
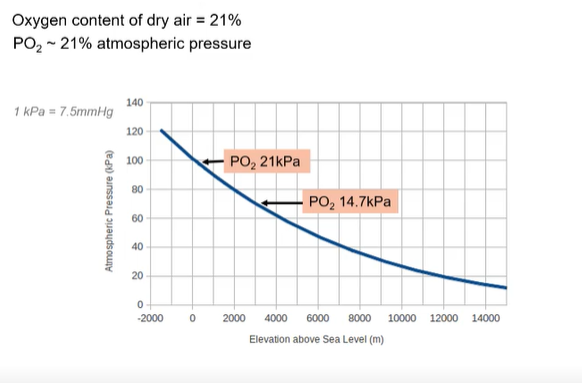
The oxygen content of dry air remains constant at approximately 21% regardless of altitude.
However, atmospheric pressure decreases with increasing elevation.
Consequently, the partial pressure of oxygen (PO2) also decreases proportionally (since PO2 is ~21% of atmospheric pressure).
Sea level: Atmospheric pressure ~100 kPa, PO2 ~21 kPa.
~3000m: Atmospheric pressure ~70 kPa, PO2 ~14.7 kPa.
~8000m (e.g., near Everest summit): Atmospheric pressure ~35-40 kPa, PO2 ~8.4 kPa.
Low Ambient Temperature:
Temperature generally decreases with increasing altitude.
Examples: -30°C at ~3000-4000m, potentially -80°C at very high altitudes like 8000m (though this seems exceptionally low for typical mountain environments and might be an extreme example or refer to specific atmospheric layers).
Responses to Altitude in Lowland Populations (Sudden Exposure >3000m)
When individuals accustomed to sea level are suddenly exposed to high altitude, a series of physiological responses are initiated to cope with hypoxia:
Rapid Ventilatory Adaptation (Acute Hyperventilation)
Hypoxemia Detection: Peripheral chemoreceptors (primarily in the carotid bodies) detect a fall in arterial PO2 (PaO2<70 mmHg or ~9.3 kPa).
Increased Ventilation: Chemoreceptor in carotid sinus, stimulation leads to an increase in minute ventilation (hyperventilation). This aims to increase alveolar PO2.
Respiratory Alkalosis: The increased ventilation exhales CO2 more rapidly than it is produced, leading to a fall in arterial PO2 (PaCO2). This causes respiratory alkalosis (increased blood pH).
Attenuation of Hyperventilation: The decreased PaCO2 and increased pH can depress the activity of chemoreceptors and the respiratory center, initially attenuating the magnitude of hyperventilation.
Experiments show that exposure to 7% O2 (simulating high altitude) or low pH (e.g., pH 6.6) can stimulate carotid body chemoreceptor firing.
Other Rapid Adaptations
Increased Heart Rate: Tachycardia occurs to compensate for arterial O2 desaturation and maintain oxygen delivery to tissues.
Fluid Loss and Reduced Plasma Volume: Increased respiration leads to greater insensible water loss. Diuresis can also occur. This reduces plasma volume, leading to hemoconcentration and an increase in hematocrit (the proportion of blood volume occupied by red blood cells).
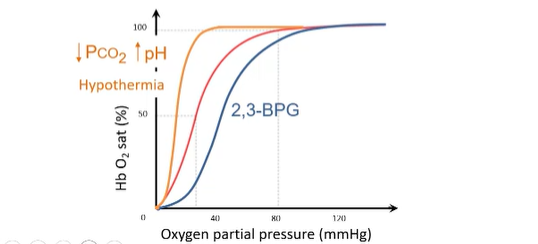
Modified Oxygen-Hemoglobin Affinity:
Initially, the respiratory alkalosis (due to hyperventilation and ↓PCO2) shifts the oxygen-hemoglobin dissociation curve to the left (Bohr effect). This increases hemoglobin's affinity for O2, aiding O2 loading in the lungs where PO2 is already low. Hypothermia at altitude can also contribute to a leftward shift.
Over time (within hours to days), levels of 2,3-Bisphosphoglycerate (2,3-BPG) in red blood cells increase. 2,3-BPG reduces hemoglobin's affinity for O2, shifting the curve to the right. This facilitates O2 unloading at the tissues. The net effect on the curve position is a balance of these opposing influences.
Pulmonary Hypertension: Hypoxia can cause vasoconstriction in the pulmonary arteries, leading to increased pulmonary arterial pressure.
Longer-Term Adaptation - Acclimatisation
With prolonged exposure to high altitude (days to weeks), further adaptations occur:
Renal Adaptation (longer term)
CSF pH Normalization: The initial respiratory alkalosis (loss of Co2) also affects the cerebrospinal fluid (CSF), making it more alkaline. Cells of the choroid plexus work to restore CSF pH by actively pumping more H+ into the CSF or transporting less HCO3− into the blood brain barrier.
Renal Bicarbonate Excretion: The kidneys respond to the systemic respiratory alkalosis by increasing the excretion of bicarbonate (HCO3−). This helps to return plasma pH to normal, a process known as renal compensation.
Sustained Hyperventilation: Once plasma and CSF pH are normalized, the primary hypoxic drive from peripheral chemoreceptors can fully stimulate ventilation without being counteracted by alkalosis, leading to a more sustained and effective hyperventilation.
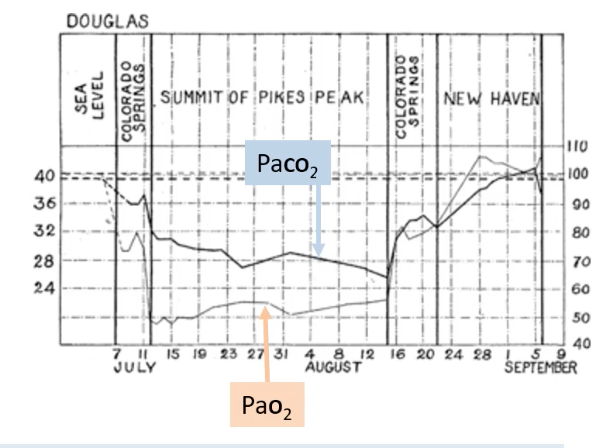
Historical research, like the Anglo-American Pikes Peak Expedition, demonstrated these changes. Over weeks at high altitude (Summit of Pikes Peak), PaCO2 gradually decreased (indicating sustained hyperventilation), while PaO2 increased from its initial nadir.
Haematological Adaptation
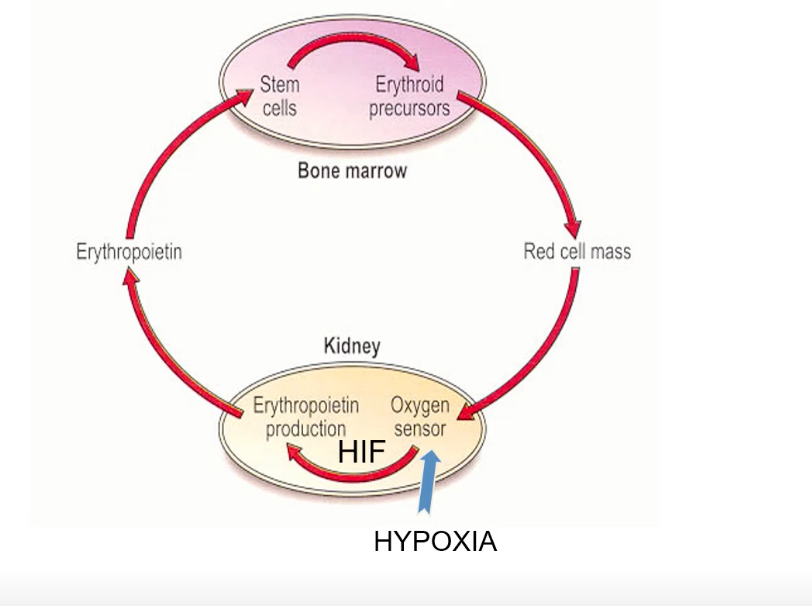
Erythropoiesis: Hypoxia stimulates the kidneys to release the hormone erythropoietin (EPO).
EPO acts on the bone marrow to stimulate the production of red blood cells (erythrocytes) from stem cells/erythroid precursors.
This leads to an increase in red cell mass and thus hematocrit (polycythemia).
The increased hematocrit raises the O2-carrying capacity of the blood.
The reduction in blood plasma volume (mentioned under rapid adaptations) also contributes to the increased hematocrit.
Adaptations in High-Altitude Native Populations
Populations that have resided at high altitude for many generations often exhibit distinct physical, physiological, and biochemical adaptations. However, these adaptations are not uniform across all high-altitude populations.
General Trends:
Ventilatory adaptation (sustained hyperventilation) may be less significant or different compared to acclimatized lowlanders.
A range of hematological and metabolic adaptations are often present, such as improved peripheral perfusion, faster O2 diffusion to tissues, and sometimes enhanced hemoglobin-O2 affinity.
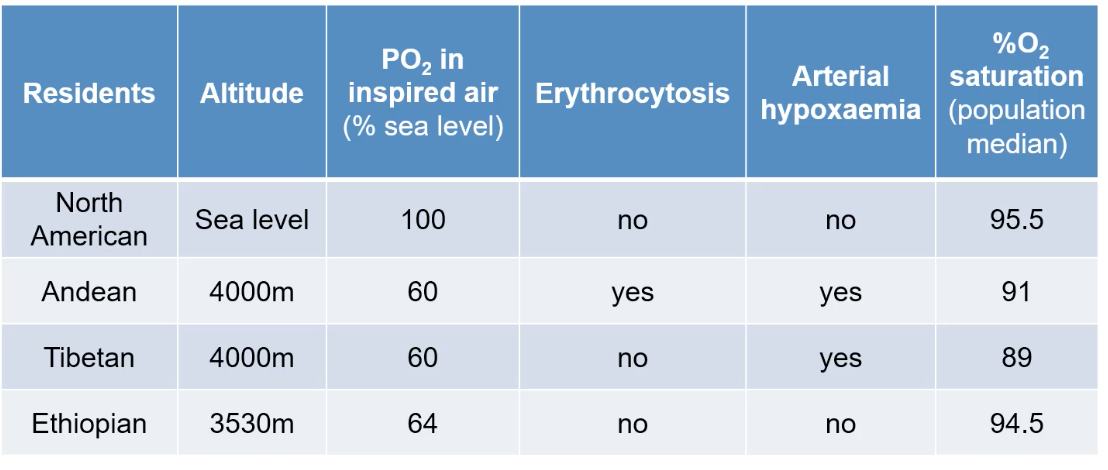
Population Variations (Examples):
Andean Highlanders (e.g., 4000m): Typically show marked erythrocytosis (high red blood cell count) and may still experience some arterial hypoxemia, with a median O2 saturation around 91%.
Tibetan Highlanders (e.g., 4000m): Often exhibit less pronounced or no erythrocytosis compared to Andeans, despite similar arterial hypoxemia (median O2 saturation ~89%). This suggests they have other mechanisms to cope with lower PO2.
Ethiopian Highlanders (e.g., Amhara, ~3530m): May show little to no erythrocytosis and minimal arterial hypoxemia (median O2 saturation ~94.5%), appearing more similar to sea-level populations in these aspects despite the altitude.
Adaptations in Sherpas (a Tibetan group):
Sherpas are particularly well-adapted, demonstrating an ability to maintain blood flow and O2 delivery to working tissues more effectively than acclimatized lowlanders.
They also show improved metabolic efficiency.
Genetic Basis of High-Altitude Adaptation
Genetic variations play a significant role in the adaptations seen in high-altitude populations.
Hypoxia-Inducible Transcription Factors (HIFs):
HIFs are master regulators of O2 homeostasis, coordinating the transcriptional response to hypoxia.
The oxygen sensor in the kidney (and other tissues) influences HIF stability and activity. Under hypoxic conditions, HIF becomes active and promotes the transcription of various target genes.
HIF Target Genes: Include erythropoietin (EPO), nitric oxide synthase (NOS, involved in vasodilation), lactate dehydrogenase, adrenergic receptors, glucose transporters (for enhanced glucose uptake), and insulin-like growth factor (ILGF).
Genetic Variations in Tibetan Populations:
Mutations in HIF2A (EPAS1) are common and correlate with a reduced hematopoietic (red blood cell production) sensitivity to hypoxia, partly explaining their lack of excessive erythrocytosis.
Mutations in EGLN1 (a gene encoding a HIF prolyl hydroxylase, which regulates HIF stability) and PPARA (a HIF target gene involved in metabolism) are associated with reduced hemoglobin concentrations ([Hb]) in Tibetans.
Angiotensin-Converting Enzyme (ACE) Gene Variants:
The ACE gene has an insertion/deletion (I/D) polymorphism in intron 16.
The insertion (I) allele of the ACE gene has been associated with reduced risk from hypoxia and better performance at extreme altitudes.
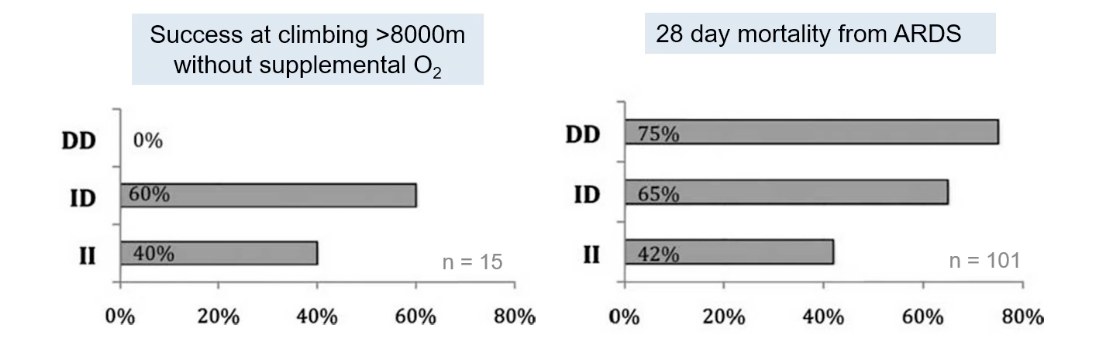
Individuals with the II genotype (homozygous for the insertion allele) are overrepresented among elite high-altitude climbers who ascend >8000m without supplemental O2. For example, in one study of 15 such climbers, 40% had the II genotype and 60% the ID genotype, with 0% having the DD genotype.
Conversely, the D allele has been linked to poorer outcomes in conditions involving hypoxia, such as Acute Respiratory Distress Syndrome (ARDS). In a study of ARDS patients, 28-day mortality was higher in those with the DD genotype (75%) compared to ID (65%) or II (42%).
Mechanism of ACE-II Benefit:
The II genotype is associated with reduced ACE activity.
Lower ACE activity leads to lower Angiotensin II activity (Ang II is a vasoconstrictor and promotes fluid retention).
This allows Nitric Oxide (NO) activity to dominate, promoting vasodilation.
Increased vasodilation can lead to improved oxygen and fuel delivery to tissues.
Relevance to Clinical Medicine: Lessons from Extreme Environments
Understanding how the human body adapts to extreme environments like high altitude can provide valuable insights into the pathophysiology and treatment of clinical conditions characterized by hypoxia. Research conducted in these environments (e.g., "lessons learnt from Mount Everest" by Dr. Daniel Martin) can inform strategies for managing patients in critical care settings