Determinants of Hormone Action
Transport of Hormones
Two chemical classes dominate reproductive endocrinology; their polarity dictates transport strategy:
Protein /peptide hormones
Hydrophilic → readily dissolve in aqueous plasma.
Circulate largely “free” (unbound) → immediately available to interact with surface receptors.
Steroid hormones
Lipophilic & water-insoluble → require carrier (transport) proteins for solubility.
Binding = “conjugation.” Multiple proteins display overlapping but unequal affinities.
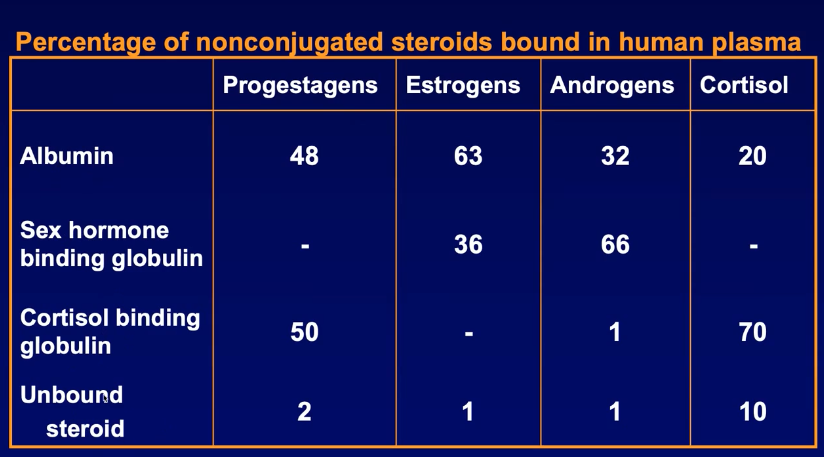
Key plasma carrier proteins & their average binding shares (human data representative of table):
Albumin (ALB) – high capacity, low affinity.
Sex Hormone–Binding Globulin (SHBG) – lower capacity, high affinity for androgens/estrogens.
Cortisol-Binding Globulin (CBG / transcortin) – high affinity for glucocorticoids & progestogens.
Typical distribution (% bound):
Biological activity derives almost exclusively from the tiny “free fraction,” because only unbound steroid can diffuse across membranes to nuclear receptors.
Clinical / pathophysiological note:
↓SHBG → ↑free testosterone → androgen excess signs
↑SHBG → ↓free testosterone → diminished androgen action.
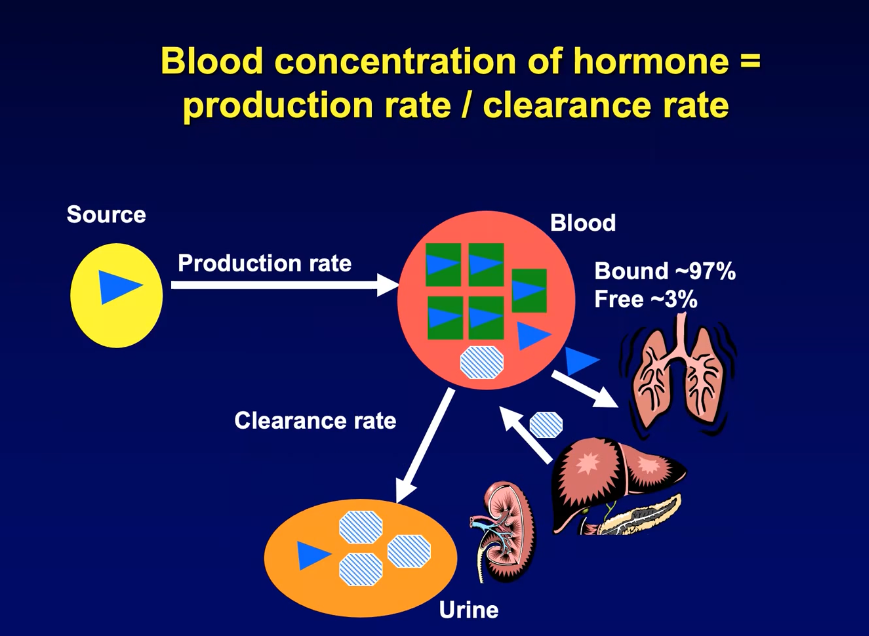
Blood Concentration & Clearance
Instantaneous plasma concentration of a hormone results from a balance:
Clearance routes & kinetics
Hepatic metabolism → conjugation/inactivation; metabolites excreted by kidney (urine).
Pulmonary “blow-off” into expired air for some volatile or small molecules.
Target-cell utilization – quantitatively minor (only a fraction of free hormones are utilized).
Half-life ( ) = time required for plasma concentration to fall to starting value.
Steroid hormones .
Prostaglandins .
Gonadotrophins (LH, FSH) . - longer half life
Functional consequence: rapid signal termination for steroids/prostaglandins vs prolonged action of glycoprotein hormones.
Metabolic Activation at Target Tissues
Enzymatic conversion of circulating hormone by target cell into more usable form.
Classic example: Testosterone → -Dihydrotestosterone (DHT)
Enzyme: -reductase within target cells.
DHT affinity for androgen receptor ≈ 10× that of testosterone → amplified response.
Developmental relevance: masculinization of external genitalia (penis, scrotum) during embryogenesis depends on local DHT production.
Secretory Patterns
Reproductive hormones are rarely secreted as smooth, steady outputs.
Three temporal motifs influence biological effectiveness:
Pulsatility
Discrete peaks separated by troughs.
Each peak - Pulse
Defined by amplitude & frequency.
Example: Testosterone pulses across 24 h; each peak lasts minutes.
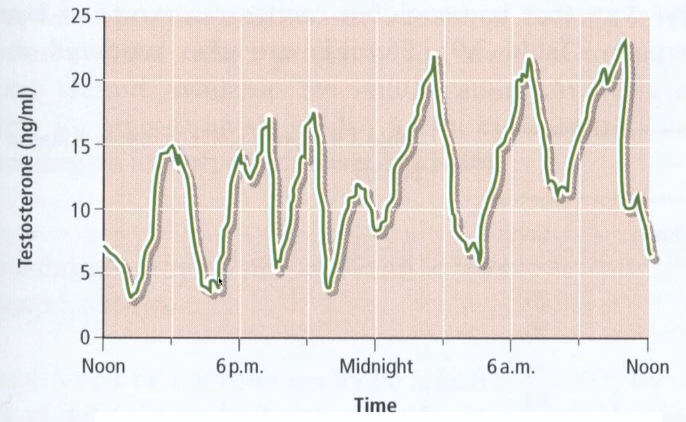
Circadian rhythm
Superimposed daily oscillation; e.g.
Testicular testosterone gradually rises during night–early morning.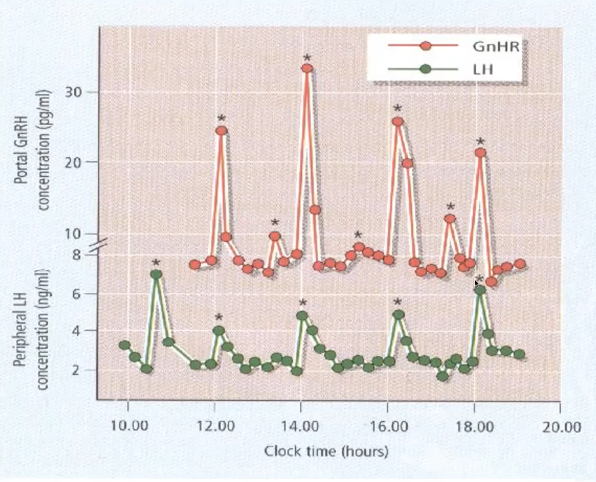
GnRH–LH paradigm (gold-standard for pulse-dependency)
GnRH released from hypothalamic neurons into hypophyseal portal blood only in pulses.
Every GnRH pulse triggers an LH pulse from the anterior pituitary; frequency helps set relative LH vs FSH output.
Exogenous pulsatile GnRH → maintains LH/FSH.
Clinical correlation: Excessive GnRH pulse frequency → favors LH > FSH → ovarian androgen excess → Polycystic Ovary Syndrome (PCOS).
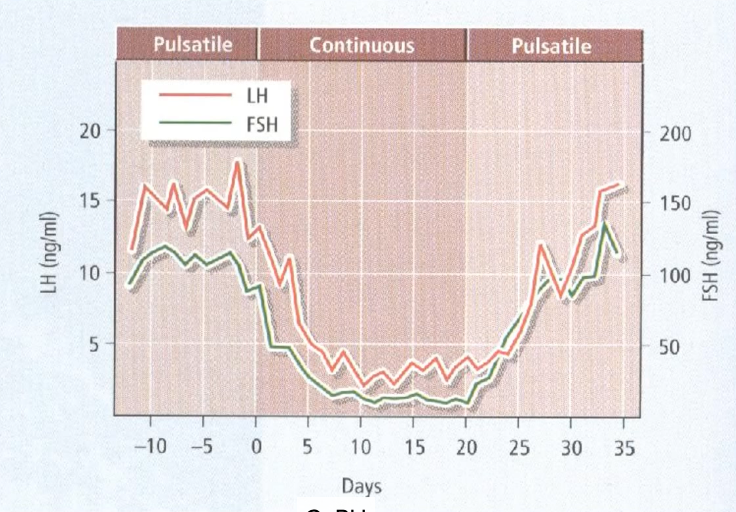
A pulsatile secretion of GnRH is need for secretion of gonadotrophin.
If its continues secretion - LH and FSH drops.
Receptor Expression & Regulation
Hormone can act only where cognate receptors are expressed.
Receptor density & sensitivity are determinants.
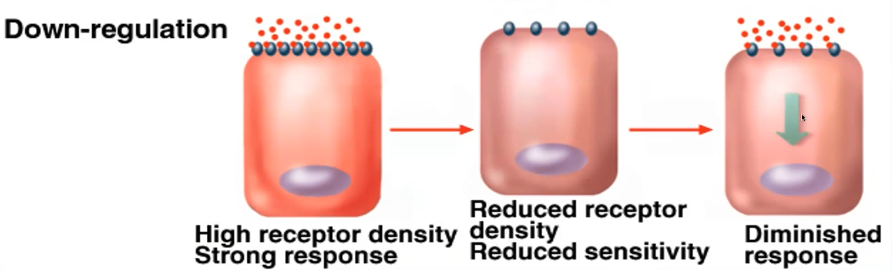
Down-Regulation
Chronic high ligand exposure → degradation of receptors or decreased synthesis.
Consequences
↓Receptor number
↓Cell sensitivity
↓Physiological response
Mechanism behind suppressed LH/FSH during continuous GnRH infusion.
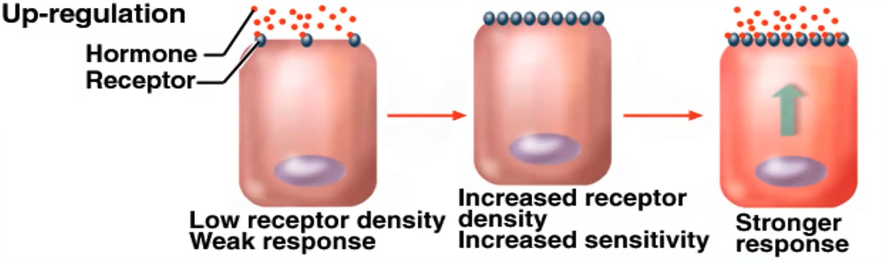
Up-Regulation
Opposite process; another hormone or paracrine factor increases receptor synthesis or membrane trafficking.
First - low receptor density and weak response.
This causes increase in sensitivity of receptors
Relevance: Dominant (pre-ovulatory) follicle up-regulates LH receptors in granulosa cells → heightened responsiveness → ovulation trigger.
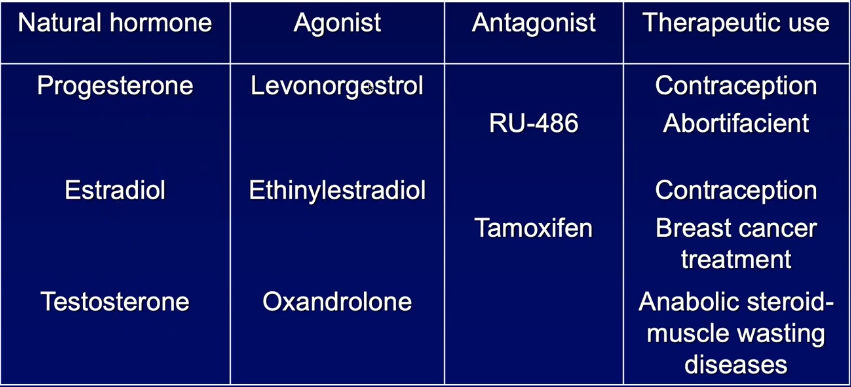
Agonists & Antagonists
Agonist = exogenous molecule that binds the receptor - promote the receptor.
Antagonist = binds receptor but blocks activation (competitive or inverse).
Therapeutic / experimental toolset (selected examples):
Progesterone analogs
Agonists: synthetic progestins (contraceptives, luteal support).
Antagonist: RU-486 (mifepristone) – emergency contraception, medical abortion.
Estrogen system
Agonists: ethinylestradiol (oral contraceptive component).
Antagonists / SERMs: Tamoxifen – blocks estradiol in breast tissue (ER-positive cancer therapy).
Androgen system
Agonists: anabolic steroids (therapeutic for cachexia, abused in sport).
Environmental Endocrine Disruptors
Industrial chemicals with estrogenic or anti-estrogenic activity:
Pesticide residues, plasticizers (e.g.
Bisphenol-A – BPA), phytoestrogens, pharmaceutical metabolites in wastewater.
Potential impacts: altered sexual differentiation, puberty timing, fertility, hormone-dependent cancers.
exogenous antagonist) can derail reproductive function and underlies numerous clinical disorders (PCOS, infertility, hormone-dependent cancers).