Matter and Change
Chemistry is a Physical Science
Intro to Chemistry:
- Chemistry- The study of the composition, structure, and properties of matter. The specific processes that matter undergoes, and any energy changes that occur in this process.
- Chemistry answers the questions of how materials go through changes and the certain behaviors that come along with reactions.
- Instruments such as balances can help us gain further observations and measurements.
Branches of Chemistry:
- Chemistry has multiple branches, that all overlap and intertwine with each other.
* Organic chemistry- The study of carbon-containing compounds.
* Inorganic chemistry- The study of non-carbon-containing compounds.
* Physical chemistry- The study of properties of matter, changes that they go through, and their relationship with energy.
* Analytical chemistry- The study of identifying compositions of specific materials.
* Biochemistry- The study of substances, and processes found in a living thing.
* Theoretical chemistry- The study of using math and computers to understand why chemicals behave in a certain way. It can also help to predict the properties of new substances. - Chemical- A substance that has a definite composition.
- No matter what area of chemistry chemicals will always be involved.
- It helps to know different properties of chemicals because then uses for specific chemicals can be found.
* Ex: Making new consumer products such as flavor enhancers and fabrics.
Basic/Applied Research & Technological Development:
- Basic research is used to get a basic idea or understanding of how chemicals interact with one another.
- Discoveries can sometimes happen through the process of basic research.
- Applied research is when information is gathered to help solve a problem.
- Technological development is when you have both production and use of products that can improve the quality of life.
- Technological applications are often behind the development of things that are eventually used as technology.
- Basic research, applied research, and technological development are often intertwined with each other. Discoveries that are made in the research process can cause applications to produce new technologies.
Matter and Its Properties
Intro to Matter:
- All things are made up of matter.
- Volume- The amount of three-dimensional space that an object occupies.
- Mass- The measure of the amount of matter.
* Generally measured using a balance or scale. - Matter- Anything that has mass and takes up space.
- ==All matter has both volume and mass.==
Basic Building Blocks of Matter:
- As mentioned before, matter comes in all sorts of forms.
- Atoms and molecules are the fundamental particles that make up all sorts of elements and compounds.
- Atom- The smallest unit of an element that keeps its chemical identity.
- Element- A pure substance cannot break down further, and is stable
- Compound- A substance that can be broken down into simpler substances that are stable. Generally, each compound is made from atoms of 2-3 elements bonded together.
- A molecule is formed by atoms that are bonded together chemically.
Properties and Changes in Matter:
- A molecule is the smallest unit of an element that retains the properties of the specific element or compound that it’s in.
- Every substance has characteristics that tell us about its properties.
- Different properties are used to distinguish substances.
- A property could easily define an entire group of substances.
- Multiple properties need to be compared to identify a specific substance.
- Extensive properties- dependent on the amount of matter present. Ex: volume, mass, energy in a substance.
* Ex: volume, mass, amount of energy in a substance - Intensive Properties- Don’t depend on the amount of matter that is present. Ex: melting point, boiling point, density, and conductibility.
* Ex: melting point, boiling point, density, conductivity, and amount of energy heat transferred
Physical/Chemical Properties & Change of State:
Physical Properties/Changes:
- Physical Property- A characteristic that can be observed or measured ==without changing the identity of the substance.==
* Ex: melting point, boiling point - Physical change- A change or substance that ==doesn't involve changing the chemical composition of a substance.==
* Ex: grinding, cutting, melting, and boiling
States of Matter:
- A change of state- a physical change of a substance from one state to another.
- Note: Most common states are solid, liquid, and gas.
- Solids- Have a definite volume and shape.
- Particles are put together in fixed positions.
- They are held together by the strong attractive force between them, and potentially vibrate at fixed points.
* Note: some solids are easily compressible compared to others - Liquid state- has a definite volume, but not a definite shape.
- It gently assumes the shape of the container.
- Particles found in liquids are close together but are able to move past each other.
- They move faster than those found in a solid, which causes the particles in a liquid to overcome the attractive force.
- Gas state- neither a definite volume nor a definite shape.
- All gasses have particles that move rapidly and are spaced out.
- The attractive force between gas particles has a lesser effect because they are spread out even further.
- A fourth state that isn't mentioned as often is Plasma.
- Plasma- a high-temperature, physical state where atoms lose a majority of their electrons.
* Ex: fluorescent bulbs, sun(stars.) - A change of state doesn't affect the identity of the substance.
Chemical Properties/Changes:
- Chemical properties- A substance undergoes changes that affect its chemical composition.
* It's easier to notice these properties when you observe new substances being formed. - Chemical change or chemical reaction- A change in one or more substances that convert into different substances.
- Parts of a Chemical reaction:
* Reactant- Substances that react during a chemical reaction.
* Products- Substances that are formed by chemical change.
Energy and Changes in Matter:
- Chemical reactions have a special way that they are written.
- Conservation of Matter- the amount of matter isn’t affected by the reactions that occur.
- When physical or chemical changes occur energy will be involved.
- Energy can be absorbed or released, but it isn’t destroyed or created. It just has a different form.
Classification of Matter:
- Matter can be found in all different forms.
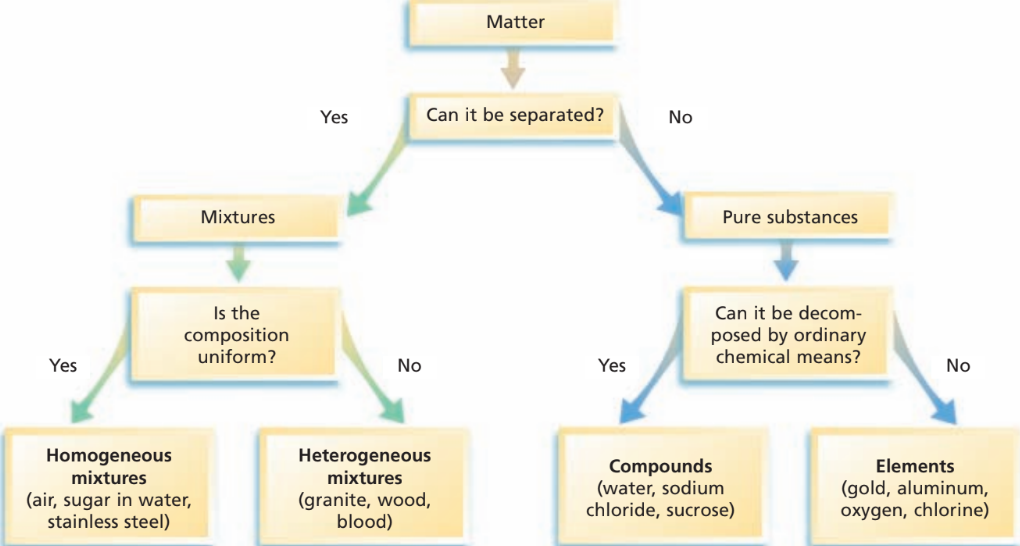
- Matter can be classified as either a pure substance or a mixture.
- Pure substance- The composition is the exact same, and doesn't vary from sample to sample. It could be either an element or a compound.
- Mixtures- Contain 1 or more substances. They change in composition and properties from sample to sample.
- Mixtures can be found everywhere. They can be simple or complicated and have unique properties.
- Mixtures can usually be separated. The properties of mixtures are classified by the properties of the different components.
- \
Homogenous- Mixtures that are uniform in composition, and are proportional to each other. Aka solutions. - Heterogeneous- Mixtures that aren’t uniform.
- Mixtures have different methods that can be used to separate their components such as filtration, or vaporization.
- A centrifuge can be used to separate solid and liquid mixtures.
- Pure substance- Has a fixed composition.
* Every sample of a pure substance has the exact same characteristics, or properties.(chemical and physical)
* Every sample of a pure substance has the exact same composition. - Note: Pure substances are their own thing. They aren't elements or compounds.
Chemicals found in labs are treated like they are entirely pure. All chemicals have impurities, and they can affect the outcome of a reaction or experiment.
- The American Chemical Society sets the standards for purity rankings.
Elements
Intro to the Periodic Table:
- Each small square on the periodic table shows the symbol for an element, and its atomic number.
- Groups or families- vertical columns of a periodic table.
* Each group has elements that have similar chemical properties. - Periods- Horizontal rows on the periodic table.
* Physical and chemical properties can vary in a period. - The Lanthanide and Actinide series are below the periodic table and fit in from elements 57-89.
They are placed below the table to keep it from becoming too wide.
Types of Elements:
- The periodic table is divided into 2 major sections. Metals and nonmetals.
- Metal Properties
* Good electrical, and heat conductors
* Lustrous
* Solid at room temperature
* Malleable
* Ductile - Nonmetal Properties:
* A poor conductor of heat and electricity
* Brittle
* Many are gaseous at room temperature - Metalloids Properties:
* Has characteristics of metals, and some nonmetals
* Solids at room temperature
* Generally less malleable than metals but aren't brittle like non-metals
* Metalloids are generally semiconductors of electricity - Noble Gases:
* Group 18 elements
* Highly unreactive
