lesson 12 -MSCs and regeneration
Page 1: Introduction to Mesenchymal Stem Cells (MSCs)
BM stroma can generate bone, fat cells, and cartilage following heterotopic transplantation in mice.
Mesenchymal stem cells (MSCs) play a critical role in this process.
MSC cells were discovered 60 years ago in experiments where they took BM of mice and put into the tissue of other mice. This study told the researchers that there are another type of BM cells that can develop into cartilage, bone, and fats that make up the various tissues within the body, indicating the potential for MSCs in regenerative medicine and tissue engineering.
Page 2: Characteristics of MSCs
Cited work: Bianco et al. Nat Med. 2013. 19:35
MSCs defined as non-hematopoietic BM multipotent precursor cells.
They possess skeletal and adipose potential.
MSCs are postnatal, self-renewing, clonogenic, and multipotent stem cells that give rise to all skeletal tissues.
All of these are mesodermal.
Page 3: Niche of MSCs
MSCs reside at the endosteal niche ( in the BM next to the bone) and the outer surface of sinusoids.
Cited work: Bianco et al., 2013; Coste et al. Front Cell Neurosci. 2015. 9:218
The MSCs that grew on the plate itself were the ones that grew into bone and muscle. Therefore they learned that the microenvironment of the plate significantly influences the differentiation and regenerative potential of MSCs, highlighting the importance of substrate choice in tissue engineering applications.
Clonogenic—do they have the ability for one cell to create a clone.
Checked this is that they grew BM cells very densely on the plate, and therefore any colony that was received came from one cell—this allowed the researchers to understand that only one cell was required for cloning
In terms of checking multipotent capability of the BM MSCs, researchers took one colony and grafted it into the mice, and saw the exact same ability to differentiate into different types of structural cells. When they removed these cells from the mice, cultured on plates, and then regrafted them, the conclusion sis that they retain their ability to be multipotent.
Page 4: Biology of Mesenchymal Stem Cells
Construct and regulate the homeostasis of hemopoietic stem cells (HSCs). In other words, they support the BM cells, giving them the signals to self-renew and differentiate
Maintain HSCs in a quiescent state characterized by self-renewal and proliferation without differentiation.
Regulate mobilization of HSCs into the vascular system. Give them the signals to do this.
MSCs respond to inflammatory cues and chemokine gradients, facilitating wound healing by suppressing inflammation. When there is a wound that leads to inflammation, the MSCs leave the BM and go to the site of the inflammation and inhibit the spread of the inflammation outside of the site.
Contribute to tissue reconstruction and skeletal physiology by producing and depositing vimentin, crucial for bone growth and turnover.
Basically, MSCs are police traffic stoppers and directors.
Page 5: Key References on MSCs
There are other tissues beyond the BM have that the same multipotent differentiation ability. Therefore this changes the definition of what the MSCs because they aren’t necessarily in the bone marrow. Therefore they determine the definition of MSCs as a broader category of cells that can be found in various tissues, including adipose tissue and dental pulp, thereby expanding their potential applications in regenerative medicine.
Therefore need to be very clear in publications with the definitions and with other labs, because each lab has their own definition.
Key references include works by Nombela-Arrieta et al. (Nat Rev Mol. Cell. Biol. 2011) and Bianco (Annu. Rev. Cell Dev. Biol. 2014).
Page 6: Mechanisms of MSC Therapy
Key mechanisms in the positive effects of MSC therapy:
Differentiation (limited selection but yes they can)
Anti-apoptosis
Angiogenesis (growth of blood vessels)
Growth factor production (structural cells)
Antifibrosis
Immunomodulation (suppress inflammation which can be very important in terms of host-graft relationship)
Reducing inflammation and suppressing immune cell function in various settings.
Advantages of MSCs: convenient isolation and low immunogenicity allowing for allogenic transplantation.
Page 7: MSC-Based Therapy Strategies
In general there are two strategies for cell therapy: cell replacement and protective/trophic therapy. MSCs can be used to promote tissue regeneration through both strategies, as they have the ability to differentiate into various cell types and secrete factors that enhance healing.
Modes of MSC-based therapy:
Cell replacement
Cell “empowerment.” Their trophic capabilities are broad and therefore can be helpful for cell therapy based on trophic therapy.
Cited works: Wang et al. Nature Immun. 2014; Maldonado et al. Journal of Biological Engineering (2023).
Page 8: Clinical Applications of MSCs
Immunosuppression in donor organ transplants (like kidney)
Prevention of graft-versus-host disease (GVHD)
Treatment of ischemic heart diseases by differentiating into cardiac tissue and secreting cytokines.
Neurological disorders treatment by promoting neuron recovery and reducing apoptosis.
Page 9: First Clinical Applications of MSCs (Immunosuppression)
Significant report of MSC application in treating severe acute GVHD using haploidentical MSCs.
They used the MSCs to reduce the issue of host-graft rejection and inflammation.
Noted clinical response showing MSCs' potent immunosuppressive effects.
Page 11: Osteogenesis Imperfecta (OI) (Bone/cartilage defects)
This is an example of cell replacement therapy
Defined as a genetically heterogeneous disorder, leading to atypical skeletal development and fractures. This already appears in vitro in fetuses
Mutations in COL1A1 and COL1A2 genes account for 90% of cases.
Type I collagen, most abundant in bones and connective tissues, is primarily affected in OI.
Page 12: Mouse Model Study for OI
They grafted the MSCs, and they moved themselves to the muscles that were infected, and showed an improvement in muscle regeneration in the mice.
Reference: Niyibizi et al. Int J Clin Rheumatol. Feb 1, 2009.
Study of MSC therapy specific to OI.
Page 13: Fetal MSC Transplantation for OI
Pre- and postnatal fetal MSC transplantation findings and safety conclusions. They grafted the MSCs to a fetus in mice and saw that the treated mice exhibited improved bone density and structural integrity compared to the control group, indicating a potential therapeutic benefit of MSC transplantation in the context of osteogenesis imperfecta. However, it did need two grafts to actually work. Here there is a replacement of the cells that were faulty.
Ongoing studies needed, though initial results are promising.
Page 15: ALS Trials
Neurological disorders such as ALS can also be treated. These can’t be used in terms of cell replacement, because we aren’t sure if they can differentiate into the neurons themselves, however they can secret growth factors and signals that lead to the protection of the affected cells. For example, Parkinson’s can use cell replacement to replace damaged dopaminergic connections as we saw in a previous slideshow, but here, it uses MSCs to protect against the progression of ALS.
Information regarding clinical trials of MSC-based treatment for ALS, referencing multiple phases and types of trials. This was developed in Israel!
Page 16: Limitations of MSC Therapy
Poor engraftment, limited differentiation, aging impact, and unwanted mesenchymal lineages pose challenges.
Highlight the need for further research on the mechanisms regulating MSC functions.
Ton of studies being done on MSCs.
Page 18: Common Stem Cells in Trials
Pluripotent Stem Cells
Multipotent Stem Cells
Hematopoietic Stem Cells
Mesenchymal Stem Cells
Epidermal Stem Stem Cells
Endothelial stem cells
Neuronal Stem Cells
Moving on from Cell Therapy to Regenerative Medicine
In general, we need removal of cells from patient and host, and put the cells into the patient.
Integration into the desired tissue/organ and correction/replacement of the abnormal cells
Focus on Regeneration
Important to understand that we haven’t even gotten to clinical trials of this strategy. But if we could regenerate limbs, would be amazing for a lot of reasons
Liver somewhat does this but we will discuss later on into the course.
Studying the Axolotl for their unique regenerative abilities. They can even regenerate the telencephalon or spinal cord, their reproductive system, and even a third of the heart ventricles. They do this through transforming nearby cells to the wound into STEM cells which regenerate the impacted tissue.
Regeneration: the replacement of lost body parts, restoring mass and function.
The ability of regeneration reduces with evolution, making mammals the least capable of regeneration. Why is this?
Page 24: Species-Specific Regeneration
Two hypothesis:
regeneration is a fundamental property of all organisms, and something stops it in many species
Regeneration is an adaptive property that has been GAINED during evolution in some species
Page 25: Scar Formation vs. Wound Healing
Comparison of scar formation and true tissue regeneration.
Reduces the ability of regeneration with age; we have some regenerative ability as we are younger, such as healing of wounds. When we are younger, there is more skin regeneeration of a cut on the skin, whereas elderly are more prone to develop scars.
Page 26: Zebrafish Cardiac Regeneration
Insight into how adult zebrafish regenerate hearts without scarring.
They have a regenerative ability of the heart, even in adults. It was known that this regeneration happens through cardiomyocyte proliferation.
Embryonic mammalian heart is capable mounting a hyperplastic response to injury—there leads to the question if the mammal has abilities to regenerate itself also after birth.
Cardiac Regeneration Limitations in Mice
Observation of limited regenerative capabilities following myocardial injury in young mice. They saw that there was a lack of regeneration after a week, and scarring appeared. they also did a similar study where they did a surgical resection of the heart after a day after birth, and it healed perfectly without scarring. Therefore they concluded that there was a correlation between age and wound healing.
Option 1: Lack of resident cardiac stem cells in adult mice
Option 2: Regeneration is not dependent on stem cells, but inability to reenter the cell cycle and complete division (could be due to either intrinsic cell cycle block or loss of mitogenic stimuli as the heart ages or both.)
Studies on Pig Heart Regeneration
They did a study on pig hearts at different days after birth of the heart, and follow-up assessments were conducted up until 12 weeks. They did this because the pig and humans have similar heart structures, and it serves as a good model organism for this type of study.
The scarring increased significantly with age, indicating that older pig hearts had a reduced capacity for regeneration and healing compared to younger hearts.
Hearts of large mammals have regenerative capacity, likely driven by cardiac myocyte division, but this potential is lost immediately after birth.
Large Animal Studies on Regeneration
Heart regeneration capacity (and other organs) decreases with evolution and with age.
Even in animals that undergo regeneration really well, age still limits their ability of regeneration.
Page 33: Questions on Regeneration
What define sand controls regenerative potential?
Why did it reduce with evolution and age?
What are the cellular sources of regeneration?
What factors initiate regeneration? How is their activation targeted to an injured area?
What signals control proliferation and patterning during regeneration? How is the process completed appropriately—in other words, salamanders can regenerate the same limb every time? How do they regulate this?
What defines and controls regenerative potential?
Mammals don’t possess the genes? Or maybe they are silenced? We aren’t sure and are still investigating through scanning the axolotl’s genoem to discover.
Immune System's Role in Healing
The immune system of evolutionary more primitive organisms is slightly different. In our enhanced immune system, ther is a suppression of regeneration. In other words, this is the evolutionary cost that we paid for a better immune system.
Page 36: Hallmarks of Regeneration
Identification of key aspects that define successful regeneration processes. We aren’t getting into it.
Age-related limitation of the regeneration: Lin28 and Gene Regulation
Immune system improvements—our immune system weakens with age because we can live longer, slightly connected to regeration as well.
Lin28: is a gene (that isn’t most commonly researched, Alon just likes it) that was discovered to have some tissue repair through reprogramming cell metabolism.
Lin28 is a highly conserved RNA-binding protein. It is heterochronic gene that is relevant to the speed of development, and it is involved in oncogenesis (overexpression = cancer).
Testing in Mice: Transgenic Lin28 Activation Mechanism
Description of transgenic methods for regulating Lin28 expression and resulting effects.
They overexpressed Lin28 in mice. Tet-on system is an inducible transgenic promotor that only goes on when tetracycline is added to the organism.
Lin28's Role in Wound Repair
Effects of Lin28 on wound repair improvement in mouse models. IN general, we tag mice through holes in the ear, and in the transgenic mice, the holes kept closing! This observation indicates that Lin28 significantly enhances the wound healing process, potentially by promoting cell proliferation and reducing apoptosis in the surrounding tissue.
However, this was also potentially a leaky gene effect, where it was closing before the promotor was activated. So they checked again in more limited circumstances, and saw that when the promotor was activated, there was reduced healing time.
Lin28 Hair Regrowth
Description of hair growth cycles and implications for regeneration. They shaved mice and saw that Lin28 increased hair regrowth. Even without adding the tetracycline, they saw the transgenic mice had increased hair regrowth, therefore it is likely there is another factor involved in the Lin28 process.
The Hair regrowth process is highly regulated, and wanted to see at which stage of the hair follicle regrowth Lin28 activation helped.
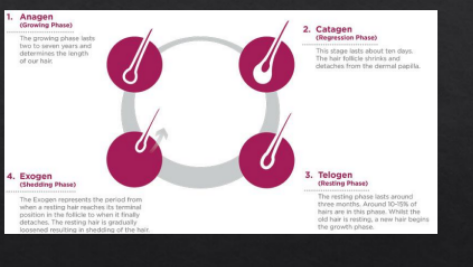
Induction of the anagen stage in adult hair follicles in mice.
Page 43: Digit Regrowth After Amputation
Examination of Lin28a's impact on regrowing neonatal digits post-injury.
Page 44: Mechanism of Lin28 in Tissue Repair
Changes in the metabolism in the mice to be similar to the developmental stages of the fetus of the mice.
“Our studies support the concept that mammalian tissues can be substantially improved by engineering activation of genes in the metabolism of the fetus metabolism.”
They do not use the term regeneration in the paper: this is rather based on genetic overexpression of juvenile bioenergetic genes such as Lin28 to lead to tissue repair.
Context-Specific Effects of Lin28
Noted limitations of Lin28-related healing in specific tissues post-injury.
One of the trials they did was that they made a cut in the heart of these transgenic mice, and saw no improvement of the Lin28 digits.
STOPPING HERE for the course. Don’t need to know after this
Page 46: In-vivo Reprogramming Techniques
Outline of various pathways for reprogramming adult cells within the body.
Page 47: Discovery of Inducing Pluripotent Stem Cells
Overview of Yamanaka's discovery on pluripotency and reprogramming factors.
Page 48: In-vivo Reprogramming Outcomes
Summary of findings on how in vivo reprogramming relates to teratoma formation.
Page 49: Applications and Indications of Reprogramming
Benefits of partially reprogramming cells for therapy and regenerative purposes.
Page 50: Cancer Risks from Reprogramming
Potential cancer risks associated with in vivo reprogramming highlighted.
Page 51: Teratoma Formation Studies
Analysis of factors related to teratoma formation following reprogramming.
Page 52: Wilms Tumor Analysis
Examination of Wilms tumor as a failure of kidney differentiation in pediatric cases.
Page 53: Reprogramming and Aging Mechanisms
Summary of how reprogramming actions can improve age-related hallmarks.
Page 54: Induction Effects on Age-Related Genes
Impact of reprogramming on the expression of age-related genes and potential cancer development.
Page 55: Lifespan Extension via Reprogramming
Findings on how reprogramming influences lifespan and aging traits.
Page 56: Epigenetic Changes and Aging
Discussion of how reprogramming affects aging at the cellular level.
Page 57: From Senescence to Pluripotency
Overview of transitions from cellular aging states to pluripotent characteristics.
Page 58: Research Goals on Reprogramming
Focus on re-establishing cellular identity amid challenges from oncogenic factors.
Page 59: Vision Restoration Approaches
Study results showing how reprogramming methods restored vision in mice.
Page 60: Long-Term Effects of Reprogramming
Safety assessments of continuous OSK expression in vivo.
Page 61: Axonal Regrowth Post-Injury
Effects of RGC treatment on optic nerve injury recovery and vision restoration.
Page 62: Direct Reprogramming Overview
Context for various developmental and reprogramming strategies.
Page 63: Historical Context for Reprogramming
Reference to foundational literature on cellular programming techniques.
Page 64: Inducing Pancreatic Cells
Study outcomes focusing on converting pancreatic cells for therapeutic applications.
Page 65: Neuron Conversion Techniques
Examination of successful demonstrations reprogramming fibroblasts into neurons.
Page 66: In-vivo Applications in Regenerative Medicine
Discussion of the potential for in vivo reprogramming strategies.
Page 67: Challenges in Direct Conversion
Outline of major challenges and strategies for achieving direct conversion effectively.
Page 68: Reprogramming Mechanisms in Cardiac Therapy
Detailed analysis of ways to achieve cardiac repair through reprogramming strategies.
Page 69: Advantages of In-vivo Reprogramming
Comparative advantages of in vivo reprogramming over traditional therapies highlighted.
Page 70: In Situ Cardiac Reprogramming Observations
Demonstrations of effective in situ reprogramming outcomes in cardiac tissues.
Page 71: Methodologies in Cardiac Reprogramming
Examination of various methodologies for in vitro and in vivo cardiac reprogramming.
Page 72: Efficiency of Cardiac Reprogramming
Insights into cardiac reprogramming efficiency and factors affecting success rates.
Page 73: Challenges in Cardiac Reprogramming Efficiency
Addressing barriers faced by in vitro reprogramming in future studies.
Page 74: Murine Cardiac Study Findings
Notable results from studies on cardiac fibroblast reprogramming methods and outcomes.
Page 75: Cardiac Repair Strategies
Reformative results from reprogramming cardiac fibroblasts into functional cardiomyocytes.
Page 76: In Vivo Reprogramming Efficiency
Continued focus on the improved efficiency noted in vivo versus in vitro reprogramming.
Page 77: Remaining Questions in Cardiac Reprogramming
Recognition of knowledge gaps and necessary future research avenues.
Page 78: Emphasis on Continued Research
Advocated need for further research to optimize cardiac reprogramming protocols.
Page 79: Sendai Virus in Reprogramming Context
Introduction of Sendai virus properties and potential applications in reprogramming endeavors.
Page 80: Direct Reprogramming in Liver Fibrosis Treatment
Highlights on direct reprogramming of myofibroblasts to mitigate liver fibrosis effects.
Page 81: Characterization of Direct Hepatic Reprogramming
Detailed descriptions of functional changes in liver-related cellular models.
Page 82: Cre-Lox Mechanism Overview
Technical aspects regarding DNA excision techniques utilized in experimental setups.
Page 83: Lineage-Tracing for Hepatic Function
Demonstrative models for observing the formation of induced hepatocytes.
Page 84: Liver Function After Reprogramming
Functional testing and characterization data post reprogramming in hepatic studies.
Page 85: Characterization of iHeps
Insights on the functionalities of induced hepatocyte-like cells and their performance in assays.
Page 86: Strategies for Hepatocyte Induction
Overview of transcription factors utilized for facilitating hepatic reprogramming.
Page 87: Astrocyte to Neuron Conversion Pathway
Examination of strategies converting glial cells into neuron-like cells for treatment of disease.
Page 88: Glia-to-Neuron Conversion in PD Treatment
Advances in therapeutic approaches using glial cells for neuronal replacement in Parkinson's disease.
Page 89: Reversing Parkinson's Disease Symptoms
Comprehensive examination of neuroprotective strategies employed to alleviate symptoms in animal models.
Page 90: Outlook on In Vivo Reprogramming Challenges
Final thoughts on the current state of in vivo reprogramming and its future directions.
Introduction to Mesenchymal Stem Cells (MSCs)
Mesenchymal stem cells (MSCs) are a type of non-hematopoietic multipotent stem cell found primarily in the bone marrow (BM) stroma. They have gained significant attention in regenerative medicine due to their capacity to differentiate into various cell types, including bone, adipose, and cartilage cells. In particular, MSCs play a crucial role in tissue regeneration following heterotopic transplantation in experimental models, notably in mice, indicating their potential therapeutic benefits.
Characteristics of MSCs
Definition: MSCs are defined as non-hematopoietic bone marrow multipotent precursor cells.
Potential: They possess the ability to differentiate into skeletal and adipose tissue.
Properties: Postnatally derived, MSCs are self-renewing and clonogenic with the unique capacity to give rise to all skeletal tissues.
Clinical Relevance: The clinical applications of MSCs are vast, and ongoing research is focused on enhancing their therapeutic applications in various diseases and injuries.
Niche of MSCs
Location: MSCs reside primarily in the endosteal niche at the bone surface and surrounding the outer surface of sinusoids. This specialized microenvironment is crucial for maintaining their stem cell properties and regulating their activity.
Cited Work: Significant contributions to understanding the niche of MSCs have been made by Bianco et al., (2013) and Coste et al. (2015).
Biology of Mesenchymal Stem Cells
Role in Hemopoiesis: MSCs are integral in constructing and regulating the homeostasis of hemopoietic stem cells (HSCs) within the bone marrow. They maintain HSCs in a quiescent state through their self-renewal and proliferation.
Response to Inflammation: MSCs exhibit a notable capacity to respond to inflammatory cues, which allows them to mobilize HSCs into the vascular system when needed. They also play a role in wound healing by suppressing inflammatory responses and facilitating tissue repair.
Skeletal Physiology: In addition to their roles in hemopoiesis and wound healing, MSCs contribute to bone physiology by producing vimentin, a vital component in bone growth and turnover.
Key References on MSCs
Crucial literature on MSCs includes Nombela-Arrieta et al. (2011) and Bianco (2014), which provide in-depth insights into their properties and applications.
Mechanisms of MSC Therapy
Key mechanisms underlying the therapeutic effects of MSC treatment include:
Differentiation: MSCs can differentiate into various cell types, which is fundamental for tissue repair.
Antiapoptosis: Their ability to prevent cell death is essential in maintaining tissue integrity during injury.
Angiogenesis: MSCs promote the formation of new blood vessels, supporting healing and tissue regeneration.
Cytokine Production: They secrete growth factors and cytokines that facilitate healing processes.
Antifibrosis: MSCs can mitigate fibrosis by influencing the behavior of fibroblasts in healing environments.
Immunomodulation: They play a vital role in reducing inflammation and suppressing immune responses, making them suitable for conditions such as graft-versus-host disease (GVHD).
Advantages: MSCs are advantageous for their convenient isolation from various tissues and low immunogenicity, which supports their applicability in allogenic transplantation.
MSC-Based Therapy Strategies
Modes of MSC-based therapy encompass two primary strategies:
Cell Replacement: Direct replacement of damaged or dysfunctional cells with healthy MSCs.
Cell Empowerment: Enhancing the endogenous repair mechanisms by treating the patient with MSCs that modulate their immune response or promote healing processes.
Cited Works: Relevant studies include those by Wang et al. (2014) and Maldonado et al. (2023).
Clinical Applications of MSCs
Immunosuppression: In donor organ transplants, MSCs help to manage rejection and facilitate acceptance.
Graft-Versus-Host Disease (GVHD): MSCs are used to prevent and treat GVHD, a significant complication in stem cell transplant patients.
Cardiac Applications: They have also shown promise in treating ischemic heart diseases by differentiating into cardiac tissue and releasing cytokines that support heart function.
Neurological Disorders: MSC therapy is employed to promote recovery in neurological disorders, enabling neuron recovery and reducing apoptosis in damaged tissues.
Osteogenesis Imperfecta (OI)
Osteogenesis Imperfecta (OI) is characterized by a genetically heterogeneous background, leading to atypical skeletal development and recurrent fractures. Most cases are attributed to mutations in the COL1A1 and COL1A2 genes, which encode type I collagen, the most abundant collagen type in bone and connective tissues. Recent MSC therapy has been explored as a treatment avenue for OI, aiming to supplement the deficits in collagen production.
Limitations of MSC Therapy
The therapeutic potential of MSCs is tempered by several limitations, including poor engraftment at target sites, limited differentiation capabilities, the impact of aging on MSC functionality, and the risk of unwanted differentiation into mesenchymal lineages. These challenges underscore the critical need for further research to understand and regulate MSC functions effectively.
Future Directions in Research
Enhanced understanding of MSC biology and mechanisms will inform the development of improved therapeutic approaches and strategies. Continuous research is essential to overcome the barriers currently limiting the application of MSCs in clinical settings and to exploit their full potential in regenerative medicine.