L11 - Nonlinear Pharmacokinetics
Overview of Nonlinear Pharmacokinetics
Nonlinear pharmacokinetics (PK) is characterized by dose dependency where pharmacokinetic parameters change with dose.
Unlike linear pharmacokinetics, where parameters such as clearance and volume of distribution are consistent with dose and time, nonlinear PK is time-dependent, meaning these parameters change over time, especially during continuous administration.
Key Concepts
Differences Between Linear and Nonlinear Pharmacokinetics
Linear pharmacokinetics:
PK parameters (e.g., clearance) remain consistent regardless of the dose.
Changes in plasma concentration are proportionate to changes in dose.
Nonlinear pharmacokinetics:
Exhibits saturation and dose dependency, meaning that as the dose increases, the increase in plasma concentration is not linear.
Typical examples include older anticonvulsants like phenytoin, which have a narrow therapeutic range and require careful monitoring.
Saturable Processes
Nonlinearity can arise from saturable processes including:
Capacity limited metabolism (saturable metabolism).
Saturable absorption or protein binding mechanisms.
Example drugs include phenytoin, carbamazepine, sodium valproate, and phenobarbital.
Michaelis-Menten Kinetics
Michaelis-Menten parameters play a crucial role in understanding drugs with capacity limited metabolism.
Vmax: Maximum rate at which enzymes metabolize a drug (average for phenytoin is ~7 mg/kg/day).
Km: Affinity of the enzyme for the drug, which indicates the drug concentration at which the reaction rate is half of Vmax (average Km for phenytoin is ~4 mg/L).
Metabolism approaches Vmax at therapeutic concentrations, leading to changes in clearance as the dose changes.
The formula for clearance in this context:
[ \text{Clearance} = \frac{V{max}}{Km + [S]} ]
where [S] is the plasma concentration of the drug.
Challenges in Nonlinear Pharmacokinetics
With nonlinear kinetics:
A small increase in dose can lead to a significant increase in concentration, making it difficult to correlate dose to effect.
Variability due to genetic polymorphisms affects drug metabolism between individuals.
Drugs like phenytoin require therapeutic drug monitoring to avoid toxicity due to their narrow therapeutic index.
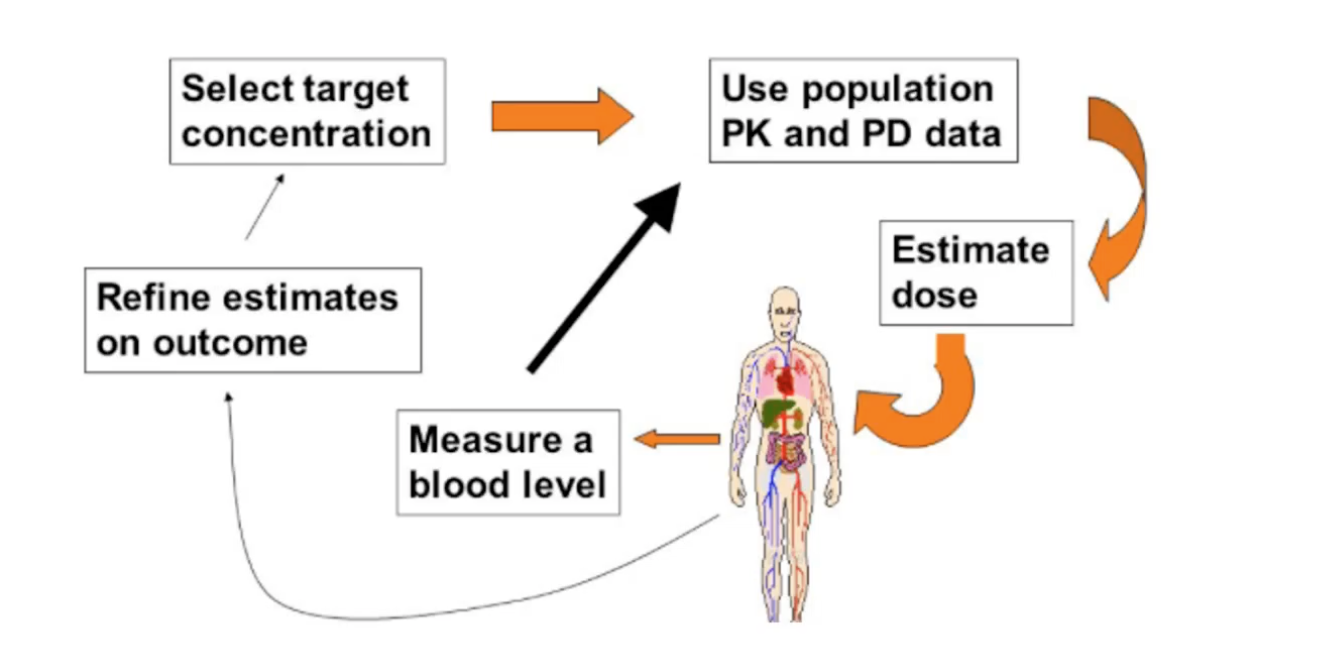
Therapeutic Drug Monitoring (TDM)
TDM is crucial for managing drugs with nonlinear pharmacokinetics, allowing for:
Monitoring plasma concentrations to maintain effective dosing without toxicity.
Assessing changes in individual metabolism (e.g., patient-specific Vmax).
Trough levels (measured just before the next dose) are optimal for steady state estimation.
Important scenarios for TDM include:
Inadequate seizure control.
Side effects or toxicity.
Influence of drug interactions or changes in patient condition (e.g., pregnancy).
Case Study: Phenytoin
Example of phenytoin's pharmacokinetics:
Average bioavailability is complete but absorption is slow.
Narrow therapeutic range (10-20 mg/L).
Therapeutic effects and toxicity must be closely monitored to avoid reaching concentrations above the therapeutic margin.
The relationship between dose and concentration is disproportionate, emphasizing the need for constant adjustment and monitoring of dosages.
Calculations in Nonlinear Pharmacokinetics
To calculate oral dose rates and steady state concentrations:
For drugs displaying nonlinear kinetics, use the Michaelis-Menten equation:
[ \text{Oral Dose Rate} = \frac{V{max} \times C{ss}}{Km + C{ss}} ]
where C_ss is the desired steady state concentration.
Example scenarios illustrate how slight changes in dose can lead to significant shifts in plasma levels.
Clinical Implications
Always aim for the midpoint of the therapeutic range due to the unpredictability of drug response.
Regular follow-up and assessment of response to treatment and serum drug levels are vital for optimal management.
Newer anticonvulsants generally demonstrate linear pharmacokinetics and wider safety margins than older drugs like phenytoin.
Conclusion
Understanding nonlinear pharmacokinetics is essential for effective drug management, particularly for drugs with narrow therapeutic ranges.
Effective therapeutic monitoring and dose adjustment can mitigate risks of toxicity and improve therapeutic outcomes for patients.