Chapter 9.3-9.5
9.3 Molecularity of a Reaction
Rxns rarely take place as shown by the balanced equation.
Most rxns take place through multiple steps
Overall rxn = sum of several steps; the sequence of steps by which a rxn occurs is called the mechanism of the rxn.
Description how molecules arroach each other, how bonds are formed or broken etc. is the reaction mechanism
Molecularity - number of particles in a single elementary step
no fractional number of particles only whole numbers (realistic numbers)
1, 2, or 3
Elementary step - a single step in the mechanism
Rate-determining step - the lowest step in the mechanism
Intermediate - the species which appears in one elementary step and is used up in another step
Does not appear in the overall rxn
Created in the rxn and used in the rxn
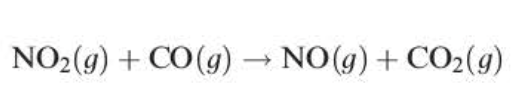
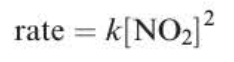
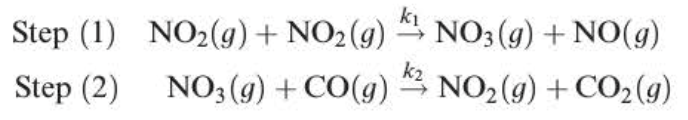
rate = k1[NO2]²
Unimolecular Reactions
Only one particle interacts; involves one reactant molecule in elementary step
Examples: isomerization, decomposition, ring opening, racemization
Follows first-order rate law
The reactant molecule collides with another molecule and becomes energetically excited at the expense of the other
Bimolecular Reactions
any elementary step that involves two reactant molecules in elementary step
Termolecular Reactions
involving three reactant molecules in elementary step
probability of this quite small
Ex: Nitric oxide and halogens reacting
M - collider molecule, usually inert gas
9.4 More Complex Reactions
Reversible Reactions
All rxns are reversible to an extent.
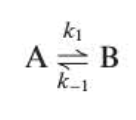
For the rate of the rxn equation
Principle of microscopic reversibility: at equilibrium, the rates of the forward and reverse processes are equal for every elementary reaction occuring
A→B is balanced by B→A
Equilibrium cannot be maintained by a cyclic process
The transition state for the forward and reverse reaction is identical
Consecutive Reactions
The product from the first step becomes the reactant for the second step
Steady state: assume the same amount of B being produced is equivalent to the amount of B being used up
Applicable if the k2 » k1
b/c b is being produced slowly and immediately used up when produced
[A]0 = [A] + [B] + [C]
[C] = [A]0(1-e-k1t)
[A] = [A]0e-k1t
[B] = k1/k2 [A]0e-k1t
Chain Reactions
9.5 The Effect of Temperature on Reaction Rate
Four types of temperature dependence for rate constants
a) Rate increases with increasing temperature (most typical)
b) Rate initially increase with temperatures, reaches maximum, then decreases (ex: enzymatic catalysis)
c) Rate decreases with increasing temperature (ex: some exothermic rxns)
d) Rate increases steadily with temperature, then increase very rapidly (ex: chain reaction)
The Arrhenius Equation
Describes the effect of temperature on the rate constant
Ea - activation energy: minimum amount of energy in order to initiate a chemical rxn
Ae - frequency factor (pre-exponential factor) : frequency of collisions between the reactant molecules
The power of e is raised to —> The fraction of molecular collisions having the enrgy equal to or higher than activation energy