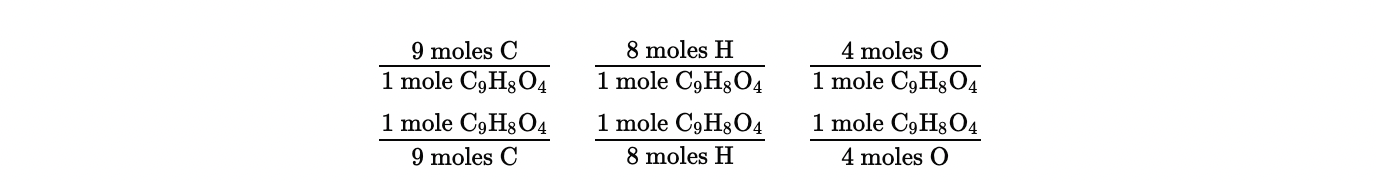Chapter 7
Avogadro’s Number
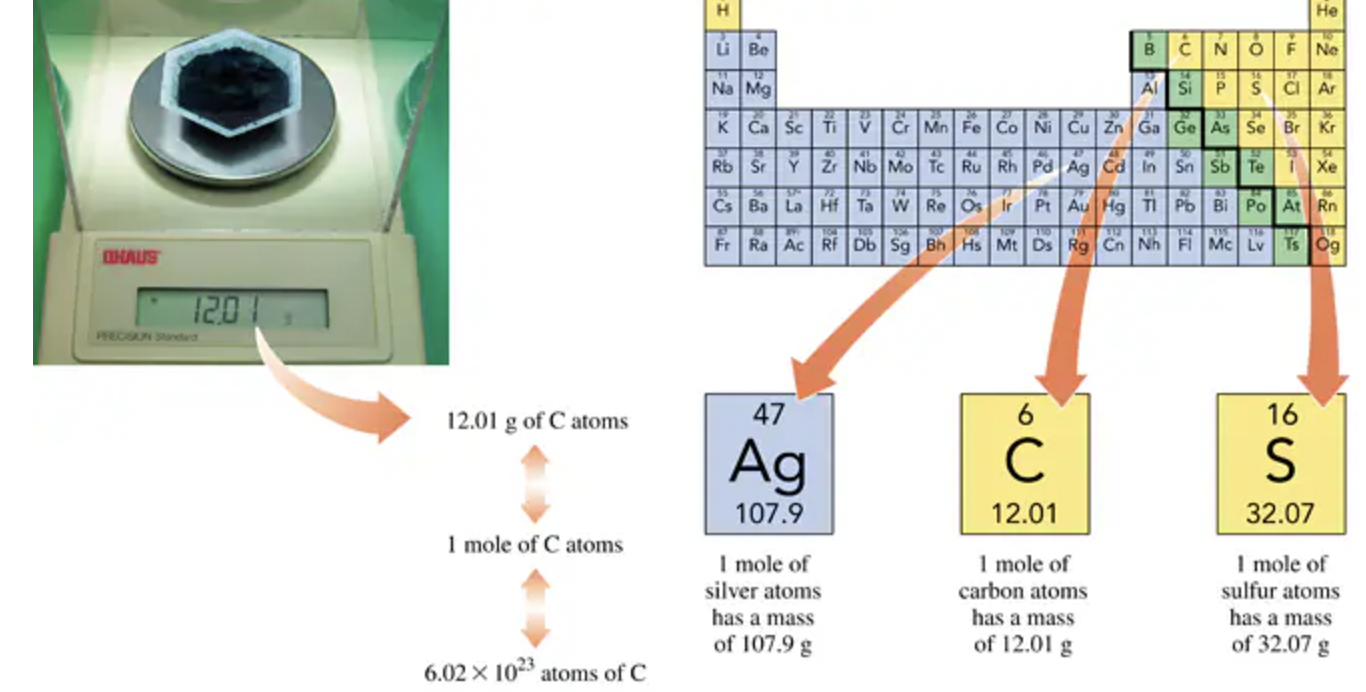
Avogadro’s number is a fundamental concept in chemistry.
Defined as: 1 mole = 6.02 × 10²³ items
This number allows for the counting of small particles such as atoms, molecules, and ions.
Named after Amedeo Avogadro (1776–1856), an Italian physicist.
Mole Representation
1 mole of an element contains 6.02 × 10²³ atoms.
Example: 1 mole of carbon = 6.02 × 10²³ atoms.
Example: 1 mole of sodium = 6.02 × 10²³ atoms.
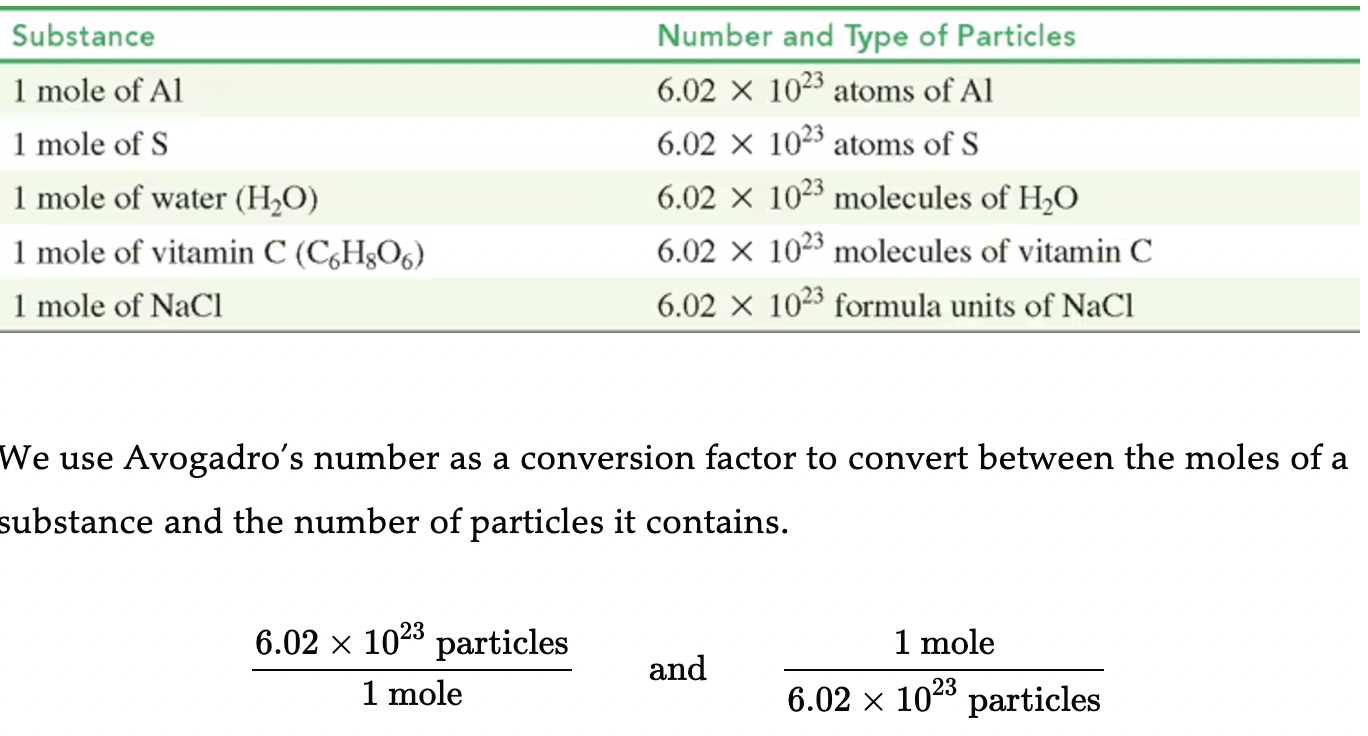
Conversion Factors Using Avogadro’s Number
Avogadro’s number can be expressed in various forms:
Equality: 1 mole = 6.02 × 10²³ particles
Conversion Factors: Conversion from moles to molecules involves multiplying or dividing by Avogadro’s number.
Converting Moles to Molecules
Identify given quantity (in moles) and calculate for molecules.
Set up a conversion plan.
Apply Avogadro’s number (6.02 × 10²³ molecules per mole).
Perform calculation to yield the final number of molecules.
Example Problem: CO₂ Conversion
Given: 0.50 mole of CO₂
Find: Number of CO₂ molecules
Plan: Use Avogadro’s number to find the conversion:
Calculation: 0.50 mole × 6.02 × 10²³ molecules/mole = X molecules.
Determining Atoms in Moles
For a sample with given moles of an element (e.g., Al):
Identify given quantity (in moles).
Write a conversion plan to determine number of atoms.
Use Avogadro’s number as a conversion factor (1 mole = 6.02 × 10²³ atoms).
Example Problem: Al Conversion
Given: 2.0 moles of Al
Find: Number of atoms in Al
Calculation: 2.0 moles × 6.02 × 10²³ atoms/mole = X atoms.
Moles of Elements in Compounds
The subscripts in a chemical formula (e.g., C9H8O4 for Aspirin) show the mole relationship.
Aspirin has:
9 moles of C
8 moles of H
4 moles of O.
Calculating Molar Mass
Molar mass of an element: quantity in grams that equals the atomic mass.
Example: Sodium (Na) has a molar mass of approximately 22.99 g/mole.
Maple masses are often rounded to two decimal places (0.01).
Example Calculation: Molar Mass of Li₂CO₃
Identify individual molar masses from the periodic table.
Adjust for number of moles indicated by subscripts in the chemical formula.
Total the masses to find overall molar mass.
Energy Considerations in Reactions
Exothermic: Reactions that release heat and have products with lower energy than reactants.
Example: C + 2H₂ → CH₄ + energy.
Endothermic: Reactions that absorb heat and produce higher energy products.
Example: N₂ + O₂ + heat → 2NO
Factors Affecting Reaction Rates
Reaction rates can increase with:
Higher temperatures
Greater reactant concentrations
The presence of catalysts.
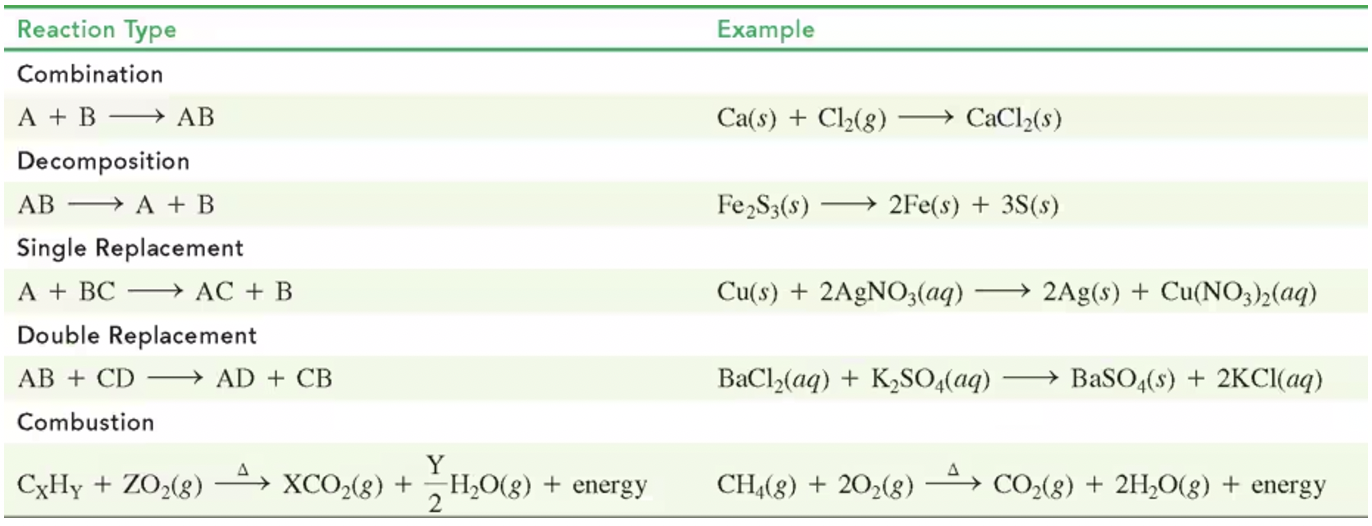
Types of Chemical Reactions
Combination Reaction: Two or more reactants combine.
Decomposition Reaction: One reactant breaks down into two or more products.
Single Replacement Reaction: One element replaces another in a compound.
Double Replacement Reaction: The ions of two compounds exchange places.
Combustion Reaction: Typically involves oxygen, producing heat, CO₂, and H₂O.