WEEK 4.1- Hair and Nail Analysis in Forensic Toxicology - Comprehensive Study Notes
Hair Analysis in Forensic Toxicology
Overview and applications
Hair analysis is routinely used to detect xenobiotics (drugs of abuse, pharmaceuticals, environmental contaminants, doping agents, etc.) across multiple domains: forensic science, traffic medicine, occupational medicine, and clinical toxicology.
Growing uses include pre-employment screening, forensic investigations, clinical applications, and doping control.
Human hair composition and structure
Composition (typical ranges):
Protein (mainly keratin): 65-95%
Water: 15-35%
Lipids: 1-9%
Minerals: <1%
Hair cycles and growth
Hair grows in cycles: growth (anagen) and quiescent phases (catagen and telogen).
Approx. 85% of ~1,000,000 hair follicles on the adult scalp are in growth phase at any time.
Growth rate:
From data: ~0.22-0.52 mm/day, which is equivalent to ~0.66-1.56 cm/month.
-一般 growth rates vary by hair type, physiology, and anatomical location.The Society of Hair Testing recommends using an average head-hair growth rate of
Hair types and classification
Types:
Vellus: fine, short, non-pigmented; covers most of the body surface.
Intermediate: features of both vellus and terminal; found on arms and legs of adults.
Terminal: coarse, long, pigmented; influenced by hormones; includes scalp, beard, eyebrows, eyelashes, armpits, pubic areas.
Ethnic/classification and subgroups
Classified as African, Asian, European.
8 subgroups based on straight or curly hair; assessment considers curve diameter, curve index, and number of waves.
Why use hair as a forensic matrix?
Advantages:
Collection is non-invasive and relatively easy to perform.
Can be done under close supervision to prevent adulteration or substitution.
Provides historical/chronological information on drug exposure, limited mainly by hair length.
Relatively stable and easy to store.
Key limitation: interpretation depends on growth history and incorporation mechanisms.
Considerations affecting hair analysis
Cuticle concerns: outer protective layer (cuticle) can be damaged over time, compromising surface integrity and exposing inner layers.
Stability depends on hair morphology and physicochemical properties of analytes.
Concentrations can decrease due to natural wash-out.
Washing and cosmetic treatment effects
Daily shampooing has little effect, but drying, curling, or straightening with heat damages the cuticle (loss or contamination).
Cosmetic treatments (dyeing, perming, lightening/bleaching) decrease drug concentrations by approximately
Hair colour variation (melanin content) may bias results; studies suggest darker hair may incorporate alkaline drugs more readily.
Results across studies are inconsistent and sometimes unclear.
Mechanisms of drug incorporation into hair
Mechanisms (at least 3):
1) From blood during formation of hair in the follicle (in situ incorporation into growing shaft).
2) Transfer from substances in sweat and sebum along the hair surface.
3) Incorporation from the external environment (external contamination).The relative contribution of each route varies by drug and is not fully understood; effects differ among drugs.
Basic drugs (e.g., amphetamines, cocaine) tend to incorporate more than neutral or acidic drugs due to pH and binding dynamics.
Factors influencing incorporation and binding: pKa, chemical structure, molecular size, lipophilicity, protein binding capacity, and melanin affinity.
pH gradient concept
Plasma pH ~7.3, while melanocytes/keratinocytes in hair follicle ~3-6, influencing ionization and trapping of drugs.
Lipid solubility is critical for passage into hair.
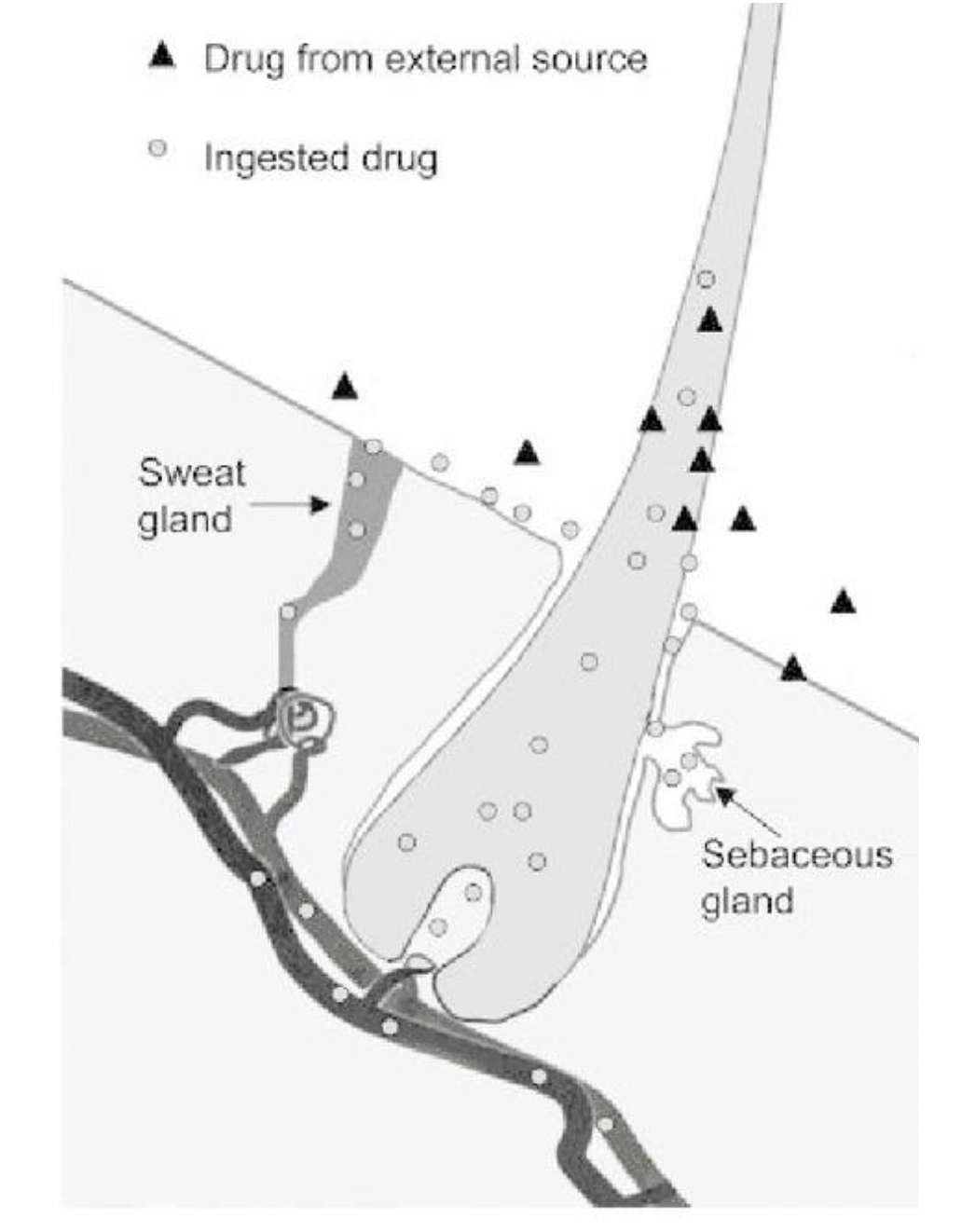
Drug movement into hair and interpretation considerations
Drug concentration in hair is expected to correlate with concentration at hair synthesis time, but does not always match blood/metabolic profiles (e.g., parent drug may predominate in hair).
Hair pigment effects: pigmentation can influence drug uptake and binding.
Non-pigmented hair uptake can still occur, but overall incorporation biased toward pigment-containing hair.
Overall: hair is a unique matrix with distinct affinity and binding mechanisms for different drugs; interpretation requires understanding the specific drug’s properties.
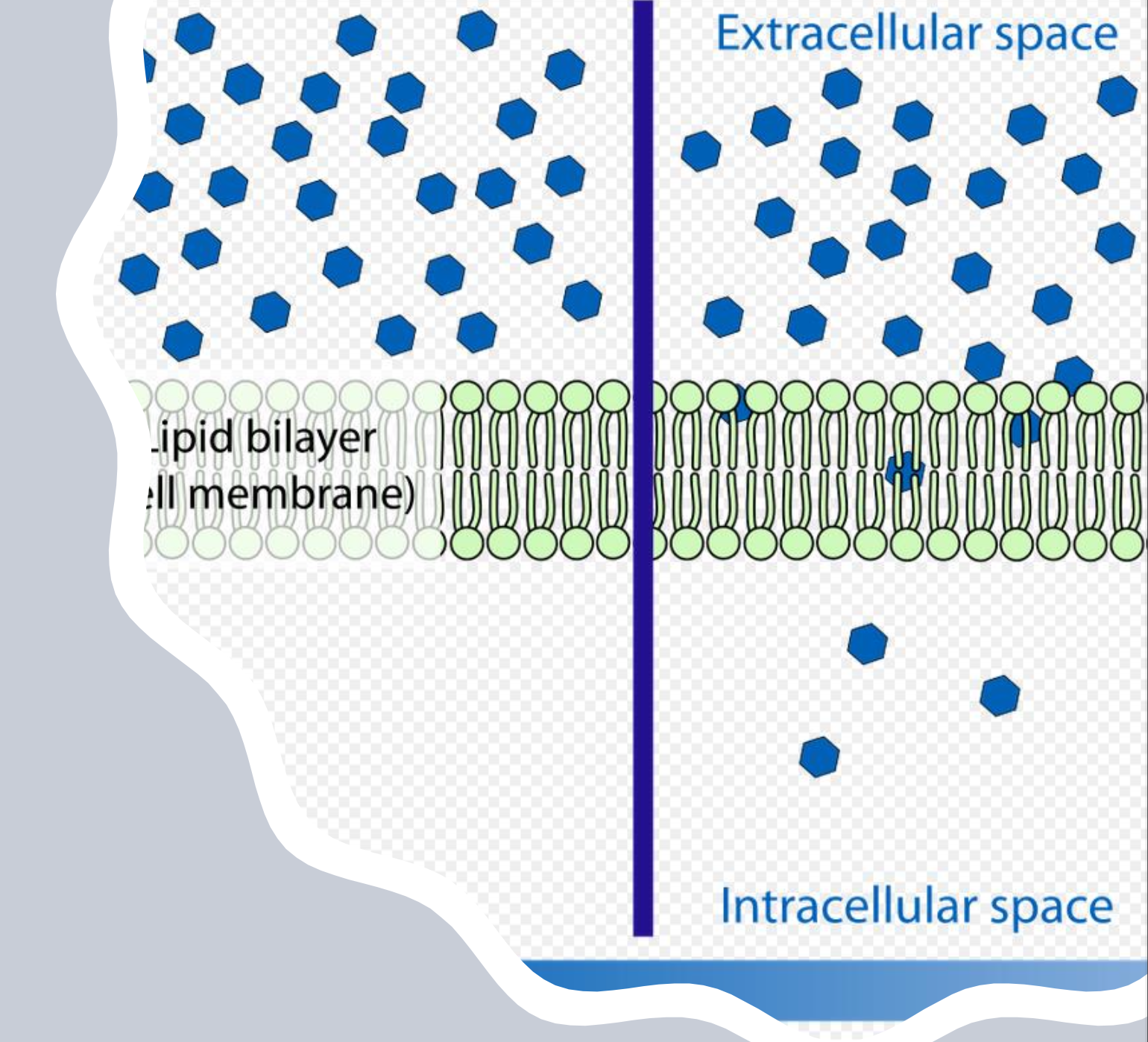
Practical testing considerations
Preferred sampling site: scalp hair, typically from the vertex posterior (back of the head).
Rationale: fastest growth rate with less variability; highest percentage of follicles in anagen; fewer age/sex influences.
Timescale interpretation
Variation in growth rate by age and individual; practical timescale estimation must consider subject age and growth variability.
It may take roughly for growing hair to reach the surface.
Analytical approaches and limitations
Testing method commonly used: Gas Chromatography/Mass Spectrometry (GLC/MS) with tandem MS often used for confirmatory testing.
Segmental hair analysis
Typically analyzes hair segments of to obtain a time course.
Dose (amount) and hair concentration do not have a simple direct relationship; quantitative correlation is not feasible.
Qualitative interpretation: concentrations are categorized as high/medium/low based on population data for the month prior to sampling.
Limitations: cannot prove impairment or intoxication at a specific moment; cannot establish degree of addiction.
Visual representations include conventional segmental analysis of bundles of hair and micro-segmental analysis of a single strand (reference figures in the literature).
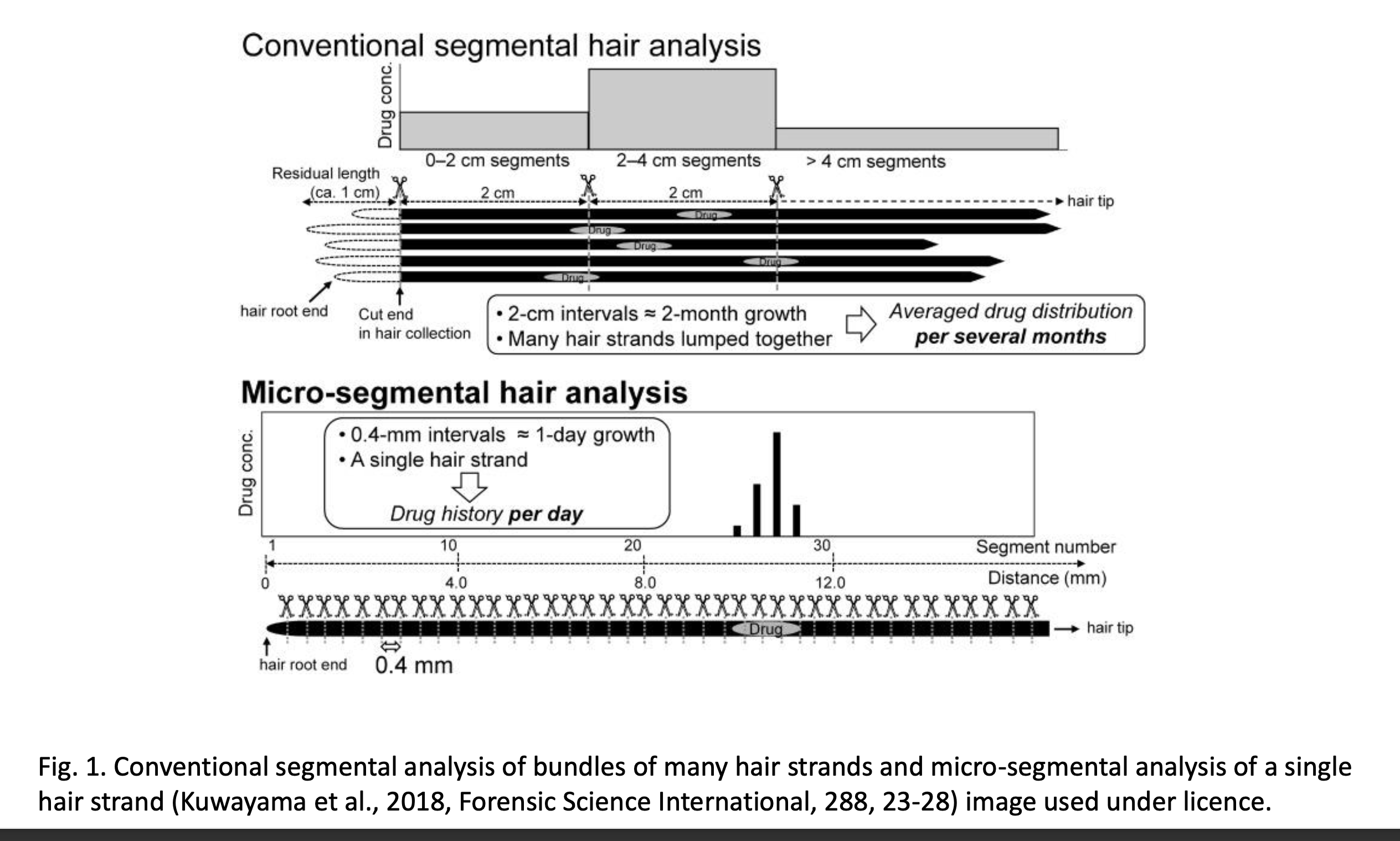
Real-world interpretation, errors, and guidelines
The most crucial issue in hair analysis is avoiding technical and evidentiary false positives.
Technical false positives arise from errors in collection, processing, and analysis.
Evidentiary false positives arise from passive exposure to drugs (environmental contamination).
Environmental contamination considerations
Passive exposure scenarios include drug-containing smoke, direct handling of drugs, and touching surfaces contaminated with drugs or another person’s hair.
Washing protocols
Proper washing procedures can remove surface contamination and help distinguish whether drugs originate from the growing hair follicle or surface fluids.
SoHT guidelines outline washing to remove gross surface contamination; selective removal of surface drugs while preserving incorporated drugs is unlikely.


Applications in forensic contexts
Verifying history of drug exposure over time; exposure history dating back to the length of hair available.
Alcohol: hair analysis cannot diagnose acute binge drinking but can reflect longer-term use.
Programmatic uses: monitoring detox programs, driver’s licences, gun licences, and child custody cases.
Doping control: major use is to identify false-negative results in blood/urine tests and to document history/frequency of drug intake via segmental analysis; longitudinal hair analysis has been proposed as a solution for documenting doping.
Hair is not universally accepted by all sports bodies; IOC/WADA acceptance is variable; in many jurisdictions, hair analysis is admissible in court even if not officially endorsed by IOC/WADA.
Special populations and scenarios Children
Hair testing in children presents challenges: distinguishing systemic incorporation from external deposition (smoke, dust, surfaces).
Children are not simply small adults; pharmacokinetics and toxicity vary across pediatric ages (ontogenic differences).
Trans-placental transfer of drugs and accumulation in fetal hair has been demonstrated.
Hair findings in newborns correlate with maternal drug consumption during pregnancy.
Postmortem toxicology
Urine and blood remain matrices of choice postmortem; hair can be useful when these are compromised or unavailable.
Hair analysis can help with long-term exposure assessment after death, especially if there was a period of abstinence or tolerance loss.
Drug-facilitated crimes (DFC)
Involves administering a substance to impair behavior or consent; includes drug-facilitated sexual crimes, elder abuse, and child abuse cases.
Common drugs include CNS depressants such as benzodiazepines, hypnotics, sedatives, anesthetics, substances of abuse, and ethanol; GHB is particularly important due to rapid elimination in biological fluids.
Blood/urine are preferred matrices if accessible in a suitable timeframe; hair provides a longer window for documentation of exposure when samples are taken long after the incident.
For GHB, hair is a unique matrix for documenting exposure when collection occurs more than 8 hours after the offense.
SoHT guidance for DFC: ideally collect four locks of ~100 hairs each, within 4-6 weeks after the incident and again about 1 month later; prioritize screening for drugs commonly associated with DFC; practical constraints may lead to analyzing a single lock first.
Negative results and interpretation caveats
A negative hair result does not conclusively exclude drug administration or exposure; it can mean one of two things:
(a) The hair owner did not take or was not exposed to the drug.
(b) The assay was not sensitive enough to detect the drug.
Negative hair results should be interpreted in the context of urine/blood results and potential exposure histories.
Interpretation pitfalls include external contamination, cosmetic treatments, ethnic bias, or uncertainty about the source of incorporation; sequential analysis can indicate contamination if results are homogeneous across segments.
Conclusions for hair analysis
Hair analysis is a promising tool for long-term exposure assessment to drugs and pharmaceuticals, complementing blood and urine analyses.
Most drugs of abuse and pharmaceuticals can be detected in hair at pg/mg to ng/mg levels, requiring sensitive and specific analytical methods and optimized sample preparation.
Mechanisms of substance incorporation into hair remain unclear, and the influence of factors like nail polish removal on concentrations is not well understood.
There is a lack of standardized sampling techniques, pre-analytical and analytical methods, making cross-study comparisons difficult; self-reported drug-use data can be unreliable, impacting study quality.
Nail Analysis in Forensic Toxicology
History and scope
Historical use began in 1965 for arsenic intoxication detection in nails.
Subsequent applications expanded to metals (mercury, cadmium, copper, lead, zinc, iron, magnesium) and other substances.
In 1984, first case reported for detection of methamphetamine and amphetamine from nails.
Today, nails are used for detecting drugs of abuse, pharmaceuticals, and their metabolites; nails have also been used for therapeutic drug monitoring of antimycotics and detection of amphetamine-like substances, cocaine, and opiates.
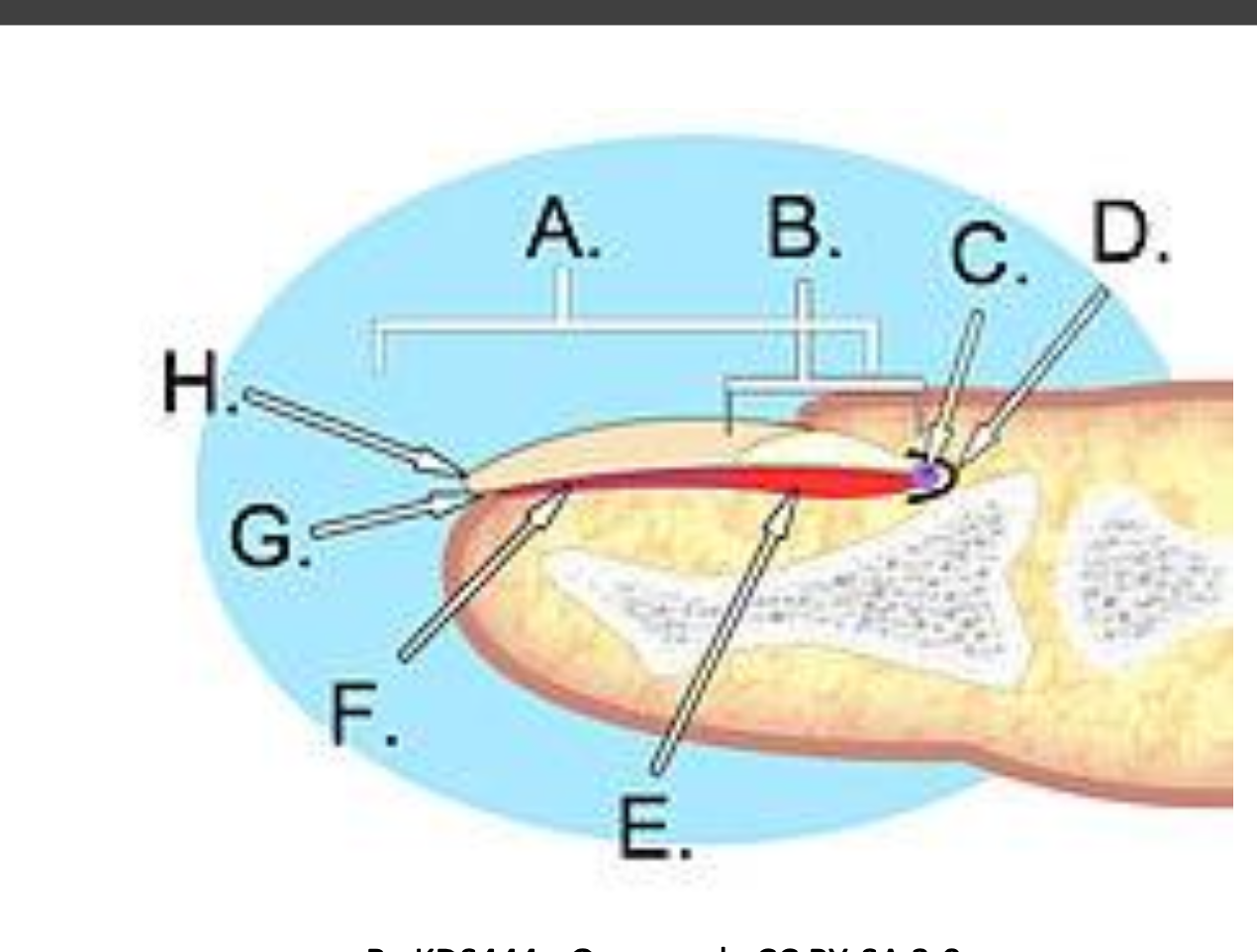
Nail anatomy and growth
Nails are made of keratin and serve to protect the distal digits.
Major components and regions:
Germinal matrix, lunula, nail bed, hyponychium, nail plate, nail folds.
Beneath the nail bed, a vascular network gives the nail its pink color; the lunula is the white crescent region.
Growth dynamics
Proliferation of the matrix involves distal growth of the nail.
Fingernail growth rate:
Toenail growth rate:
Nail growth slows with age, cold climate, disease, and malnutrition; faster in nail-biters.
Thickness increases as nail bed forms ventral layers during growth from the lunula to the free margin.
Rationale for using nails in toxicology
A large window of detection: about in fingernails and in toenails.
Non-invasive collection; does not require highly trained personnel; can be stored at room temperature.
Small sample size; collection can be supervised to prevent adulteration.
Drug presence in nails represents cumulative exposure; lower drug concentrations than hair but can reflect longer historical exposure due to slower growth.
Nails vs. hair: pros and cons
Pros
Nails are useful when hair is not available (alopecia, chemotherapy, newborns shortly after birth).
Drug stability is high in nails (less exposure to sunlight and heat) compared to hair.
Growth is continuous (not cyclical like hair), enabling accumulation and potential detection of longer-term exposure.
It is possible to detect parent compounds and metabolites in nails.
Collection is aesthetically more acceptable and less intrusive; no melanin-related bias since nails lack pigmentation that can influence results.
Cons
Generally lower drug concentrations than hair; interpretation can be more challenging due to variability.
Lower correlation with exact timing of drug consumption; timing estimates are less precise.
Considerations when testing nails
Drug concentration range is broad and depends on drug physicochemical properties, notably lipophilicity.
Interaction of drugs with the keratin matrix is influenced by drug properties; the exact mechanism is not fully understood.
Factors affecting results include amount of drug, level of exposure, individual characteristics (age, health, growth rate, gender), and cosmetic treatments.
Chemical and cosmetic treatments can interfere with analytes; bacterial or fungal infections can complicate interpretation.
Time delays between drug intake and detection in nails have not been fully characterized yet.
Mechanisms of drug incorporation into nails
The primary mechanism is believed to be incorporation from blood circulation during nail growth.
Other possible pathways: diffusion from biological fluids such as sweat, sebum, and saliva; minimal diffusion from the external environment.
Studies with antifungals demonstrate three mechanisms:
1) Contamination from sweat detectable within ~24 h after drug intake.
2) Incorporation from the nail bed (vertical growth) detectable after ~2 weeks.
3) Incorporation from the germinal matrix (horizontal growth), with average peak concentrations around ~3 months after drug administration.
Analysis methodology
Sampling methods: nail clipping with clippers or scraping with a blade of the upper nail layer; can also sample whole nails during autopsy.
Sample preparation generally involves: decontamination, homogenization, extraction, and cleanup.
Analytical techniques: chromatography followed by mass spectrometry (often LC-MS/MS or GC-MS/MS depending on analyte).
Applications and utility
Clinical and forensic applications include:
Monitoring of drug abusers and treatment programs; evaluating long-term adherence to therapy.
Workplace drug testing, driving/licensing, or other compliance programs.
Differentiating between low, moderate, and heavy users based on nail concentrations over time.
Special contexts: monitoring long-term antifungal therapy (antimycotics) and potential use in antidepressant/antipsychotic therapy monitoring.
In utero exposure and neonatal nails
Neonatal nails provide a non-invasive tool to assess intrauterine drug exposure.
Alcohol and drug use during pregnancy can cause miscarriage, prematurity, increased neonatal mortality, congenital anomalies, developmental delays, SIDS, etc.
Neonatal nails form during the second trimester and persist after birth; neonatal hair forms in the third trimester and is typically shed within 8-12 weeks after birth.
There is a useful correlation between maternal drug consumption during pregnancy and drug findings in the newborn’s nails.
Postmortem and forensic implications
Nails can aid postmortem toxicology when conventional matrices are compromised by decomposition.
In cases of drug-facilitated crimes or suspected chronic exposure, nails can provide long-term integration of drug use history.
Nails may be preferred when hair analysis results are difficult to interpret due to external contamination or cosmetic interventions.
Practical conclusions and limitations
Nail analysis is a promising tool for long-term drug exposure assessment, complementing blood and urine analyses.
Most drugs of abuse and pharmaceuticals can be detected in nails in the pg/mg to ng/mg range, requiring sensitive analytical methods and optimized sample preparation.
The mechanisms of drug incorporation into nails remain incompletely understood; external factors like nail polish or acetone removal effects are not fully characterized.
There is a lack of standardized sampling, pre-analytical, and analytical methods, which complicates cross-study comparisons.
The reliability of self-reported drug use remains a challenge for interpreting results.
