Lattice enthalpy
Chemical bonds
How can you compare the strength of a covalent bond? What data do you use?
Bond enthalpy
What would the equivalent be for an ionic bond? What is the added complication for an ionic bond?
You have to allow for the giant ionic lattice with an ionic compound
For an ionic compound we use lattice enthalpy
Lattice enthalpy is a measure of the strength of the ionic bonding in a giant lattice
Lattice enthalpy of formation, ΔLH0
Is the standard enthalpy change when one mole of a solid ionic compound is formed from its gaseous ions
e.g. Li+(g) + Cl-(g) → LiCl(s)
Lattice enthalpies of formation are always exothermic, i.e. have negative values
Lattice enthalpies cannot be determined directly, they must be determined using other data
To calculate lattice enthalpies we need to use a range of other pieces of data
Lattice enthalpy of dissociation, ΔLH0
Is the standard enthalpy change when 1 mole of a solid ionic compound dissociated into its gaseous ions
e.g. LiCl(s) → Li+(g) + Cl-(g)
Lattice enthalpies of dissociation are always endothermic i.e. have positive values
Lattice enthalpy of dissociation has the same magnitude as lattice enthalpy of formation but the opposite sign
Read question carefully to determine which one it’s referring to
Standard molar enthalpy change of formation, ΔfH0
Is the enthalpy change when 1 mole of a compound is formed from its constituent elements under standard conditions, all reactants and products in their standard states.
e.g.
Li(s) + 1/2Cl2(g) → LiCl(s) -408.6kJmol-1
Standard enthalpy change of atomisation, ΔatH0
Is the enthalpy change which accompanies the formation of 1 mole of gaseous atoms from the element in its standard state under standard conditions
e.g.
Li(s) → Li(g) +159.4kJmol-1
1/2Cl2(g) → Cl(g) +121.7kJmol-1
For a covalent element, the enthalpy change is ½ of the bond enthalpy of the element.
First ionisation energy, ΔLH0
The standard enthalpy change when one mole of gaseous atoms is converted into a mole of gaseous atoms each with a singe positive charge
e.g.
Li(g) → Li+(g) + e- +520kJmol-1
If you have a metal that wpuld be a 2+ ion then you would have to also use the 2nd ionisation energy as well
e.g.
M+(g) → M2+(g) + e-
First electron affinity ΔEAH0
The standard enthalpy change when a mole of gaseous atoms is converted to a mole of gaseous ions, each with a single negative charge
e.g.
Cl(g) + e- → Cl-(g) -348.8kJmol-1
Is a 2- ion then do the same as first ionisation energy - use 2nd electron affinity as well.
Note: whilst the first electron affinity is negative, all subsequent ones will be positive
Born Haber cycles
Hess’ law states that the overall energy change is the same no matter which route is taken
Born-Haber cycles are just an extension of this with more steps
An enthalpy scale is drawn on the Y-axis with higher enthalpies at the top
An endothermic reaction will have an arrow pointing upwards
An exothermic reaction will have an arrow pointing downwards
Every step in the Born-Haber cycle must be thoroughly labelled, with state symbols etc.
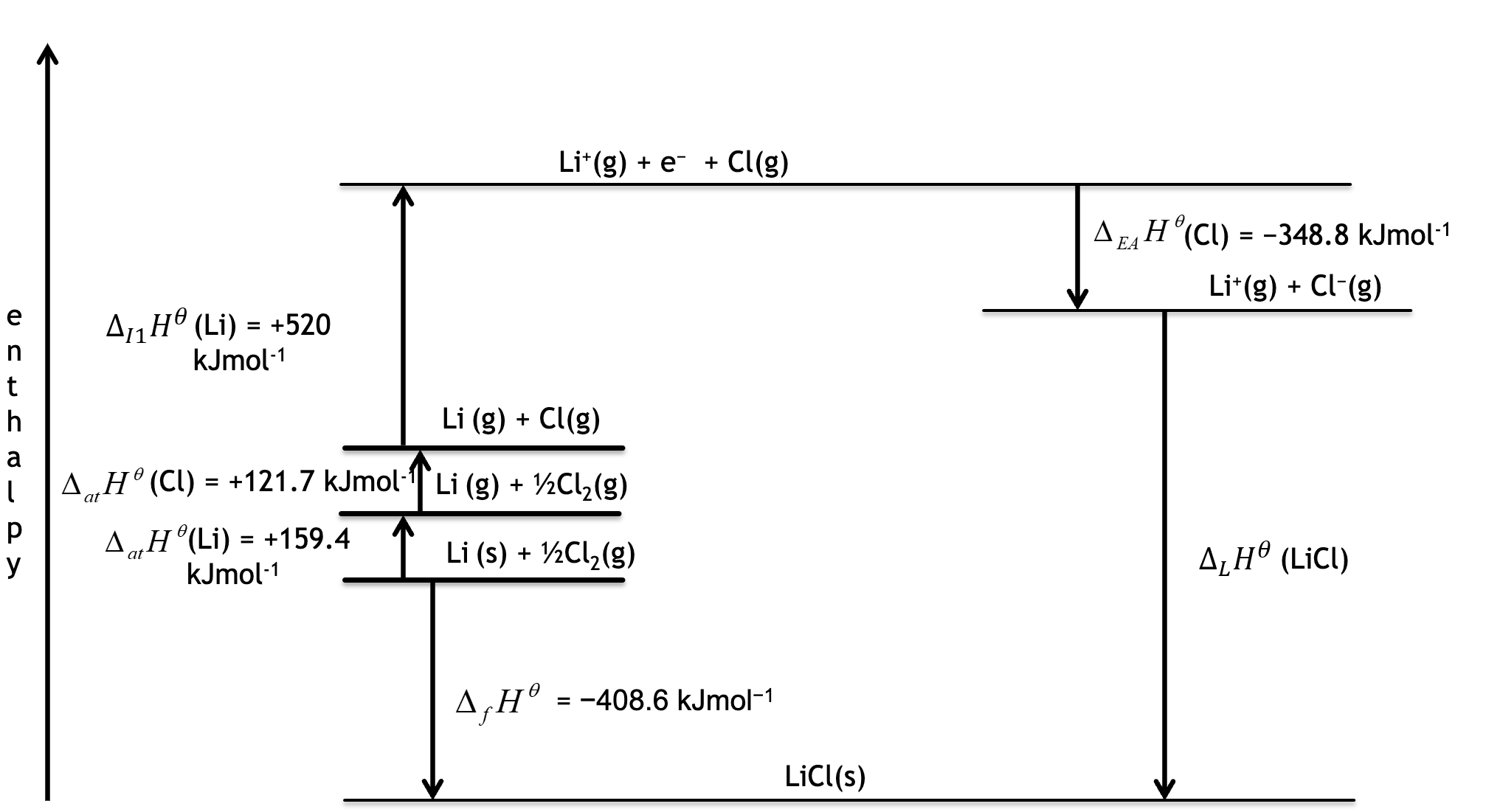
Use travelling pathway - get the answer -860.9kJmol-1