Ch6b: Gases
Composition of Air
Air is a mixture consisting primarily of the following gases:
Nitrogen: Approximately 78%
Oxygen: Approximately 21%
Argon: Small amounts
Carbon dioxide: Small amounts and other trace gases
Behavior of Gases
Air can be treated as a single gas for calculation purposes, despite being a mixture.
Key properties to consider:
Pressure (P)
Volume (V)
Temperature (T)
Using the Ideal Gas Law, the total number of moles in air can be calculated from these properties:
Even though the gases are different, if we know P, V, and T, we can determine the number of molecules of each gas component.
Partial Pressure
Defined as the pressure that a single gas in a mixture would exert if it occupied the entire volume by itself.
Equation derived from: Pi = (na/ntotal)Ptotal = XiPtotal
The partial pressure of a component in a gaseous mixture is its mole fraction multiplied by the total pressure.
To calculate the partial pressure (P_i) of gas A:
Use the equation:
Pi = Xi times PtotalWhere:
Xi is the mole fraction of the gas
Ptotal is the total pressure of the mixture
Dalton's Law of Partial Pressures states:
P{total} = P1 + P2 + … + PnThis means individual gases in a mixture can be considered independently for calculations.
Mole Fraction
The mole fraction (X) of a gas in a mixture can be calculated as: Xi = Pa/Ptotal = [na(RT/V)]/[(ntotal)(RT/V)] = na/ntotal
Where:
na = number of moles of gas A
ntotal = total number of moles of gas in the mixture
Alternatively, the mole fraction can be obtained from the percentage by volume of the gas, divided by 100.
The number of moles of a component in a mixture divided by the total number of moles in the mixture is the mole fraction (Xa).
For gases, the mole fraction of a component is equivalent to its percent by volume divided by 100%.
Example: Calculation of Mole Fraction and Partial Pressure
For nitrogen at 78% volume in air:
Mole fraction,
Given total pressure, , partial pressure of nitrogen is:
P{N2} = X{N2} times P_{total} = 0.78 times { 1atm}
For oxygen at 21% volume in air:
Mole fraction,
Follow similar calculations for argon and other gases based on their volume percentages.
Practical Example: Helios Air Mixture Calculation
A 12.5L container holds a mixture of helium and oxygen:
Given:
Mass of helium: 24.4 grams
Mass of oxygen: 4.3 kilograms (4300 grams)
Temperature: 298K
Conversion of grams to moles:
Moles of helium:
Moles of oxygen:
Total moles:
n{total} = n{He} + n{O2} = 6.05 + 134.375 = 140.425 mol
Calculating total pressure using the Ideal Gas Law:
P{total} = ntotal times R times {T}{V}Where:
R = 0.0821 L·atm/(K·mol)
Substituting values:
Calculation of Partial Pressure
Mole Fraction of Helium:
Partial Pressure of Helium:
P{He} = X{He} times P{total} = 0.0431 times P{total}
Collecting and Measuring Gases
Method of Collection:
Gases can be collected over water, resulting in a mixture of the gas and water vapor due to vapor pressure.
Vapour Pressure of Water at Different Temperatures:
Example at 25 °C: 23.78 mmHg.
Using total pressure measurement and subtracting vapor pressure gives the partial pressure of the collected gas.
Example: Oxygen Collection from Aquatic Plants
Total pressure of collected gas: 755 mmHg
Initial temperature: 293K (20°C).
The vapor pressure of water is looked up (e.g., 17.55 mmHg).
Calculating the pressure of O2:
P{O2} = P{total} - P{H2O}
Conversions and calculations lead to determining:
n{O2} = {P{O2} times {V}{R}{T}Finally, to find mass of O2 in g, multiply moles by its molecular weight (32 g/mol).
Gases in Chemical Reactions
In reactions involving gaseous reactants or products, we often specify the quantity of a gas in terms of its volume at a given temperature and pressure.
As we have seen, stoichiometry involves relationships between amounts in moles.
For stoichiometric calculations involving gases, we can use the ideal gas law to determine the amounts in moles from the volumes, or to determine the volumes from the amounts in moles.
When gases are at STP, use 1 mole = 22.4 L.
The general conceptual plan for these kinds of calculations is as follows:


Example: Synthesized Methanol
Methanol (CH3OH) can be synthesized by the reaction:
CO(g) + 2H2(g) → CH3OH(g)
What volume (in liters) of hydrogen gas, at a temperature of 355 K and a pressure of 738 mmHg, is needed to synthesize 35.7 g of methanol?
g Methanol to mol Methanol to mol H2 to VH2 using v=n(rt/v)
To calculate the volume of hydrogen gas required, we first need to determine the number of moles of methanol being produced using its molar mass (32.04 g/mol), which gives us approximately 1.11 moles of methanol. Since the reaction shows that 1 mole of methanol is produced from 2 mole of hydrogen, we will need 2.22 moles of hydrogen gas as well.
Then from mol H2, we get the volume using V = n(RT/P)
P = 738 mmHg × (1 atm/760 mmHg) = 0.970 atm
V = 2.22 mol(0.08 × 355K/0.970) = 66.90 L
This calculation demonstrates how to convert moles of hydrogen gas into volume under the given conditions, highlighting the application of the ideal gas law.
Kinetic Molecular Theory
Gases are made of tiny particles (atoms or molecules) that are far apart and move constantly in random directions.
The particles are very small compared to the empty space between them, and there are no significant attractions or repulsions between them.
Collisions are perfectly elastic — when particles collide, they may exchange energy, but no total energy is lost.
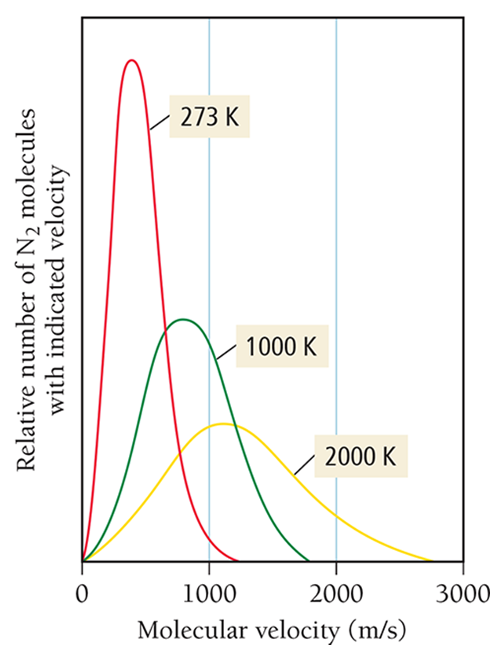
Temperature (in kelvins) measures the average kinetic energy of the gas particles — as temperature increases, particles move faster on average.
Not all particles move at the same speed, but at higher temperatures, the range of speeds shifts toward faster motion.
👉 In short: gas particles move freely, bounce off without losing energy, and their average speed increases as the Kelvin temperature rises.
The average kinetic energy of a particle is proportional to the temperature in kelvins.

Because the gas particles are constantly moving, they strike the sides of the container with a force.
The result of many particles in a gas sample exerting forces on the surfaces around them is a constant pressure.
P = F/A
Boyle’s Law: Kinetic Energy
Boyle’s law states that the volume of a gas is inversely proportional to the pressure.
Decreasing the volume forces the molecules into a smaller space.
More molecules will collide with the container at any one instant, increasing the pressure.
Charle’s Law: Kinetic Energy
Charles’s law states that the volume of a gas is directly proportional to the absolute temperature.
According to kinetic molecular theory, when we increase the temperature of a gas, the average speed, and thus the average kinetic energy, of the particles increases.
The greater volume spreads the collisions out over a greater surface area, so that the pressure is unchanged.
Avogadro’s Law: Kinetic Energy
Avogadro’s law states that the volume of a gas is directly proportional to the number of gas molecules.
Increasing the number of gas molecules causes more of them to hit the wall at the same time.
To keep the pressure constant, the volume must then increase (naturally)
Dalton’s Law: Kinetic Energy
Dalton’s law: The total pressure of a gas mixture is the sum of the partial pressures.
According to kinetic molecular theory, the particles have negligible size, and they do not interact.
Particles of different masses have the same average kinetic energy at a given temperature.
Because the average kinetic energy is the same, the total pressure of the collisions is the same.
Ideal Gas Law: Kinetic Energy
The kinetic molecular theory is a quantitative model that implies PV = nRT.
The pressure on a wall of a container occupied by particles in constant motion is the total force on the wall (due to the collisions) divided by the area of the wall.
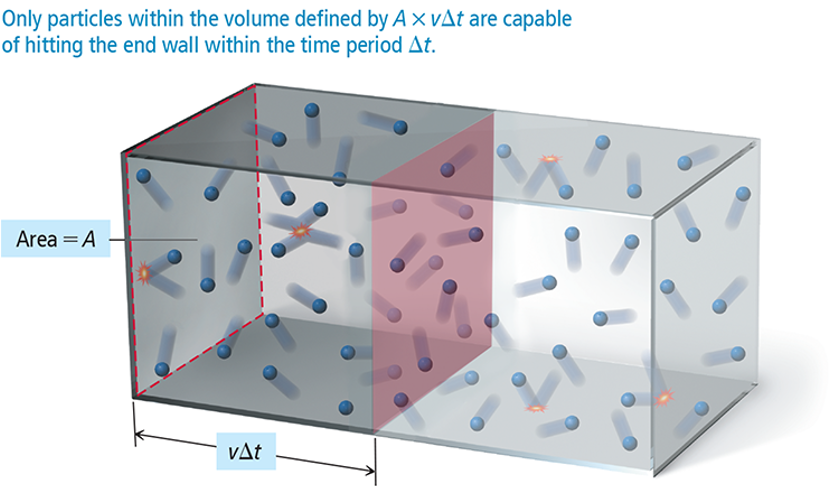
P = Ftotal/A
Kinetic Molecular Theory: Temperature and Velocity
Average kinetic energy of the gas molecules depends on the average mass and velocity.
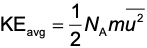
KE of a single molecule = ½ mv², where:
m = mass in kg
v = speed
NA = Avogadro’s number , which is approximately 6.02 x 10²³ molecules per mole
u² = average of the squares of particle velocities
Gases in the same container have the same temperature; therefore, they have the same average kinetic energy.
If they have different masses, the only way for them to have the same kinetic energy is to have different average velocities.
Lighter particles will have a faster average velocity than more massive particles.
Root Mean Square
Gas molecules all move at different speeds — some faster, some slower.
You can’t just take a regular average, because some particles are very fast.
Instead, rms is calculated by:
Squaring each particle’s speed
Taking the average of those squares
Taking the square root of that average
So rms speed is a kind of “effective average speed” that gives more weight to faster particles — perfect for calculating kinetic energy.
The root mean square velocity of a collection of gas particles is proportional to the square root of the temperature (K) and inversely proportional to the square root of the molar mass of the particles (in kg/mol)
R = 8.314 J/(mol•K)
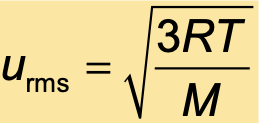
Kinetic Molecular Theory: temperature and velocity
Newton’s 2nd Law: the change of motion of an object is proportional to the force impressed; and is made in the direction of the straight line in which the force is impressed.
F = mass x acceleration
To have the same average kinetic energy, heavier molecules must have a slower average speed.
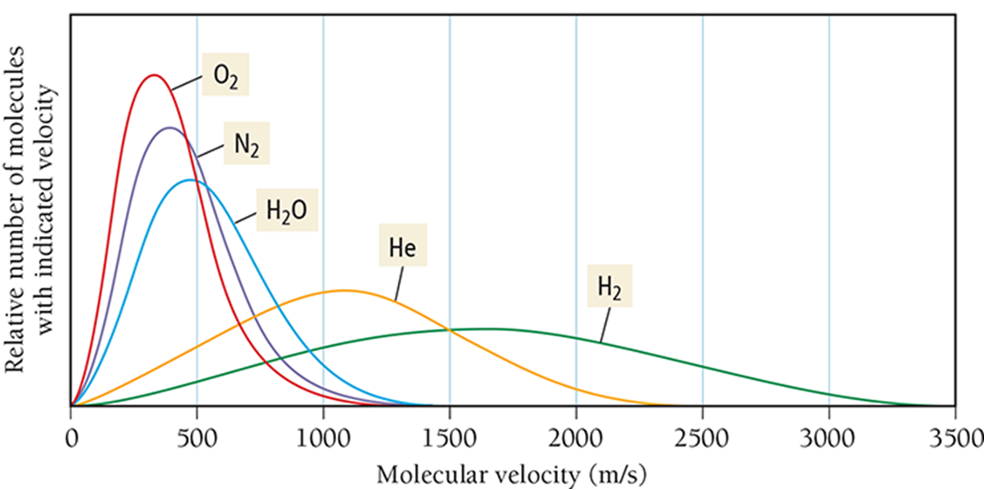
As the temperature of a gas sample increases, the velocity distribution of the molecules shifts toward higher velocity.
The distribution function “spreads out,” resulting in more molecules with faster speeds, as energy is distributed unevenly among molecules. Some molecules “get more energy” and move much faster, while others don’t speed up as much.
This uneven distribution causes the velocity distribution curve to spread out, with more molecules at higher speeds — the “tail” of the distribution stretches to the right.
So the average speed goes up, and the range of speeds becomes wider, even though every individual molecule is still moving.