C7: Acids, bases, salts
Acids: proton donors
Bases: proton acceptors
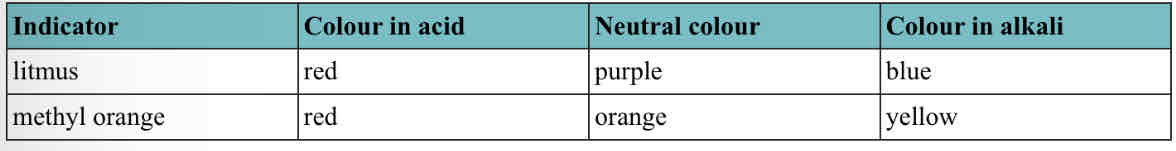
Indicators: substances that change color if they are put into an acid or alkaline solution.
Litmus and methyl orange:
Universal indicator: mixture of indication dyes

pH scale
Acids: pH < 7, more acidic → lower pH
Neutral substances: pH 7
Bases: pH > 7, more basic → higher pH
Bases and neutralization
Acids are a source of hydrogen ions, H+
Bases (or alkalis) are a sources of hydroxide ions, OH–
they react together in a neutralisation reaction, the H+ ions react with the OH– ions to produce water
acid + base → salt + water (neutralization)
net ionic equation: H+ (aq) + OH– (aq)⟶ H2O (l)
Most bases are insoluble in water
Alkalis: bases that are soluble in water
Characteristic reactions of acids
metal + acid → salt + hydrogen
Hydrochloric acid → chloride salt
Nitric acid → nitrate salt
Sulfuric acid → sulfate salt
Acid + base → salt + water
Acid + metal carbonate → salt + water + carbon dioxide
Oxides
Compounds made from one or more atoms of oxygen combined with another element
Acidic oxides:
Non-metal + oxygen
acid oxide + base → salt + water
Produce an acidic solution when dissolved in water
Eg. CO2, SO2
Basic oxides:
Metal + oxygen
basic oxide + acid → salt + water
Produce a basic solution when dissolved in water
Eg. CuO, CaO
Amphoteric oxides:
Can react with both bases/acids → salt + water
Eg. ZnO, Al2O3
Neutral oxides:
Oxides that do not react with acids/bases
Eg. N2O, NO, CO
Preparation of salts
Solubility of salts
All common sodium, potassium, ammonium salts are soluble
all nitrates are soluble
most chlorides and sulfates are soluble
all carbonates are insoluble
Preparing soluble salts:
Method A: Acid + solid metal/base/carbonate
Warm acid, add excess solid metal/base/carbonate until no more reaction is shown (no longer turns blue litmus paper red for base)
Filter mixture to get rid of excess into evaporating dish
Gently evaporate to concentrate salt solution (water bath/sand tray)
Let crystals from as solution cools → filter, wash, dry
Method B: Acid + alkali by titration (neutralization)
Acid is poured into a burette. A known volume of alkali is placed in a conical flask using a pipette.
A few drops of indicator (methyl orange/thymolphthalein) is added to the flask
Acid solution is slowly run into the flask until indicator just changes color
Swirl conical flask gently to ensure everything is mixed
Activated charcoal can be added to remove indicator and filtered out later
Anhydrous: dehydrated salt/powder
Preparing insoluble salts: precipitation reaction
Dissolve soluble salts in water and mix together using a stirring rod in a beaker
Filter to remove precipitate from mixture
Wash the residue with distilled water to remove traces of other solutions
Leave in an oven to dry