Energy Wavelength and Frequency Flashcards
Lyman series → Ultraviolet
Balmer series → Visible
Psdchan series → Infared
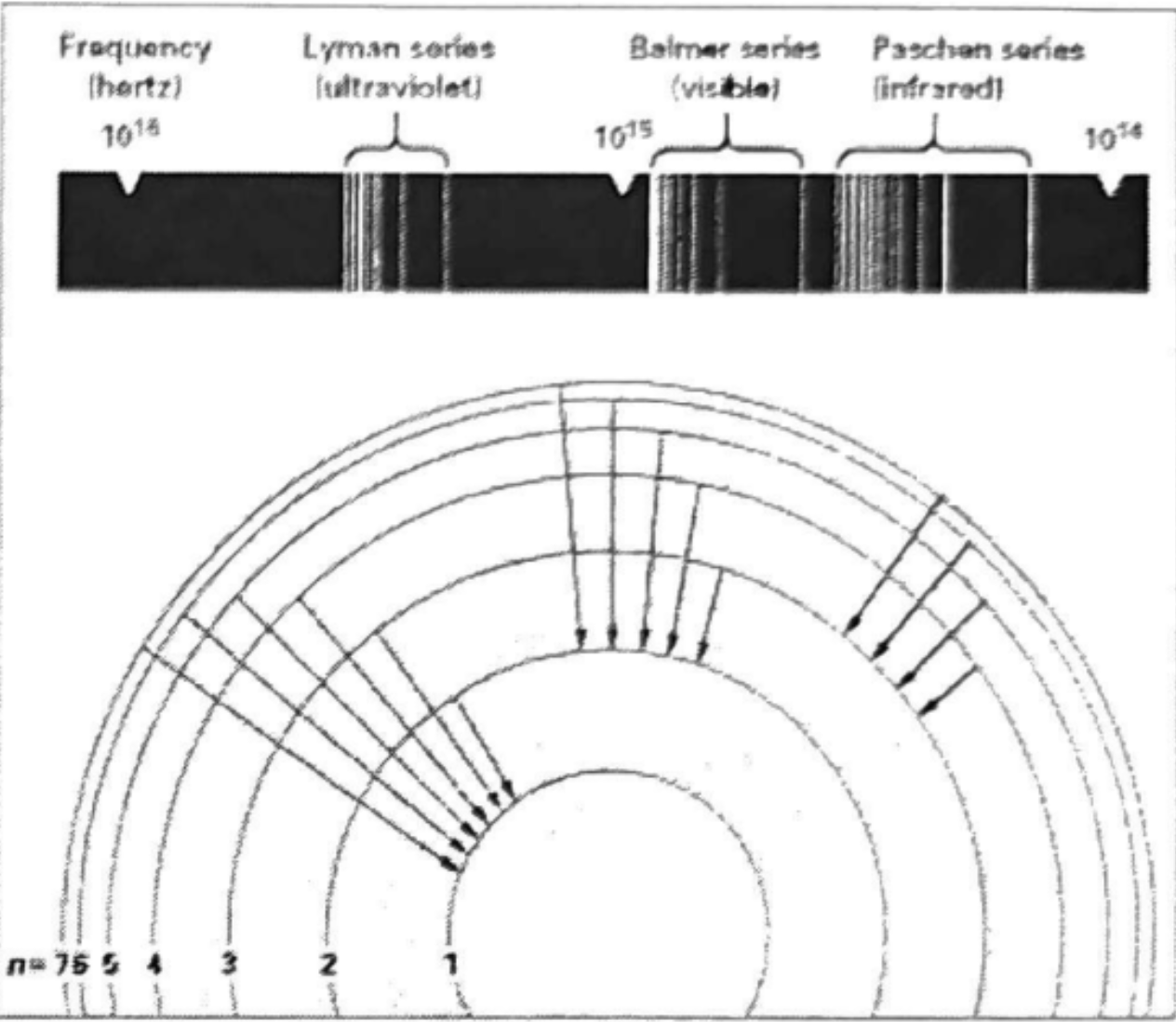
Bhor model key limitation
Only accurate for hydrogen molecules
Assumes only one electron
Does not account for electron–electron repulsion
Electromagnetic Spectrum (EMS)
Encompasses all types of electromagnetic energy
Gamma rays → Radio waves
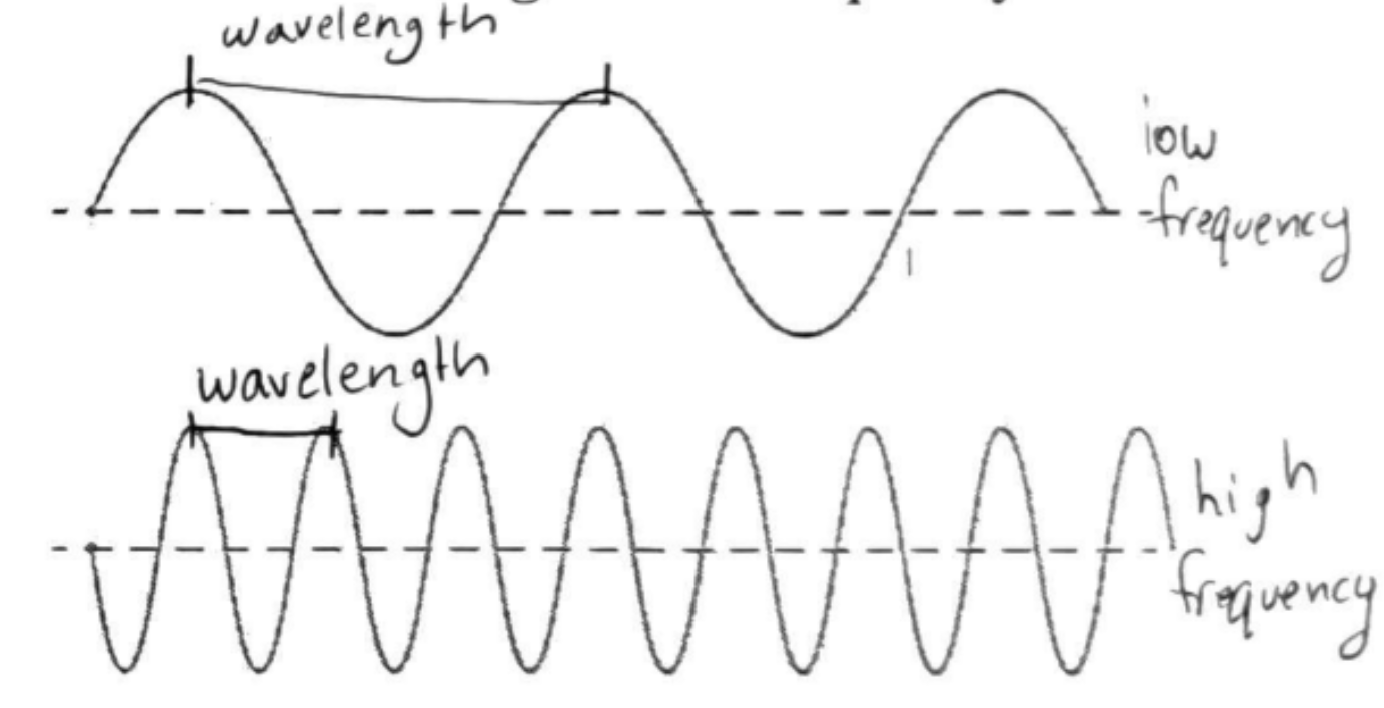
Wavelength (λ)
Distance from peak to peak
Meters (m)
Nanometers (nm): 1 × 10⁹
Frequency (ν)
How often waves oscillates
1/s
s⁻¹
Hertz (Hz)
KHz: 1×10³
MHz: 1×10⁶
GHz: 1×10⁹
THz: 1×10¹²
c=λν
C: Speed of light
3.00 × 10⁸ m/s
λ (wavelength): meters
ν (frequency): s⁻¹ or Hz
E=hv
E: Energy (joules)
h: Planck’s constant
6.63×10-34 J×s
v: Frequency s⁻¹ or Hz