Ch. 4.7 Electron Configuration and Orbitals
Orbital Diagrams
Orbital Diagrams use boxes to show how electrons:
are arranged in the orbitals of an atom
fill the orbitals within the same sublevel at one time, before pairing the electrons
Electron Configurations
Chemists use a notation called electron configuration to:
Indicate the placement of electrons in an atom
Show how electrons fill energy levels in order of increasing energy
write an abbreviated form using a noble gas to represent all electrons preceding it
Electron Configurations and the Periodic Table
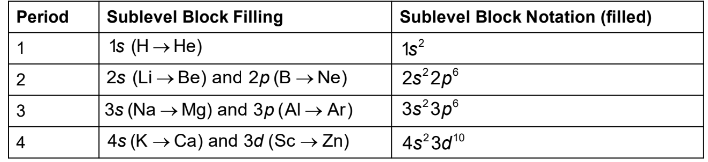
The electron configurations of elements are related to their positions on the periodic table. Different sections or blocks correspond to sublevels s, p, d and f
Blocks on the Periodic Table
The f block, the inner transition elements, is the two rows of elements at the bottom of the periodic table
There are 14 elements in each f block, because seven f orbitals can hold a maximum of 14 electrons
The s block contains elements in Groups 1A (1) and 2A (2) which means the final one or two electrons are in the s sublevel
The p block consists of elements in Group 3A (13) to Group 8A (18). There are six p block elements in each period, because three p orbitals can hold a maximum of six electrons
The d block, which contains transition elements, first appears after calcium (atomic number 20). There are 10 elements in the d block, because five d orbitals can hold a maximum of 10 electrons
The 4s sublevel fills before the 3d sublevel, because the 4s sublevel is slightly lower in energy than the 3rd sublevel
The 5s sublevel fills before the 4d sublevel
The 6s sublevel fills before the 5d sublevel
Guide to Writing Configurations Using Sublevel Blocks
Use sublevel blocks on the periodic table to write the electron configuration of an element
STEP 1
Locate the element (selenium in this example) on the periodic table. Selenium (atomic number 34) is in Group 6A (16) and Period 4
STEP 2
Write the filled sublevels in order, going across each period
STEP 3
Complete the configuration by counting the electrons in the last occupied sublevel block. Because Selenium is the fourth element in the 4p block, there are 4 electrons in the 4p sublevel

For Chromium (Cr), moving one of the 4s electrons to the 3d sublevel adds stability with a half-filled d subshell, and the resulting configuration is
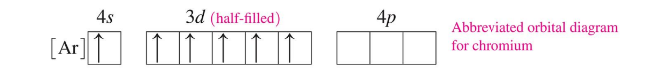
3d occupancy quick note: in the transition metals, the 3d subshell is filled after the 4s but can be rearranged in observed configurations due to stability considerations.