5.2 Enthalpy
- Enthalpy: (H) is the total amount of thermal energy in a substance.
- Can not be calculated but we have some theoretical values from observing changes and bond types.
- Enthalpy Change: (ΔH) is the energy absorbed from or released to the environment during a chemical or physical change.
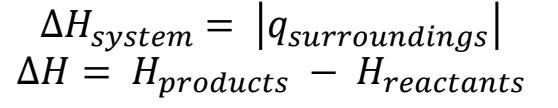
If:
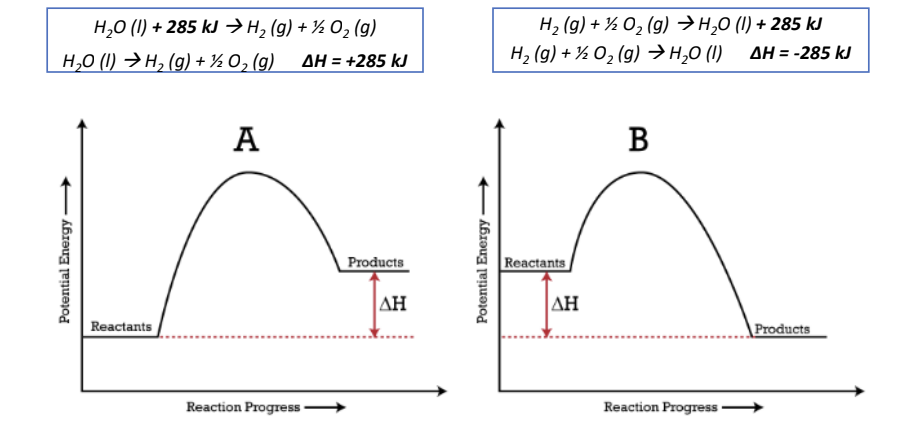
- ΔH > 0, endothermic reaction
- ΔH < 0, exothermic reaction
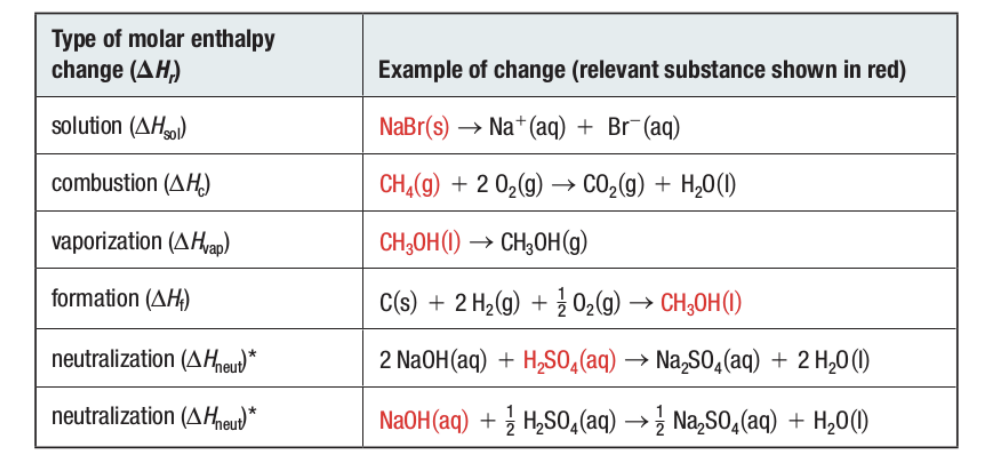
Molar Enthalpy Change
- Energy that it takes when 1 mol of a substance undergoes a physical/chemical/nuclear change.
- Measured in joules per mole (J/mol)
- ΔHr (r = reaction type)
- Ex: molar enthalpy of a physical change: vaporization of water
- H2O(l) → H2O(g)
- ΔHvap = 40.8 kJ/mol
Types of Molar Enthalpy Changes
Formula
- To obtain the change in energy involved in a given change, you must look up the molar enthalpy and apply it to a known amount of a substance.
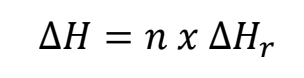
Where:
- n is the amount in moles
- ΔHr is the molar enthalpy change of the reaction
Representing Molar Enthalpy Changes
- Endothermic and exothermic reactions can be represented by thermochemical equations.
- These are balanced chemical reactions, which include the energy changes that occur in the system.
Thermochemical Equations
- A chemical equation that describes the enthalpy change of a reaction is called a thermochemical equation
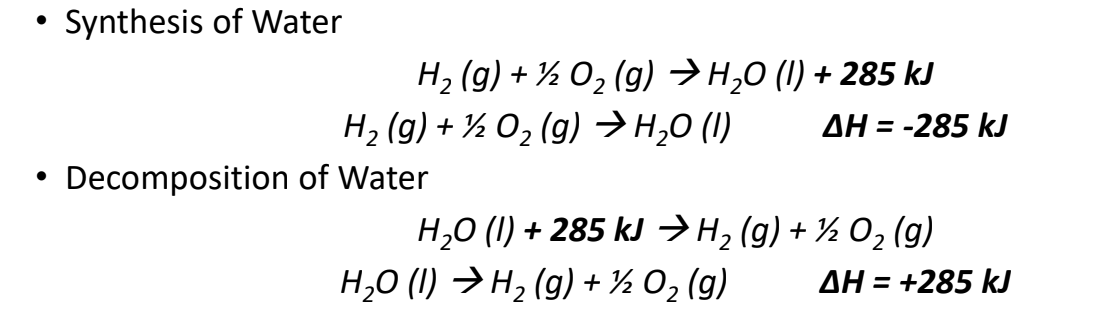
Potential Energy Diagrams