Chem Unit test
Investigating Matter
Matter: anything with mass and volume
Mass: amount of matter in a substance, measured in grams(g)
Volume: amount of space a substance occupies, measured in litres(L)
Density: Density compares the mass of a substance to its volume
mass/volume = g/mL or g/cm³
Changes in Matter
A) Chemical → New Matter
B) Physical → No new matter
A) Chemical Change
A chemical change is a change in matter when substances combine(or separate) to form new substances
These new substances have new properties
Things to look for: bubbles form, precipitate(a solid forms after mixing 2 liquids), colour change, smell and sound
B) Physical Change
A physical change is a change in matter when no new substance is formed
The substance may have a different appearance but its properties stay the same
This is because no chemical bonds are formed or destroyed in a physical change
Changes of state between solid, liquid, and gas do not produce new substances and are physical changes
The Kinetic Molecular Theory
Kinetic energy
The energy of motion
All particles are moving and have kinetic energy
The more energy the particles have, the faster they move and the farther apart they get
The Kinetic Molecular Theory states:
All matter is made up of tiny particles
Different substances have different particles
The particles are in constant motion
energy makes particles move
Solid: particles are close together and can only vibrate
Liquid: particles are further apart and slide by each other
Gas: particles are very far apart and move quickly
The Kinetic Molecular Theory and Changes of State
Changes of state: changing from one state to another
A change of state happens when energy(heat) is added or removed
Solids have the least amount of energy
Gasses have the most amount of energy
gas→liquid = condensation
liquid → gas = evaporation
liquid → solid = solidification
solid → liquid = melting
solid → gas = sublimation
gas → solid = deposition
Temperature and Changes of State
Boiling point: temperature to change from liquid to gas
Condensing point: temperature to change from gas to liquid
Melting point: temperature that changes solid to liquid
Freezing point: temperature that changes liquid to solid
Describing Matter
a) Physical properties (characteristics that can be observed or measured without changing its chemical identity of matter) of matter can be classified as:
qualitative properties: descriptions that are not measured with a number(quality)
quantitative properties: descriptions that are measured with a number(quantity)
qualitative | quantitative |
state(solid, liquid, gas) | temperature of melting/freezing/boiling/condensing |
colour | density |
magnetism | measurements |
b) Chemical properties: describe the ability of matter to react with another substance to form different substances
examples: combustibility, lack of reactivity, reactivity with oxygen, etc.
Classification of Matter
All substances can be classified as: i) pure substances or ii) mixtures
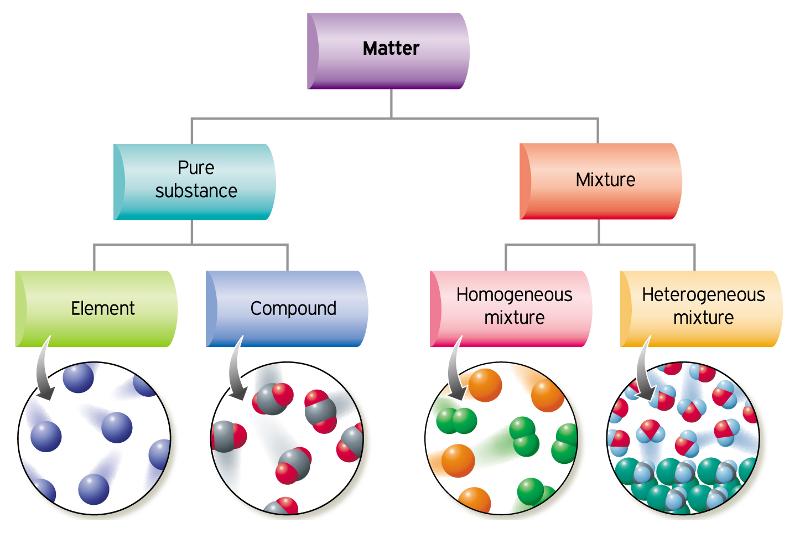
i) Pure substance: substance that is made up of one type of particle; cannot be separated by physical means
a) elements - made up of one type of atom; cannot be broken down further
eg. Oxygen(O), Hydrogen(H)
b) Compounds - substance made up of two or more different kinds of elements that are held together by chemical bonds; can be broken down into simpler substances
eg. Water(H2O), Salt(NaCl)
ii) Mixture: contains two or more pure substances; can be separated by physical means
eg. salt water
a) Homogeneous mixtures (solutions): mixed uniformly; cannot see their components
example: air(nitrogen, oxygen, hydrogen), steel(iron and other elements)
b) Heterogeneous mixture: have different components that you can see
ex. beach sand, salad dressing
The Atomic Theory
The Atomic Theory: All matter is made up of atoms, which are particles too small to be seen.
The atom: smallest unit of an element that has the properties of that element
Inside the Atom
Each element is classified by its own kind of atom
Each atom is made up of three subatomic particles:
proton
neutron
electron
Most of the mass of the atom is in the nucleus.
subatomic particle | electric charge | location in the atom |
Proton (p+) | + | nucleus |
Neutron (n0) | no charge | nucleus |
Electron (e−) | - | energy shells |
Elements
Elements are the building blocks of matter.
Chemical Symbols
Chemical symbol: symbolic system to describe all elements using letters from their Greek or Latin name
To give every element a unique symbol:
Every element is represented by the first letter of its name or by the first two letters of its name
When the first letter was previously used, a letter from the rest of the name is added
eg. C = carbon so Ca = calcium, Co = cobalt, Cr = chromium
The first letter is always capitalized and the second is lower case
Describing Elements
We can describe elements by:
physical properties: the appearance of a substance (eg. colour)
chemical properties: substance’s ability to chemically react with other substances to form new products
Elements are classified as a) metals, b) non-metals, c) semi-metals/metalloids
A) Metals:
most elements are metals
metals have the following properties:
property | characteristic |
lustre | shiny |
malleability | malleable(can be formed or shaped) |
ductility | ductile(can be stretched into wire) |
conductibility | good conductors of heat and electricity |
state | solid at room temperature(except mercury) |
density | usually denser than non-metals |
reactivity | active metals react with acid, and very active metals react with water |
B) Non-metals:
non-metals have the following properties:
property | characteristics |
lustre | dull |
malleability | likely to be brittle or shatter when struck |
boiling and melting points | usually lower melting and boiling points than metals |
state | often gases at room temperature |
conductibility | poor conductors of heat and electricity |
C) Semi-metals/Metalloids
elements that share some properties with metals and some properties of non-metals
property | characteristics |
lustre | shiny |
ductility/malleability | likely to be brittle or shatter when struck |
state | solid at room temperature |
conductibility | poor conductors of heat |
The Periodic Table and Chemical Properties
Mendeleev’s Table
Elements are organized based upon some important methods first introduced by Dmitiri Mendeleev:
Arranged elements by increasing atomic mass
Grouped elements into “families” based on similar properties(density, melting point)
Made the assumption that some elements were still undiscovered and left areas of the table blank
Modern periodic table is ordered by increasing atomic number
The Periodic Table
A chart that organizes elements according to their chemical and physical properties.
Each box in the periodic table gives the element’s:
name
symbol
atomic number
atomic mass
common ion charge(s)
Atomic number:
number of protons in an atom’s nucleus
identifies the element
pattern: atomic number increases by 1 across rows
Atomic mass:
average mass of all atoms of one element
measured in atomic mass units(amu)
pattern: atomic mass tends to increase along with the atomic number
Ion charge:
electric charge of the ion (atom gains/loses electrons)
negative charge = gained electrons
positive charge = lost electrons
if more than one charge is written = that an atom can gain/lose a different number of electrons
pattern:
elements on the left(metals) tend to have positive ions
elements on the right(non-metals) tend to have negative ions
elements in the same chemical family form ions with the same charge
Table Arrangement: Periods and Families
The modern periodic table organizes elements in groups and periods.
Period:
horizontal row
numbered 1 to 7
Chemical family:
vertical row called a group
elements in a group have similar physical and chemical properties
numbered 1 to 18
Elements of Groups 1, 2, 3 and 13 to 18 are called main-group elements or representative elements
Elements in Groups 3 to 12 are called transitional elements
Four well-known elemental families
Alkali metals(group 1):
Li, Na, K, Rb, Cs, Fr (NOT H)
highly reactive(reactivity increases down the group)
react with water and oxygen
low melting point
very soft
Alkaline earth metals(group 2):
Be, Mg, Ca, Sr, Ba, Ra
less reactive than Group 1
burns in air if heated and produces bright flames
reacts with water(not as violent as Group 1)
Halogens(group 17):
F, Cl, Br, I, At
non-metals
highly reactive
Noble Gases(Group 18):
He, Ne, Ar, Kr, Xe, Rn
Non-metals
most stable and unreactive of all elements
The Periodic Table and Atomic Theory
patterns in the periodic table occur as a result of regular changes in the structure of the atoms of elements
elements line up in columns because they are similar in the arrangement of their electrons
Niels Bohr:
suggested that electrons move around the nucleus of an atom in orbits or shells
Bohr model:
a diagram that shows how many electrons are in each shell surrounding the nucleus of an atom
electron shells:
regions surrounding the nucleus of an atom
each shell is a certain distance from the nucleus
Each shell holds a different number of electrons
Shell number | Maximum number of electrons in each shell |
1 | 2 |
2 | 8 |
3 | 8 |
4(or greater) | 32 |
Bohr model diagrams
How to draw a Bohr Model Diagram:
Write the element symbol
Check the periodic table to see how many electrons the element has (in a neutral atom: #protons = #electrons)
paying attention to how many electrons each shell can hold, figure out how many shells the atom has
draw a circle around the element symbol for each shell
use a dot to represent an electron. Starting with the inner shell, put the correct number of electrons on the diagram
Valence electrons and Chemical Families
Valence = outer
Valence electrons: electrons in the outermost shell
Valence shell: highest occupied energy level
important patterns:
elements in the same family/group have the same number of valence electrons
elements in the same period have valence electrons in the same shell
period number indicated the number of shells that contain electrons
Noble Gas Stability
when a valence shell is full, the atom is stable
noble gas atoms(group 18) have filled valence shells, which makes them virtually unreactive
atoms in other groups need to gain or lose electrons to become stable(full valence shell) during a chemical reaction
How Atoms Become Ions
Ion: atom that has gained or lost electrons, and as a result has a charge
Positive Ion:
if an atom loses electrons(loses negative charges), then the atom becomes positively charged
since atoms of metals have 1, 2, or 3 valence electrons, they want to lose their valence electrons to become positive ions
Negative Ion:
if an atom gains electrons(gains negative charges), then the atom becomes negatively charged
since atoms of non-metals have more than 3 valence electrons, they want to gain valence electrons to become negative ions
Reactivity
1) Reactivity of an element is linked to how close it is to having a full valence shell
most reactive elements: groups 1 and 17(elements are only one electron away from a full valence shell)
example: Sodium(group 1) easily gives up an electron, since it exposes the full valence shell underneath
2) Reactivity increases as you move down the family
example: group 1 metals: potassium is more reactive than sodium
Atomic Size Trends
1) Atomic size increases moving down a group
As you move down a group, elements have atoms with increasing numbers of energy shells
The greater the number of shells, the farther the valence electrons are from the nucleus, and the larger the atom
2) Atomic size decreases moving left to right across a period
elements have increasing numbers of electrons across a period
number of occupied valence shells stay the same, but the number of protons in the nucleus increases
attraction between valence electrons and the nucleus increases because a greater positive charge on the nucleus pulls more strongly on the electrons
therefore, the electrons are pulled more tightly towards the nucleus, leading the decreasing atomic size
Compounds
Compounds account for the huge variety of matter on Earth
Compound: a pure substance made up of 2 or more different kinds of elements that are held togther by chemical bonds