Lesson 12: Percentage Yield
Percentage Yield
Theoretical Yield is the amount or mass of product predicted based on the stoichiometry of the chemical reaction. \n
Actual Yield is the amount or mass of product actually collected during and experiment or industrial process \n
Reasons for a Difference between the Theoretical and Actual Yield
- Experimental procedure
- Impurities
- Competing side reactions
\
Percentage Yield is the ratio, expressed as a percentage, of the actual or experimental quantity of product obtained (actual yield) to the maximum possible quantity of product (theoretical yield) derived from a stoichiometric calculation. \n
Percentage Yield = actual yield x 100%
theoretical yield
\
“Slaked lime,” Ca(OH)2, is produced when water reacts with “quick lime,” CaO. If you start with 2 400 g of quick lime, add excess water, and produce 2 060 g of slaked lime, what is the percent yield of the reaction?
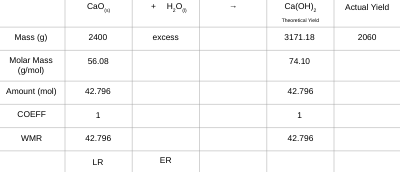
Percentage Yield = actual yield x 100%
theoretical yield \n
% yield = 2060 x 100%
3171.18 \n
% yield = 64.96%
\
% yield = 65% (sig figs)