The periodic table
1.1 groups and periods
Stile
creating new elements
The periodic table was first created in 1869 by Dmitri Mendeleev.
The most recent changes were in 2015 by shaming atoms together using a particle accelerators.
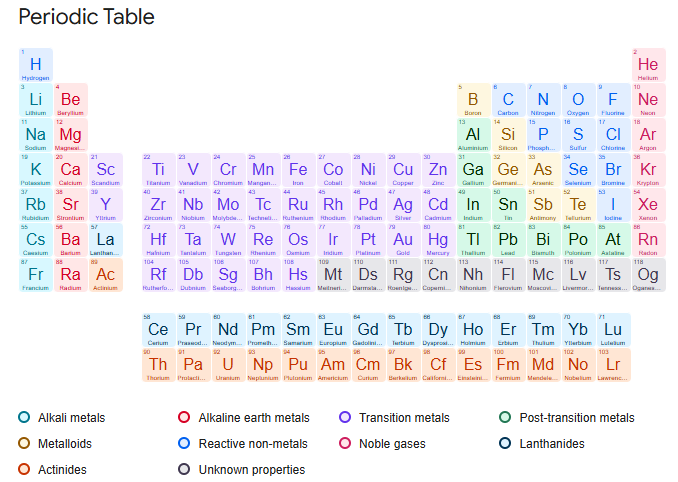
The organization of the periodic table
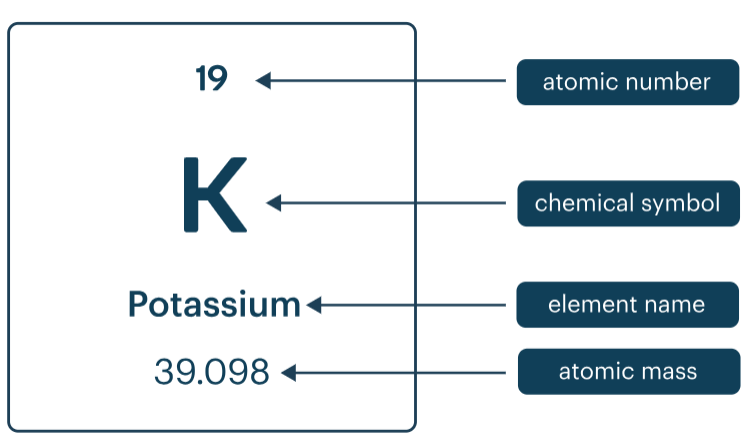
Elements are organised by atomic number.
the atomic number is the number of protons
The rows are called periods.
The columns are called groups.



4.1 Review of chemical equations
what is a chemical equation?
chemical equations are used to represent the reactants and products in a chemical reaction
chemical reactions occur if a chemical change occurs when substances re added together
this means a new substance has formed
chemical changes can be indicated by observations including colour changes, gas formations(bubbles), a solid forming, or heat/light energy being released or absorbed
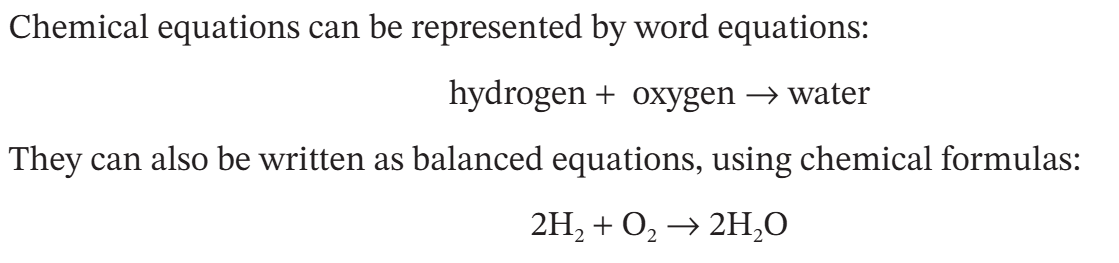
both types of equation show reactants on the left side
these are the chemical required to start the reaction
products that form in the reaction are found on the right
an arrow separates the reactants and products
chemical equations never include an equal sign
Balanced equation
a balanced equation provides detailed information about the atoms in a chemical reaction
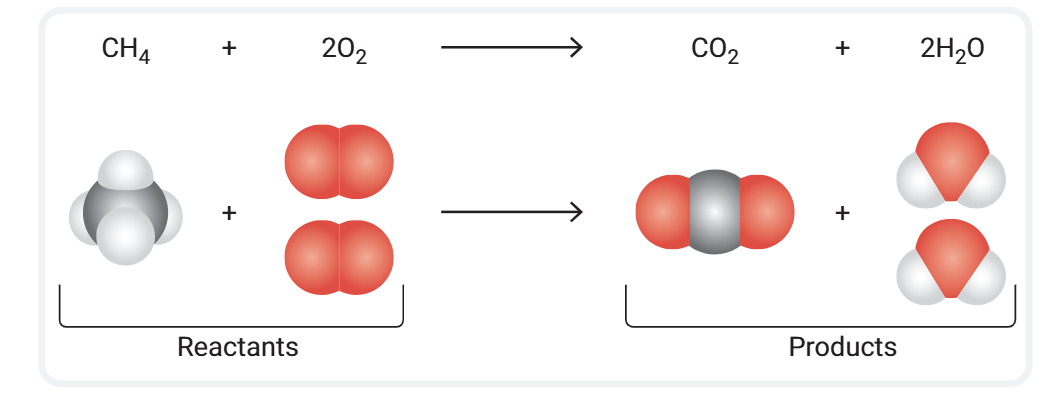
showing the reaction between methane (CH4) and oxygen (O2) to form carbon dioxide (CO2) and water (H2O)
the first thing you can see from the equation is the type and number of atoms in each reactant and product
for example (CH4) is methane ()the gas used for cooking and heating)
Understanding reaction rates
Explore factors influencing chemical reaction speed
chemical reactions vary in speeds (slow or fast) e.g. the rapid bubbling when hydrochloric acid reacts with magnesium is different from the “oxidation of iron” known as rusting
What is reaction rate?