Using resources
Ceramics, composites and polymers
Different materials have different properties, which make them useful for different things
Ceramics are hard, brittle, heat-resistant and corrosion-resistant materials
They are made by shaping and then firing a non-metallic material (like clay) at high temperatures
Mainly glass or clay ceramics (brick, china and porcelain)
Clay ceramics are made by shaping wet clay and heating to a high temperature in a furnace so it hardens
It has a high compressive strength - why it is used for building
Glass ceramics are mainly soda-lime glass
Made by melting a mixture of sand (silicon dioxide), sodium carbonate and limestone
The molten liquid is cooled, so it solidifies
Borosilicate glass is made by heating sand with boron trioxide - has a much higher melting point than soda-lime
Glass is transparent, strong and a good thermal insulator so is good for in windows
Composites are materials that consist of two or more materials with different properties, that have been combined to produce a more desirable material
Most have two components - one for reinforcement, like long solid fibres, and a matrix, which binds it together (starts soft and hardens)
Polymers are large molecules of high Mr, made by linking together lots of monomers
Properties depend on the original monomers and the conditions of the chemical reaction
Generally they are flexible, easily shared and good for electrical and thermal insulators
Poly(ethene) is a common polymer, and is has two main forms
Low-density poly(ethene) and high-density poly(ethene)
LDPE is made in moderate temperatures, high pressures and with a catalyst
It is more flexible but weaker
It is used in carrier bags, etc.
HDPE is made in low temperatures and pressures, with a catalyst
It is more rigid but stronger
It is used for drain pipes, etc.
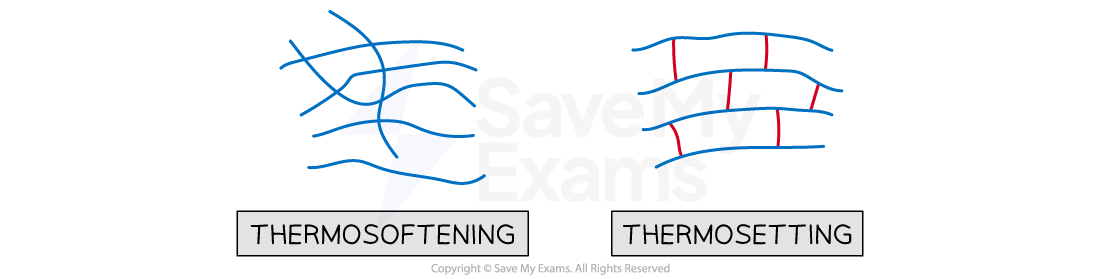
Thermosoftening polymers are made by lots of polymer chains held together by weak intermolecular forces
These weak bonds break easily when heated, causing the polymer to break so it can be remoulded and cooled so it hardens again
Thermosetting polymers are made from lots of polymer chains held together by strong covalent bonds
Lots of energy is needed to break them, so they don’t soften when heated - are hard, rigid and strong
Corrosion
Corrosion is the process by which metals are slowly broken down by reacting with substances in their environment
Iron + oxygen + water → hydrated iron oxide (rust)
Fe → Fe3+ + 3e- (oxidised)
O2 + 4e- → 2O2- (reduced)
Conditions required for rust are oxygen and water
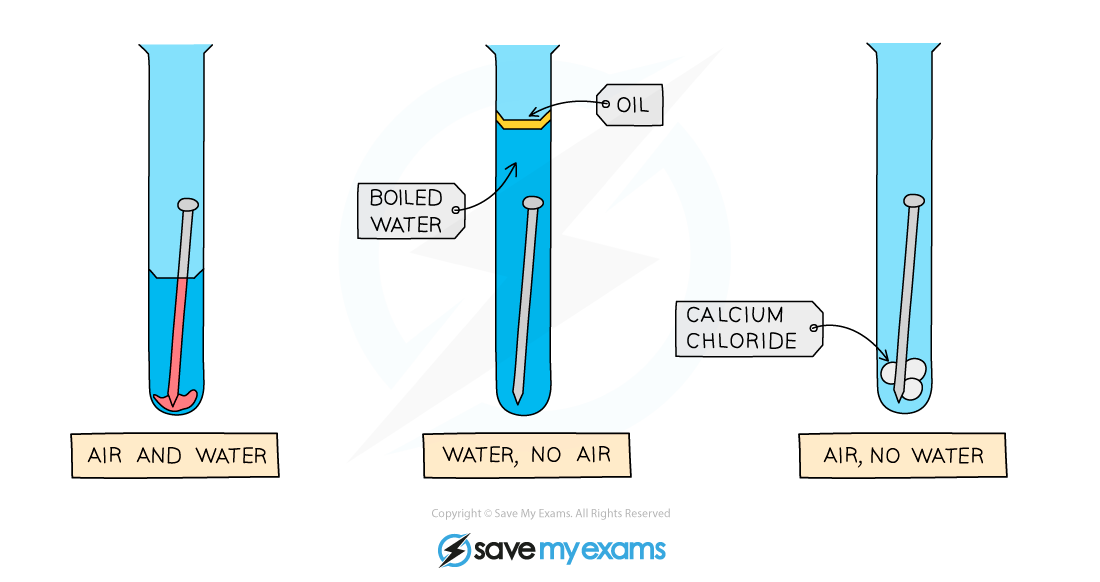
Only the surface metal corrodes
Layers can break away and completely break down with iron and rust
For aluminium, only surface atoms are affected - protected layer is formed
Prevention from rusting
Barriers - prevents oxygen and water from being in contact
Paint, oil/grease, electroplating (using electrolysis to cover the iron in a thin layer of another metal)
Sacrificial - adding a more reactive metal to the iron, like zinc or aluminium
If the object is exposed to oxygen, the more reactive metal will be oxidises instead
Galvanising - both barrier and sacrificial
A layer of zinc is added to protect, but if scratched the oxygen won’t reactive with the iron as the zinc is much more reactive
Sustainable development - recycle and reuse
Sustainable development is an approach to human and economic development that meets the needs of current generations without damaging future generations
It shouldn’t damage the environment, use up limited resources or add to global warming
We can use renewable resources
Wood instead of plastic (from crude oils) as it is more sustainable
We have to take the energy required for the manufacturing process into account, like if it uses fossil fuels
Ways to reduce the environmental impact of manufacturing:
More efficient processes - tiny amounts of oil needed
Catalysts
Optimum pressures and temperatures
After a product has been used, it is best to reuse or recycle it
Reuse - either for the same purpose or different
Recycle - if the products are too damaged, parts or elements can be separates, crushed/melted and made into new products of similar or different types
Fewer quarries and mines are needed
Less crude oil used, so land fill is reduced
Less energy is required than making new
Life cycle assessments (LCA’s)
LCA’s analyse the different stages in a products life cycle, and assess its environmental impacts
Extracting and processing raw materials
Manufacturing and packaging
Using the product
Disposal
Extracting and processing
Can directly damage local environment, through mines or deforestation
Indirectly damage the environment through huge amounts of energy being used, and pollutants being released (global warming)
Manufacturing and packaging
Energy use
Pollution - carbon monoxide or hydrogen chloride
Waste products - usually useless
Impact of using product
Damage it does during its lifetime
Pollutants from a car, fertiliser run-off
Length it is used for
Re-useable vs single use
Disposal
Land fill - takes up space, chemical damage
Burn waste - releases pollutants to atmosphere
Energy required for transport to these places
Limitations of LCA’s
Difficult to quantify exact amounts
Difficult to assess harm of each step and their comparisons
Can be manipulated to support a campaign
Potable water
Potable water is water that is safe to drink
Not necessarily pure (only H2O)
Criteria required for water to be potable
Levels of dissolved substances need to be fairly low
pH has to be between 6.5 and 8.5
There can’t be any microorganisms (bacteria or fungi)
Fresh water - only has a small amount of dissolved substances
For example, rain
Surface water - lakes, rivers, reservoirs (exposed at surface)
Ground water - aquifers (permeable rock that traps water in ground)
Surface water is easy to access and is frequently replaced, however it can dry up if hot, so ground water is relied on
Fresh water needs to be treated
Filter through a wire mess (removes large bits)
Pass through a bed of sand and gravel - filters small bits out
Sterilise it - bubble chlorine gas through, UV light or exposure to ozone
Some countries have little rain, so rely on desalination (extraction from sea water)
Desalination is done through distillation or reverse osmosis
Both are expensive as they require lots of energy - not practical in large amounts
Distillation - boil and collect water vapour to get pure distilled water
Reverse osmosis - salt water is passed through a membrane, which removes salt and ions to get pure distilled water
Waste water treatment
Sources of waste water:
Domestic - household waste (sinks, toilets, showers)
goes to sewers and then sewage treatment plants
Agricultural systems - nutrient run-off and animal waste
Industrial - from factories with chemicals
All waste water has to be treated
Domestic and agricultural - organic matter and harmful microbes have to be removed, as they could pollute fresh water (health risk)
Industrial - contain chemicals, so needs extra treatment
Sewer treatment
Screening - removing large objects
Sedimentation - the sewage is left in a settlement tank so that solids settle to form sludge and effluent
Effluent is separated and biological breakdown by microorganisms occurs to remove organic matter
Sludge occurs in anaerobic conditions - sealed so anaerobic digestion happens
Methane is produced, which can be trapped and burned as fuel
Waste can be used as a fertiliser
Effluent occurs in aerobic conditions
Air is pumped in - oxygen is supplied
Breaks down by aerobic digestion
Water is now safe to be released back into the environment
For toxic substances, additional stages have to occur
Adding chemicals to precipitate out metals
UV radiation
Sewage treatment is easier than desalination, though fresh water is best
The Haber process
The Haber process is the industrial production of ammonia from nitrogen and hydrogen
N2 + 3H2 ⇌ 2NH3 (and heat)
An iron catalyst, 450c and 200 atm
The ammonia produced is used for nitrogen based fertilisers, to grow all food needed
Nitrogen is easy to acquire - from air
Hydrogen is harder as it has to be hade from hydrocarbons (like methane)
It is an exothermic reaction, and is a reversible reaction
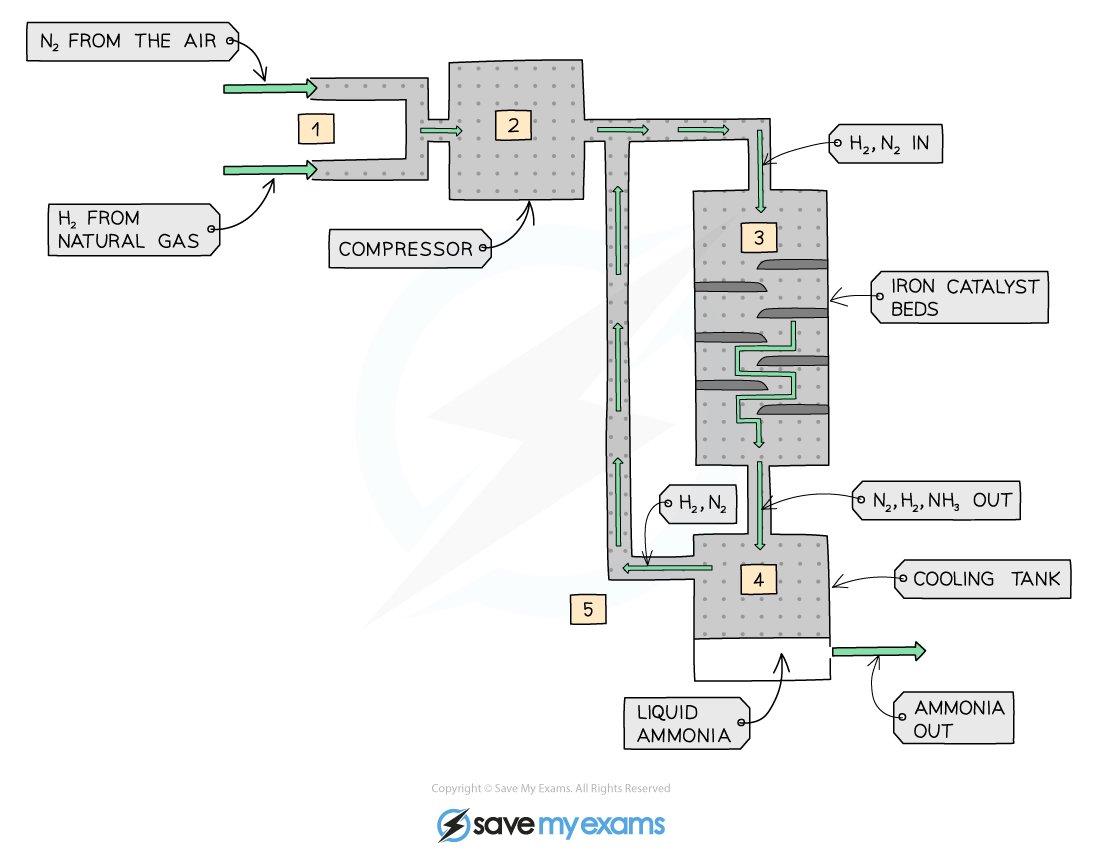
Nitrogen and hydrogen enter and are compressed
They pass through the reaction vessel (450 degrees and 200 atm), where they react together to form ammonia
Not all hydrogen and nitrogen reacts - reversible reaction
The spare nitrogen and hydrogen and produced ammonia enter the cooling tank, where ammonia condenses as it has a lower boiling point
The nitrogen and hydrogen cycles back around, and the process repeats
The process produces liquid ammonia 🙂
Why 450 degrees?
As it is an exothermic reaction, we need a low temperature so the forward reaction is favoured
For a higher % yield
For a higher rate of reaction, we need a high temperature - energy for more frequent particle collisions (reach activation energy easier)
450 degrees is used as a compromise - lower yield, but higher rate of reaction
Any higher would be too costly
Why 200 atm?
For a high % yield, we want a high pressure so the reaction favours the right
Less molecules on right side
High pressure also means high rate of reaction
High pressure is expensive, and can be dangerous if it goes wrong
200 atmospheres balances it out
NPK fertilisers
A fertiliser is a substance that is applied to soil in order to supply plants with nutrients
In the past, fertilisers were usually made from animal waste (cow manure) but now they are usually formulated fertilisers (made in factories)
A formulation is a mixture that has been designed as a useful product
If we make fertilisers by combining chemicals in a specific ration it would be a formulated fertiliser
Formulated fertilisers contain nitrogen, phosphorus and potassium
NPK - main elements plants need from the soil
NPK fertilisers are formulations of various salts containing appropriate ratios of the elements
Nitrogen fertilisers
Important for plants to make amino acids and hence proteins (essential for growth)
Main source is ammonia, made in the Haber process
Ammonia can make nitric acid (O2 + H2O + NH3 react) and ammonia salts (reacts with acids)
Phosphorus fertilisers
Can be mined as phosphate rock
Phosphate salts in the rock are insoluble, so can’t be used
We react the rock with acids to produce a soluble salt
Nitric acid and rock produces phosphoric acid and calcium nitrate
Sulfuric acid reaction produces calcium sulfate and calcium phosphate (singe superphosphate)
Phosphoric acid and rock produces calcium phosphate (triple superphosphate)
Potassium fertiliser
Potassium chloride and sulfate can both be mined from the ground and used directly in fertilisers
DONE!!!