alkanes
These compounds are described as being saturated hydrocarbons because they only contain single covalent bonds. Any substance with double or triple Carbon-Carbon bonds are described as being unsaturated. Most of the hydrocarbons in crude oil are saturated hydrocarbons called alkanes.
Series of alkanes
Each member of the alkane homologous series shares:
the same general formula:
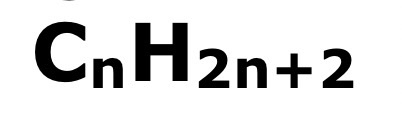
The same functional group (atom and groups responsible from most of the chemical reactions of a compound)
They have similar chemical properties
Naming alkanes

1) the first alkane has 1 carbon and 4 hydrogens, methane
2) the second alkane has 2 carbon and 6 hydrogens, ethane
3) the third alkane has 3 carbon and 8 hydrogens, propane
4) the fourth alkane has 4 carbons and 10 hydrogens, butane
Boiling points
Due to the weak intermolecular forces between molecules of alkanes they have low boiling points, however these do increase as the molecules get bigger.