kinetic theory of laws
A gas consists of tiny particles, either atoms or molecules, moving about at random.
The volume of the particles themselves is negligible compared with the total volume of the gas; most of the volume of a gas is empty space.
The gas particles act independently of one another; there are no attractive or repulsive forces between particles.
Gas particles can move wherever they want. There are NO attractive or repulsive forces between the particles. Every direction of movement is equally probable.
The average Kinetic Energy of the gas particles is proportional to the Temperature (expressed in units of K) of the sample.
Kinetic energy (KE) refers to the energy associated with movement or motion. It is determined by the mass (m) and velocity or speed (v) of a substance, according to the relationship:
KE=\frac12mv^2
v - velocity
m - mass
2. Ideal gas law
PV=nRT
P is pressure (measured in atm)
V is volume (Liters)
n is moles of gas present
R is a constant that converts the units. It’s value is 0.08221 atm.L/mol.K
T is temperature measured in Kelvin
Simple algebra can be used to solve for any of these values
p=\frac{nRT}{V}
V=\frac{nRT}{P}
n=\frac{PV}{RT}
\top=\frac{PV}{nR}
R=\frac{PV}{nT}
Relationship between V, T AND P
Relationship between volumen and pressure
Constant:
n (mol)
R (ideal gas constant)
T (temperature)
P\propto\frac{1}{V}
The one we need to know:
p1 x V1 = p2 x V
Boyle's law
All the particles (atoms and molecules) of a substance are continually moving and so possess kinetic energy. In gases the movement of the particles is highly energetic and this is the reason why gases form, the particles have enough energy to overcome the attractive forces holding the particles together. In gases the particles are moving very quickly and freely in a random manner constantly bumping into each other and their surroundings. It is these collisions between the particles of the gas and the walls of the container it is confined to that creates gas pressure. The gas pressure is the overall force of all these collisions divided by the area of the walls of the container it is confined in.
The relationship of a gas with pressure and volume was developed by the scientist Robert Boyle at around 1660 and is known as Boyle’s Law.
Boyle’s law states:
"For a fixed mass of gas, at a constant temperature, the product (pressure x volume) is a constant."
Pressure x Volume = constant
p x V = constant
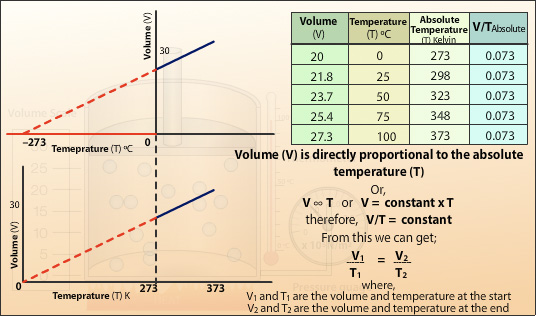
Relationship between volume and temperature
V\varpropto T
This is a constant
\frac{V}{T}
Relationship between pressure and temperature
Constant:
n (mol)
R (ideal gas constant)
V (volume)
The pressure law states:
"For a fixed mass of gas, at a constant volume, the pressure (p) is directly proportional to the absolute temperature (T)."
Pressure ∝ Temperature
Pressure | = constant |
Temperature |
Summary
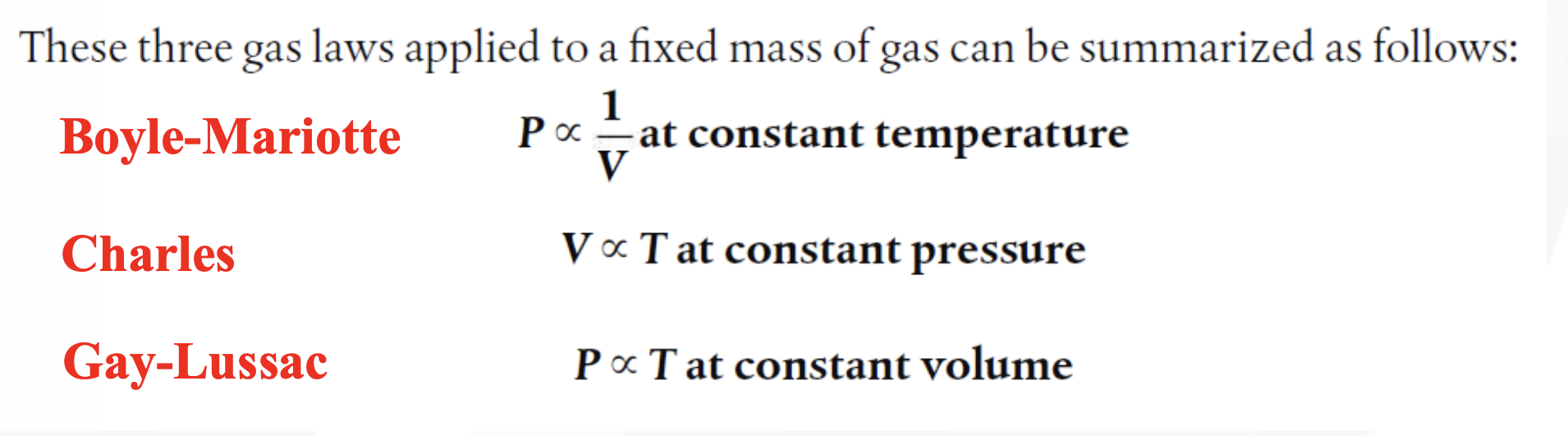
Combined gas law
hese can be combined to give one equation for a fixed mass of gas: PV T = a constant or PV1-P₂V2 (where I and 2 refer to initial and final conditions respectively) T T2 Application of this enables gas volume, pressure, and temperature to be calculated as conditions change
Exercices
1⃣ Charles’s Law (Pressure constant)
[\frac{V_1}{T_1} = \frac{V_2}{T_2}]
Given:
(V_1 = 2.5 , L), (T_1 = 300,K), (T_2 = 350,K)
[
V_2 = V_1 \times \frac{T_2}{T_1} = 2.5 \times \frac{350}{300} = 2.5 \times 1.1667 = 2.9167 , L
]
✅ Answer: (V_2 = 2.92 , L)
2⃣ Gay-Lussac’s Law (Volume constant)
[
\frac{P_1}{T_1} = \frac{P_2}{T_2}
]
Given:
(P_1 = 1.2 , atm), (T_1 = 250,K), (T_2 = 300,K)
[
P_2 = P_1 \times \frac{T_2}{T_1} = 1.2 \times \frac{300}{250} = 1.2 \times 1.2 = 1.44 , atm
]
✅ Answer: (P_2 = 1.44 , atm)
3⃣ Boyle’s Law (Temperature constant)
[
P_1 V_1 = P_2 V_2
]
Given:
(P_1 = 2.0 , atm), (V_1 = 3.0 , L), (P_2 = 3.0 , atm)
[
V_2 = \frac{P_1 V_1}{P_2} = \frac{2.0 \times 3.0}{3.0} = \frac{6.0}{3.0} = 2.0 , L
]
✅ Answer: (V_2 = 2.0 , L)
4⃣ Combined Gas Law
[
\frac{P_1 V_1}{T_1} = \frac{P_2 V_2}{T_2}
]
Given:
(P_1 = 1.0 , atm), (V_1 = 1.5 , L), (T_1 = 273,K),
(P_2 = 2.0 , atm), (T_2 = 300,K)
[
V_2 = \frac{P_1 V_1 T_2}{P_2 T_1} = \frac{1.0 \times 1.5 \times 300}{2.0 \times 273} = \frac{450}{546} = 0.824 , L
]
✅ Correct Answer: (V_2 \approx 0.82 , L)