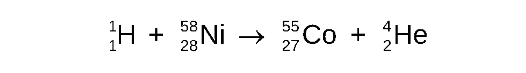Chapter 5.2 Nuclear Reactions
Nuclear Reactions
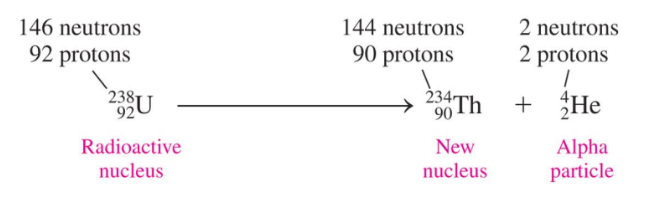
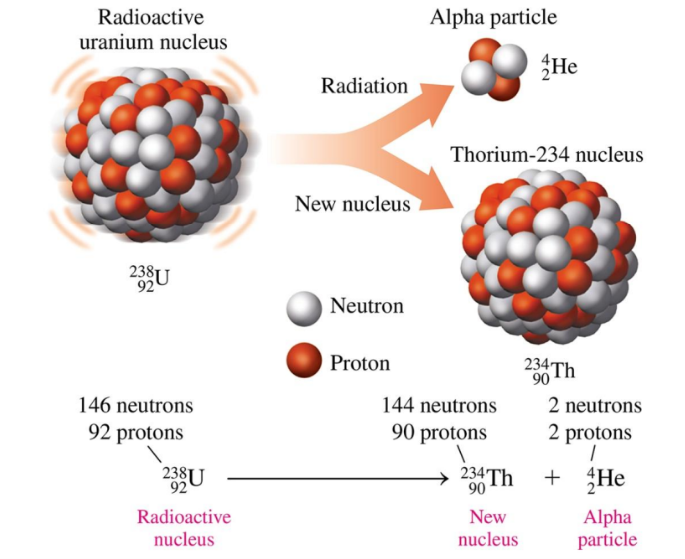
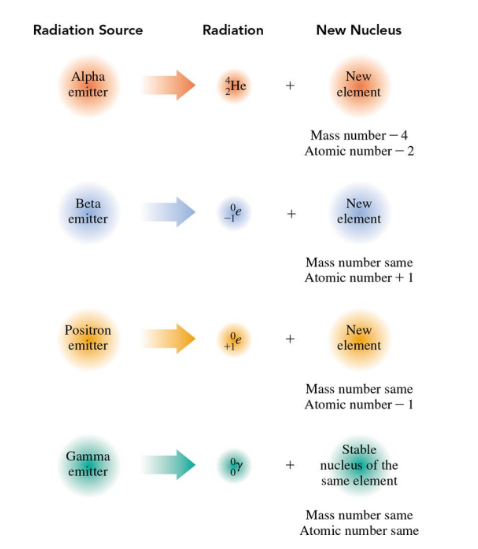
In nuclear equations for alpha decay, the mass number of the new nucleus decreases by 4 and its atomic number decreases by 2
Balancing Nuclear Equations
In a process called radioactive decay, the nucleus breaks down by emitting radiation. The process is represented by nuclear equation in which
the mass number and atomic number may change
the sum of the mass numbers and the sum of the atomic numbers are equal for the reactants and the products
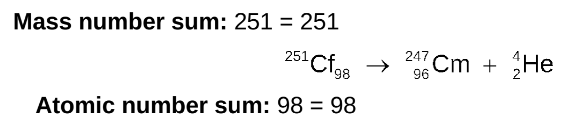
Alpha Decay
Alpha decay occurs when a radioisotope nucleus emits an alpha particle, forming a new nucleus with a mass number that is decreased by 4 and an atomic number that is decreased by 2
Equation for Alpha Emissions Super 4, sub 2, He
(americum-241)
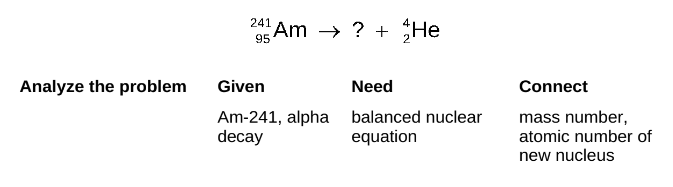
STEP 1
Write the incomplete nuclear equation
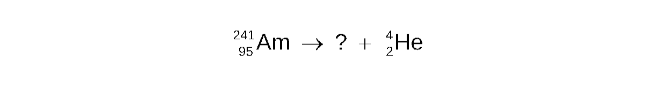
STEP 2
Determine the missing mass number
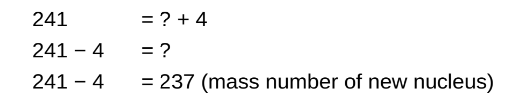
STEP 3
Determine the missing atomic number

STEP 4
Determine the symbol of the new nucleus and complete the nuclear equation
The elements that has atomic number 93 is neptunium, Np
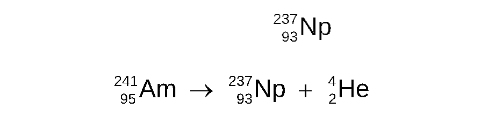
Beta Decay Super 0, Sub Negative 1, e
In the nuclear equation for beta decay, the mass number of the new nucleus remains the same and its atomic number increases by 1
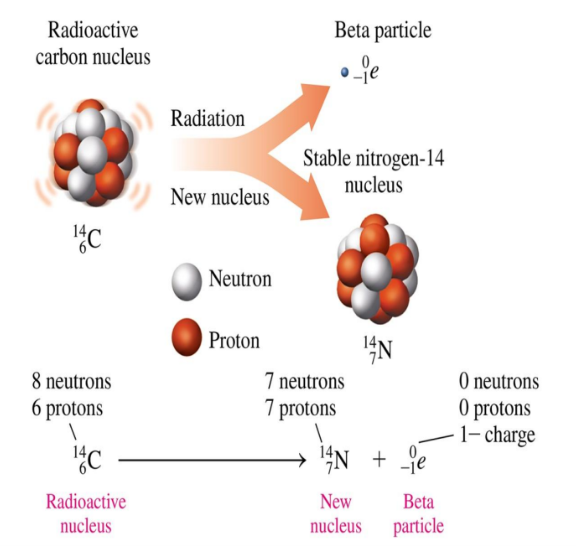
Equation for Beta Decay Super 0, Sub Negative 1, e
(potassium (K)-42)

STEP 1
Write the incomplete nuclear equation for the decay of K, a beta emitter
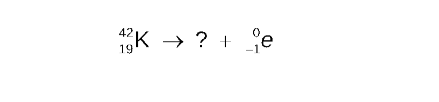
STEP 2
Determine the missing mass number
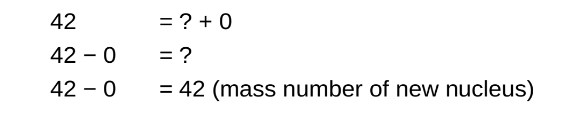
STEP 3
Determine the missing atomic number

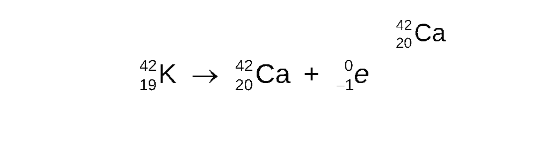
STEP 4
Determine the symbol of the new nucleus and complete the nuclear equation
The element that has atomic number 20 is calcium (Ca)
In summary, when the nuclei of alpha, beta, positrons and gamma emitters emit radiation, new and more stable nuclei are formed
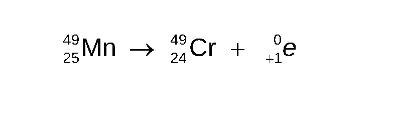
Positron Emission Super 0, Sub Plus 1, e
In positron emission, a proton is converted to a neutron and a positron
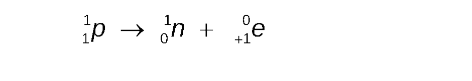
The mass number remains the same, but the atomic number decreases by 1.
Producing Radioactive Isotopes
Radioactive isotopes are produced
when a stable nucleus is converted to a radioactive nucleus by bombarding it was a small particle
a process called transmutation
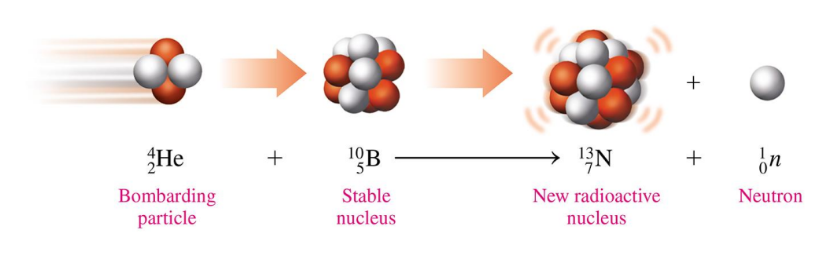
Equation for Producing New Isotopes by Bombardment
Write the balanced nuclear equation for the bombardment of nickel-58 by a proton which produces a radioactive isotope and an alpha particle

STEP 1
Write the incomplete nuclear equation
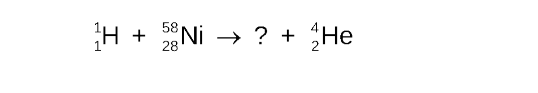
STEP 2
Determine the missing mass number

STEP 3
Determine the missing atomic number

STEP 4
Determine the symbol of the new nucleus
the elements that has atomic number 27 is cobalt (Co)
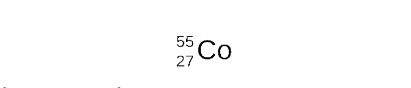
STEP 5
compete the nuclear equation