Organic chemistry
Hydrocarbons
Organic chemistry is all about compounds that contain carbon
Hydrocarbons are any compounds that are formed from carbon and hydrogen only
Alkanes are saturated hydrocarbons - every carbon has 4 single covalent bonds so no double bonds
They are a homologous series - have similar properties
Their general formula is CnH2n+2
Methane - CH4
Ethane - C2H6
Propane - C3H8
Butane - C4H10
Alkanes
An alkane is a hydrocarbon with no double bonds
As the length of the carbon chain increases
Boiling points increase - 1-4 carbons = gas at room temp
5-17 carbons = liquid
18+ carbons = solid
They become less flammable (more bonds to break)
Short alkanes are volatile, so evaporate easily
Longer alkanes are more viscous (thick and runny)
Combustion reactions
The main use of hydrocarbons are as fuel, as they release lots of energy when burned with oxygen
If there is enough oxygen available, complete combustion will occur
Hydrocarbon + oxygen → carbon dioxide + water
Releases energy - exothermic
Fractional distillation
Crude oil is a fossil fuel, and is a mixture of many different compounds (mainly hydrocarbons)
It is formed naturally from the remains of dead plants and animals (plankton) over millions of years
At high pressures and temperatures underground, the natural biomass turned into crude oil, which soaked into rock and was stored until we began to drill it out
Finite resource - non-renewable
Fractional distillation separates out the hydrocarbon in crude oil, as they each have different properties, particularly boiling points
Long chains have higher boiling points, so condense at hotter temperatures (lower levels of the column)

Crude oil is heated and turned into a gas, and then passed through the fractionating column
Boiling points of shorter chains alkanes are lower, so they condense at higher layers
These are used as fuels - mainly for vehicles (petrol, kerosene, diesel)
Some hydrocarbons never condense, like liquid petroleum gas (used as a fuel)
Longer chains condense at much higher temperatures so condense lower
These are used for heavy fuel oils - heating, fuel, lubricating oils
Bitumen (longest) is for paving, etc.
Shorter chains are more flammable, so better fuels
Longer chains (HFO and bitumen) are poor fuels so can be used for other purposes or can be broken down into smaller hydrocarbons
This can be done via cracking
Substances from crude oil are called petrochemicals
They can all be used as feedstock
Lubricants, solvents, polymers and detergents
Cracking and alkenes
Longer hydrocarbons are thick viscous liquids so are less useful than shorter chains
They can broken down into shorter, more flammable hydrocarbons through cracking
Cracking is a thermal decomposition reaction
Catalytic cracking is where long chain hydrocarbons are vaporised
Hot powered aluminium oxide is then used as the catalyst
The hydrocarbons are passed over the powder, and when in contact with the catalysts they break apart
Steam cracking is where hydrocarbons are vaporised
They are then mixed with steam and heated to very high temperatures
This causes the chains to split apart into smaller hydrocarbons
Cracking balanced equations - long chain alkanes break down into a shorter alkAne and an alkEne
For example, heptane → butane + propene
When breaking down, there aren’t enough hydrogens for both hydrocarbons to be saturated (only single bonds)
Instead one product has a double bond - alkene
These can be used to make polymers and as starting materials for other polymers
Alkenes are unsaturated hydrocarbons
They are a homologous series
They have at least one double bond (C=C)
They are more reactive than alkanes - can react with bromine
Alkenes + bromine water (orange) → colourless solution
They can be added together to make molecules as the double bond breaks open to form two more bonds
This is how we can test for alkenes compares to alkanes that don’t react
Reactions of alkenes
Alkenes are unsaturated hydrocarbons
They have at least on double carbon bond
Functional group is C=C
As they have a double bond, the can undergo addition reactions
The double bond opens up and other molecules or atoms can be added
Hydrogen + alkene → alkane
Using a catalyst (for example, nickel)

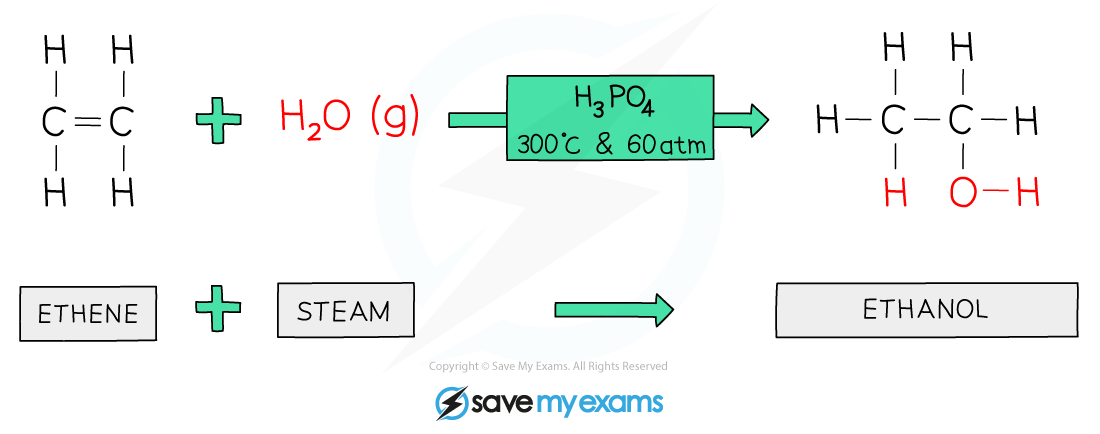
Water + alkene → alcohol
Using a catalyst, high temperatures and pressures
H2O + CH2CH2 → C2H5OH (ethene to ethanol)
Alcohol formed with OH functional group
The ethanol produced, unreacted ethene and water has to be separated out
We can cool it so that ethanol and water condense and ethene stays a gas
Water and ethanol can then be separated through fractional distillation
Ethanol has a lower boiling point so will evaporate
Halogens + alkene → _____
No catalyst required
Br2 + C2H4 → C2H4Br2

Alkenes can be tested for using bromine, which turns from orange to colourless when mixed, as all bromine bonds to alkene where double bonds have opened
Alkanes are saturated so don’t take part in addition reactions
Addition polymers
An alkene has a double carbon bond
They are unsaturated and can break open so carbons form new bonds
Monomers open up to form chains
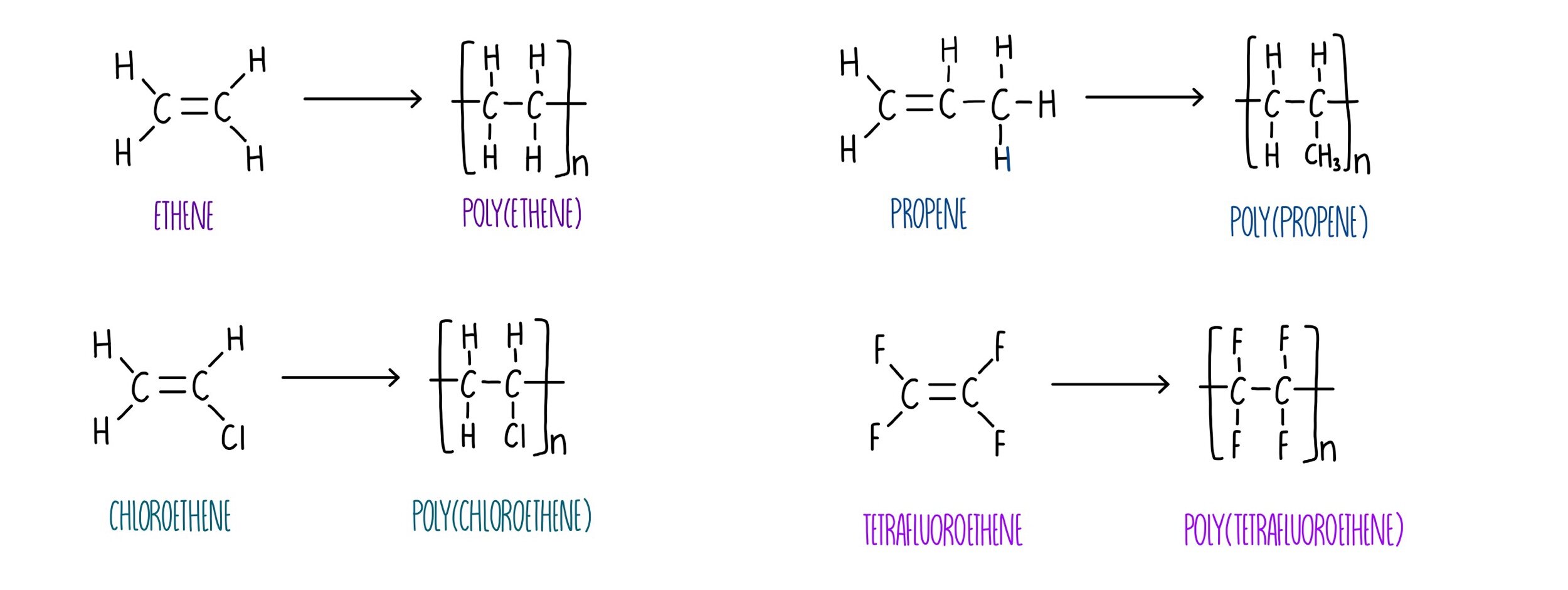
With high pressures and catalysts, lots of monomers can split open and form polymer chains
Poly(ethene), poly(butene)…
Alcohols
Alcohols are a homologous series
They have the functional group OH
Methanol, ethanol, propanol, butanol
General formula is CnH2n+1OH
Properties of alcohols
Shorter chain alcohols are more flammable
Can undergo complete combustion - alcohol + oxygen → carbon dioxide + water
Shorter chains are more soluble - can dissolve in water
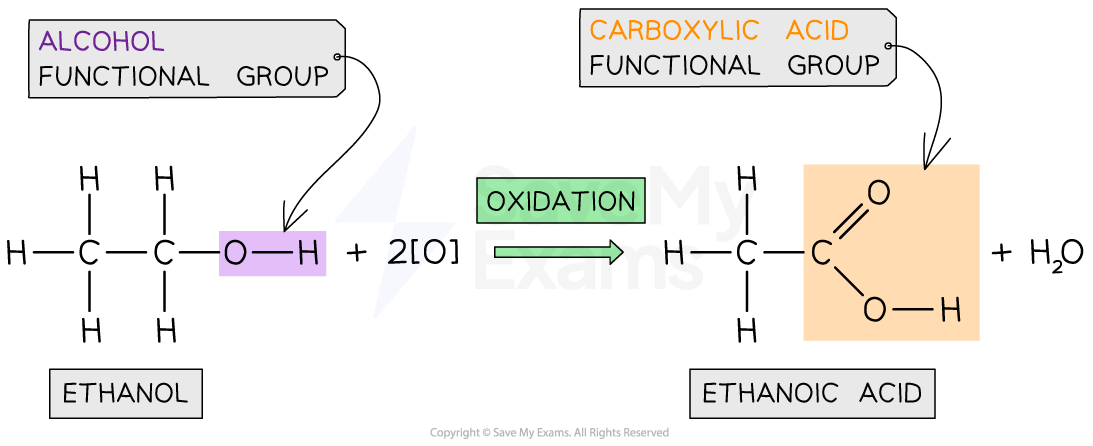
When oxidised, they form carboxylic acids with the functional group COOH
Uses of alcohols
Flammable - as fuels
Solvents in industry
Can dissolve what water can’t, like hydrocarbons and lipid compounds (fats and oils)
Production of ethanol
Ethanol is an alcohol with the formula CH3CH2OH
It is used as a chemical feedstock to produce other organic compounds, as a biofuel and in alcoholic drinks
Ethene + steam → ethanol
C2H4 + H2O → C2H5OH

It is an addition reaction
High temperatures (300 degrees) and pressures (60-70 atm) are required
Catalyst needed - phosphoric acid
Pros - ethene is cheap
Reaction is efficient and cheap
Cons - ethene is made from a crude oil, a non-renewable resource so if it begins to run out, it will become expensive
Fermentation uses glucose to produce ethanol
Glucose → ethanol + carbon dioxide
It is the anaerobic respiration of sugars by yeast cells
It is carried out in fermentation tanks, and requires yeast cells (naturally occurring enzymes - catalyst)
Temperatures of 30-40 degrees, which are optimum for enzymes
Must be anaerobic conditions to prevent ethanol being oxidised into ethanoic acid
Pros - sugar/glucose is renewable and yeast is easy to grow
Cons - it is a slow process and ethanol produced isn’t pure (needs to be distilled by fractional distillation)
Carboxylic acids
Carboxylic acids are a homologous series with the functional group -COOH
Their names end in -anoic acid
Methanoic acid - HCOOH
Ethanoic acid - CH3COOH
Propanoic acid - C2H5COOH
Butanoic acid - C3H7COOH
They are all weak acids (don’t fully ionise - don’t release all H+ ions)
In reversible reactions
Carboxylic acid + metal carbonate → salt + water + carbon dioxide
They are made by oxidising alcohols
Alcohol →(oxidising agent) carboxylic acid
Esters
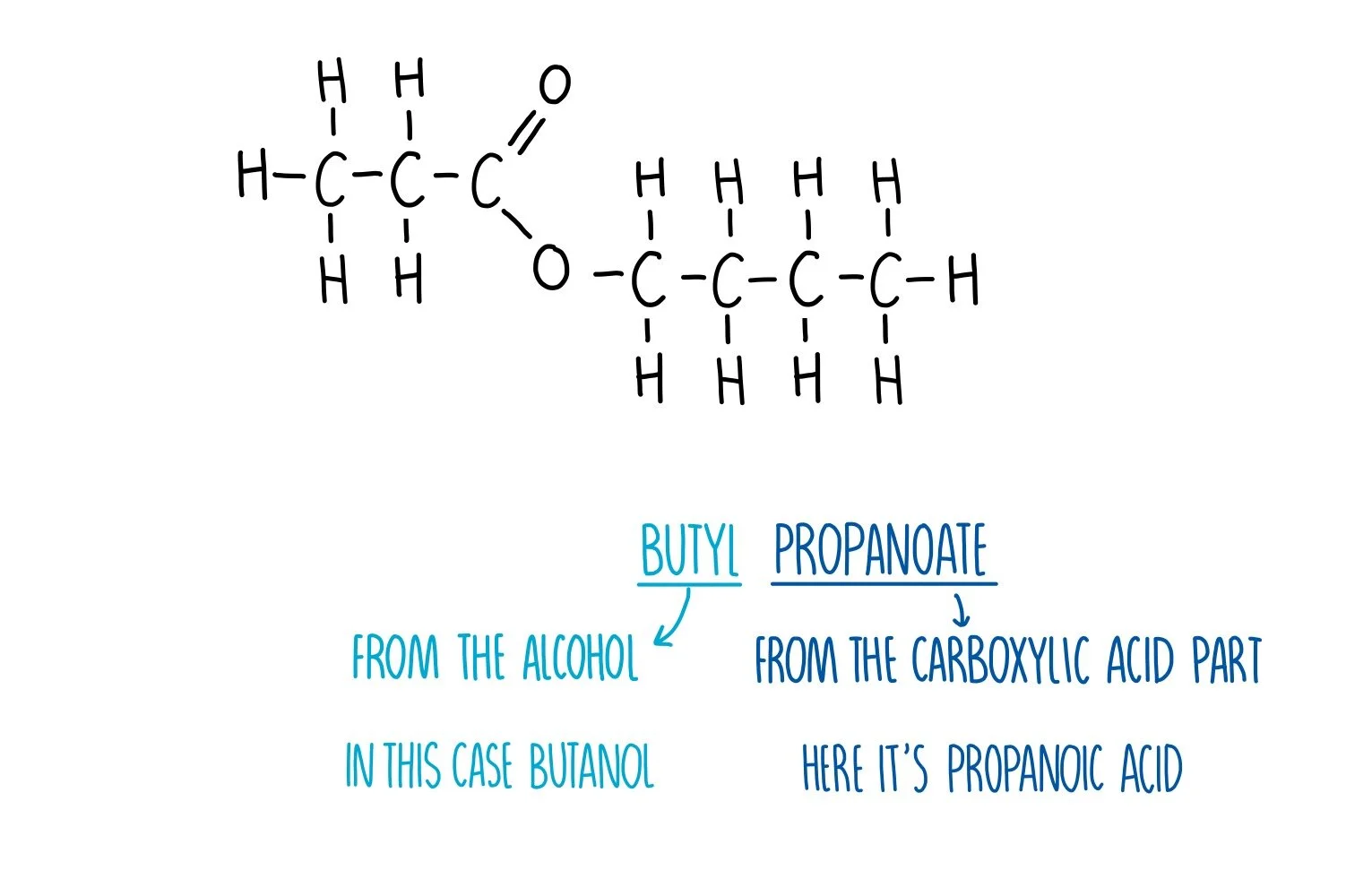
Esters have a functional group of -COO-, in the middle of the molecule
They often have pleasant smells - used in perfumes or food flavourings
They are volatile - evaporate easily
Making esters
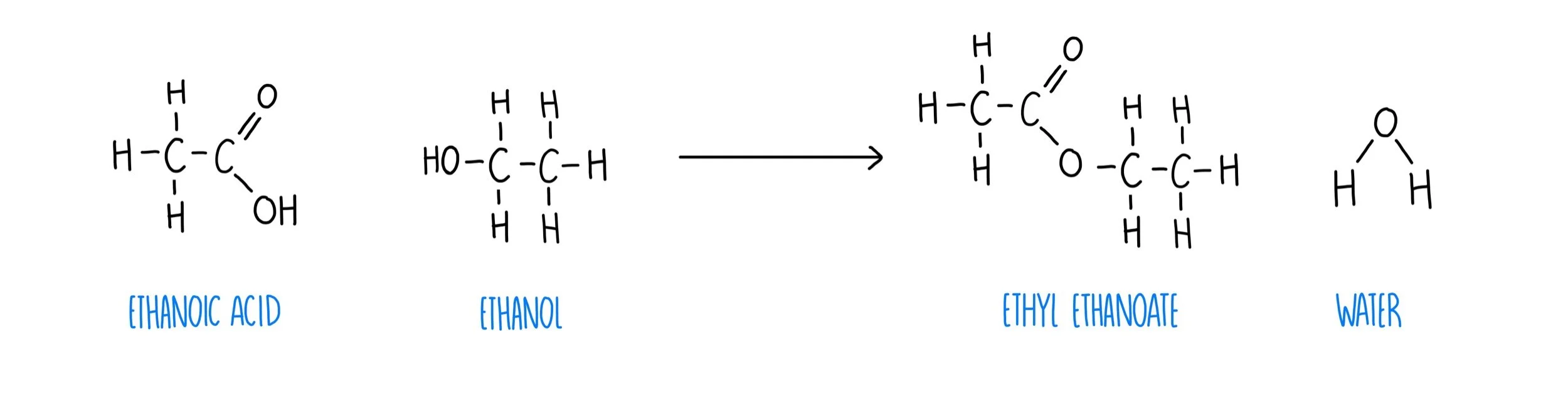
Carboxylic acid + alcohol → ester + water
For example, CH3COOH + C2H5OH → CH3COOC2H5 + H2O
Ethanoic acid + ethanol → ethyl ethanoate + water
This happens with an acid catalysts, usually sulfuric acid (H2SO4)
Ethanoic acid loses its OH, and the ethanol loses its H, which forms a water molecule
Condensation polymers
Polyesters are a type of condensation polymer and are made using an ester link (-COO-)
All polymers are made with lots of individual monomers
Polyesters are often two different monomers
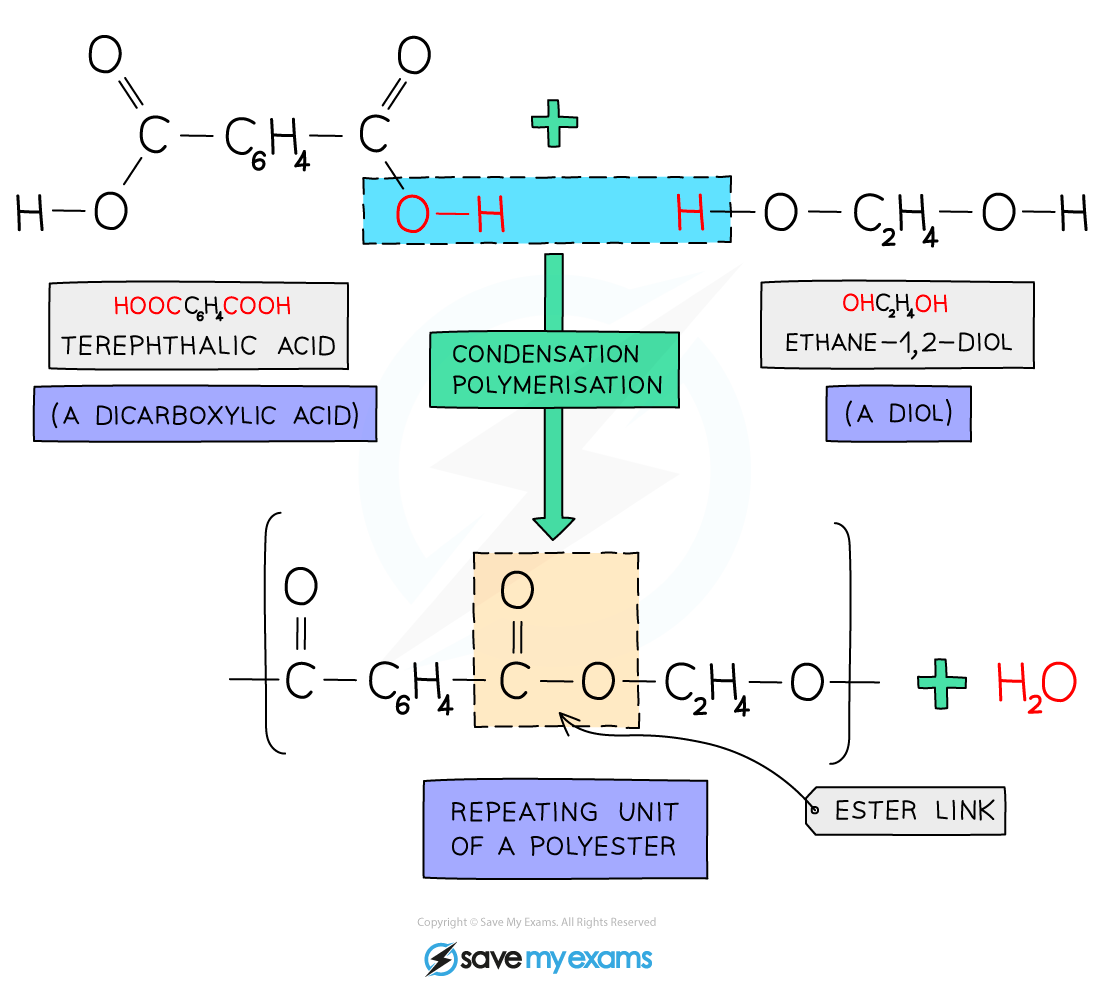
The O-H group is lost from the dicarboxylic acid, and the H is lost from the diol, which forms a water molecule
The carbon then bonds to the O (single bond), which forms an ester link (-COO-)
This forms a repeating unit of a polyester and water
Polyesters are made from a dicarboxylic acid with two -COOH functional groups, and a diol monomer with two -OH functional groups
Condensation polymers are only formed when:
Each of the monomers has at least two functional groups
There are at least two different functional groups overall
A small molecule is given up in the reaction - normally water
Polyesters are generally biodegradable, as bacteria can break down ester links
Different to addition polymers which are plastics
Naturally occurring polymers
Naturally occurring polymers include polypeptides (lots of amino acids), DNA (nucleotides) and carbohydrates (lots of sugars)
Polypeptides
Long chains of amino acids
If they fold up, or combine with other polypeptides, they form proteins
They can combine in many different combinations
They can catalyse chemical reactions as enzymes and provide structure and strength to tissues
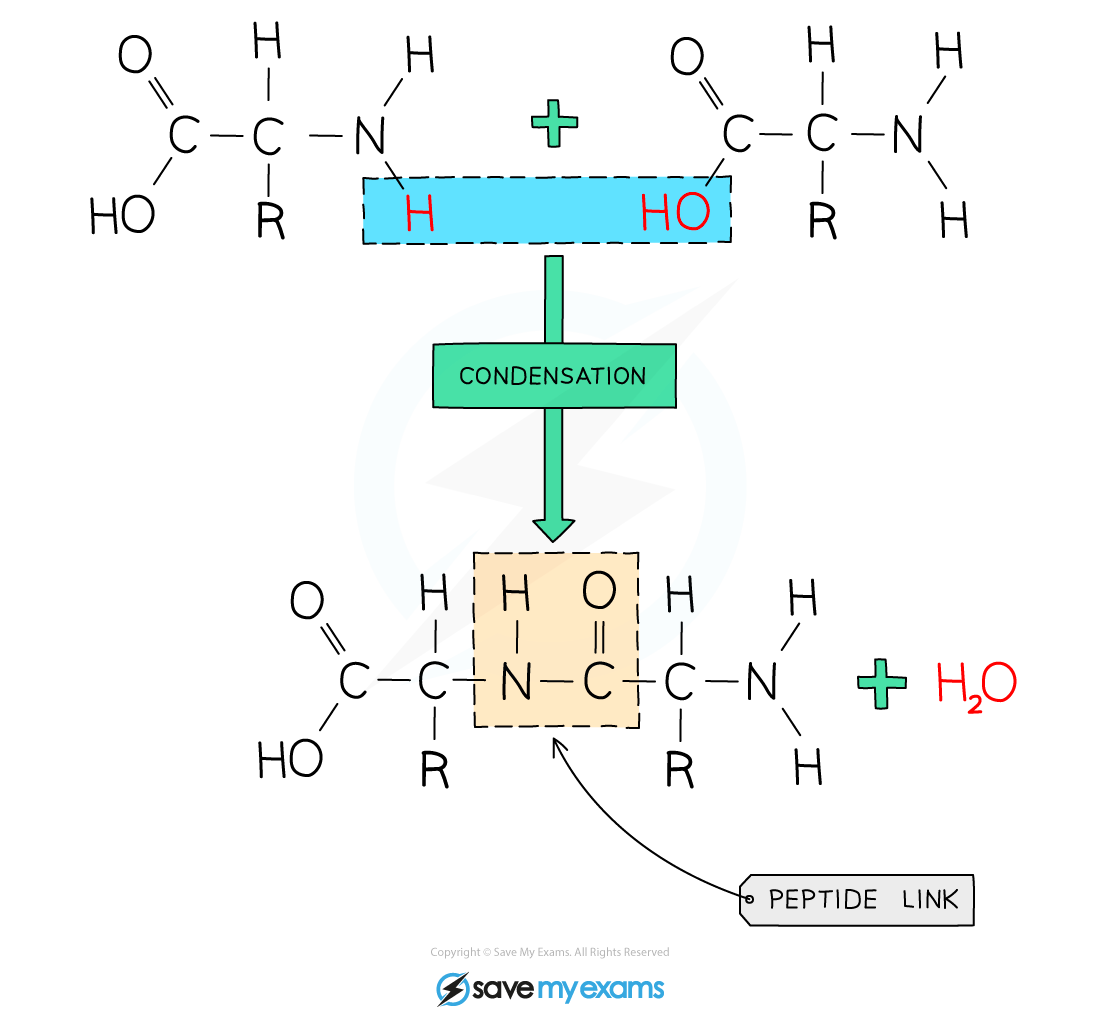
The R group changes depending on the amino acid wanted
The NH2 is the amino group, and the COOH is the carboxylic group
When they form polymers, they lose an -OH and H to join together (condensation polymerisation)
They have a peptide link (amide bond/link) - NHCO
They also form water molecules from the lost H and OH
DNA
Monomers of nucleotides - all contain a base (4 types - ATGC)
Froom the different bases they form different genes from their order
They are all held together and prevented being damaged by two polymer chains linked together - naturally coils into a double helix
The backbone of DNA is made from phosphates and sugars
Carbohydrates
Refer to a number of different polymers and monomers that we derive energy from
They are all made of only carbon, oxygen and hydrogen
Polymers can be starch, cellulose and glycogen
Monomers can be glucose and fructose (sugars) which combine to make polymers
DONE!!!