Regents Chemistry: Atomic Theory
(FIRST TOPIC: ATOMIC HISTORY)
Dalton's Atomic Theory (1803):
He said atoms were hard indivisible spheres
His model is the “Billiard Ball Model” which is just a dense ball with the same material throughout.

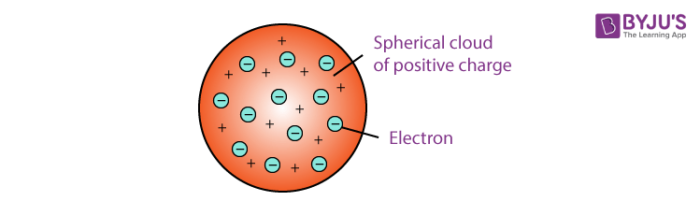
Thomson's Model (1897):
Discovered the electron through the cathode ray tube experiment
He put an electric current through a vacuum and discovered the charge was attracted towards the positive side, showing that the current had negatively charged particles, later on named electrons.
His model is the “Plum Pudding Model” which depicted a sphere with positively charged fluid and electrons as balls or the “plums”
Rutherford's Model (1911): (MOST PROMENET ON THE REGENTS!!!)
Discovered the proton through the gold foil experiment
He shot alpha particles at a gold foil and saw what happened to them. Some of them bounced off proving that atoms had positively charged dense nucleuses, However, most of them went straight through, proving that atoms were made of mostly empty space
Developed the nuclear model; (jimmy neutron atom picture, the most popular depiction of the atom. ) a nucleus made out of a positively charged nucleus with electrons circling in fixed paths
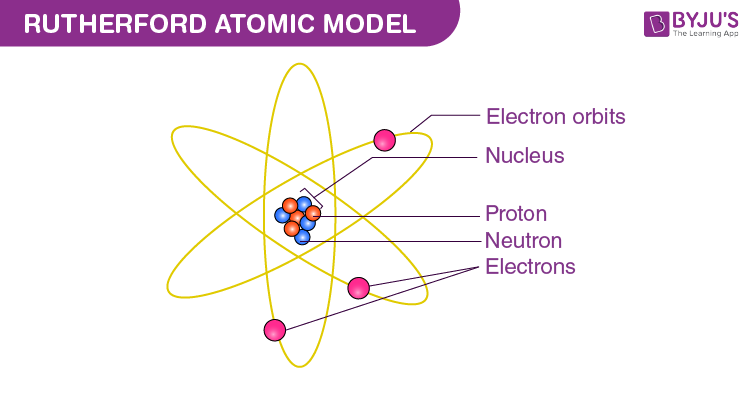
( TLDR: discovered the proton, atoms were made of mostly empty space and have a positively charged dense nucleus)
Bohr's Model (1913):
developed the planetary model, where the electrons orbited the nucleus in fixed paths like how the earth orbits around the sun in a circular pattern; different from the nuclear model where the electrons where in oval like paths
Quantum Mechanical Model (1920s):
Suggested that electrons were based on probability and not located in fixed paths. So the electrons were located in “orbitals” or in the “electron cloud” around the nucleus. And the orbital is the most probable location of finding an electron.
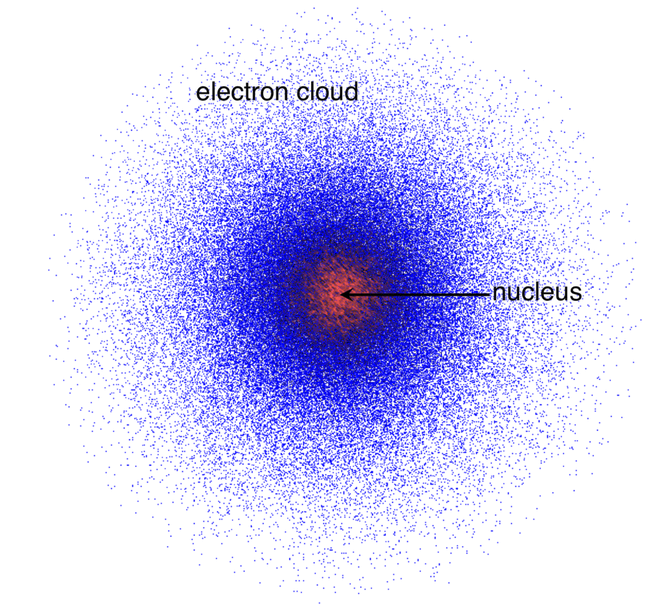
(SECOND TOPIC: SUBATOMIC PARTICLES:)
Atoms are made of three subatomic particles:
proton: positively charged particle (symbol: p+)
- Located inside the nucleus- Mass of 1 AMU
electron: negatively charged particle (symbol: e-)
- Located outside the nucleus in “orbitals” (most probable location of the electron)
- Mass of 1/1823 AMUneutron: neutral charged particle (symbol: n⁰)
- Located inside the nucleus
- Mass of 1 AMU
Atomic Mass and Atomic Number of an ATOM:
The atomic number of an ATOM is the # of protons (which identify the element; if the number of proton changes, the element changes) and it symbolizes the # of electrons too because an atom is neutrally charged.
The atomic mass of an atom is the # of neutrons AND # of protons in an atom.
Nuclear Charge: # of protons because the nucleus contains the # of protons and neutrons in an atom
Calculating # of neutrons, protons and electrons in an ATOM:
To get the # of protons and electrons, look at the atomic number
To get the # of neutrons, subtract the atomic number from the atomic mass
(Atomic mass - Atomic number = # of neutrons)
Nuclear and Hyphen notation:
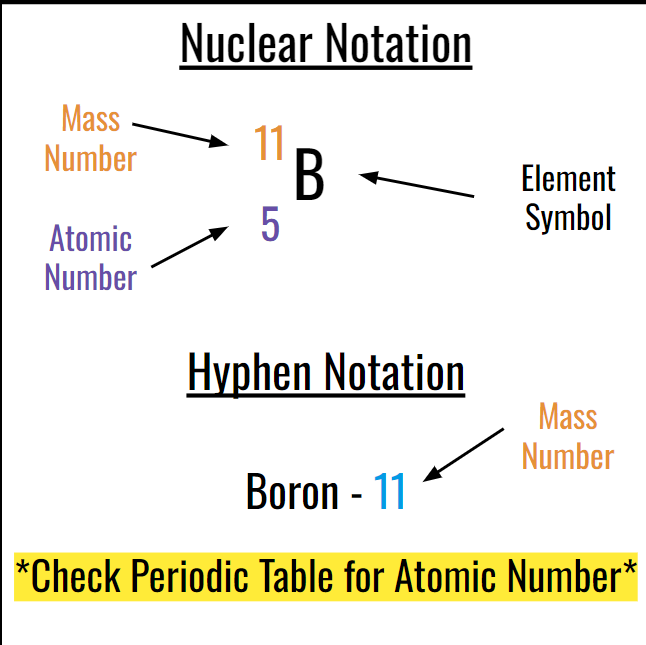
THIRD TOPIC: ISOTOPES:
What are isotopes?
- an atom of the same element that has the same # of protons but different # of neutrons.
Isotopes have the same atomic number, but different mass number.
MUST KNOW ISOTOPES:
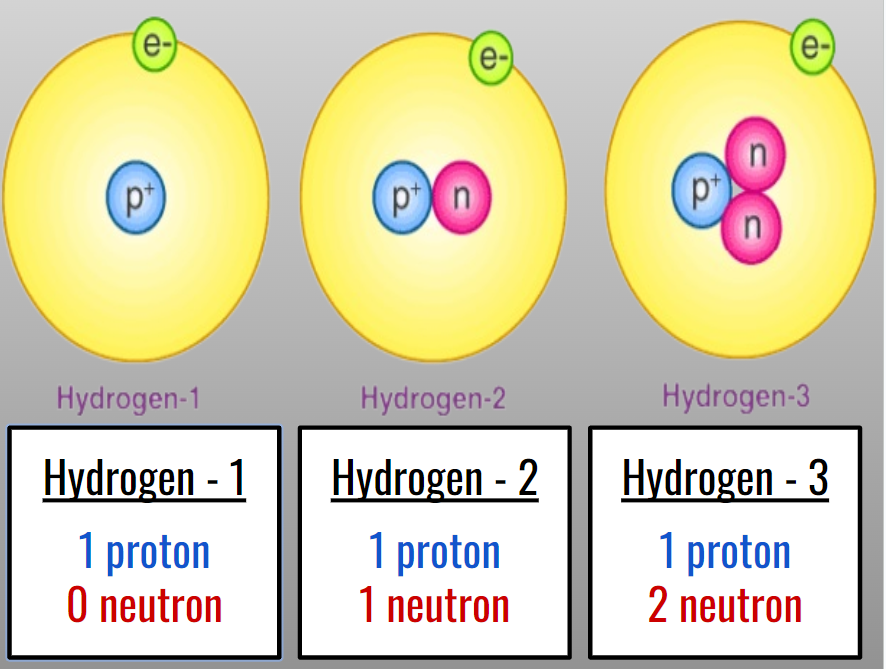
Hydrogen’s isotopes:
Hydrogen-1: (1 proton and 0 neutron)
Hydrogen-2 (Deuterium): (1 proton and 1 neutron)
Hydrogen-3 (Tritium): (1 proton and 2 neutrons)
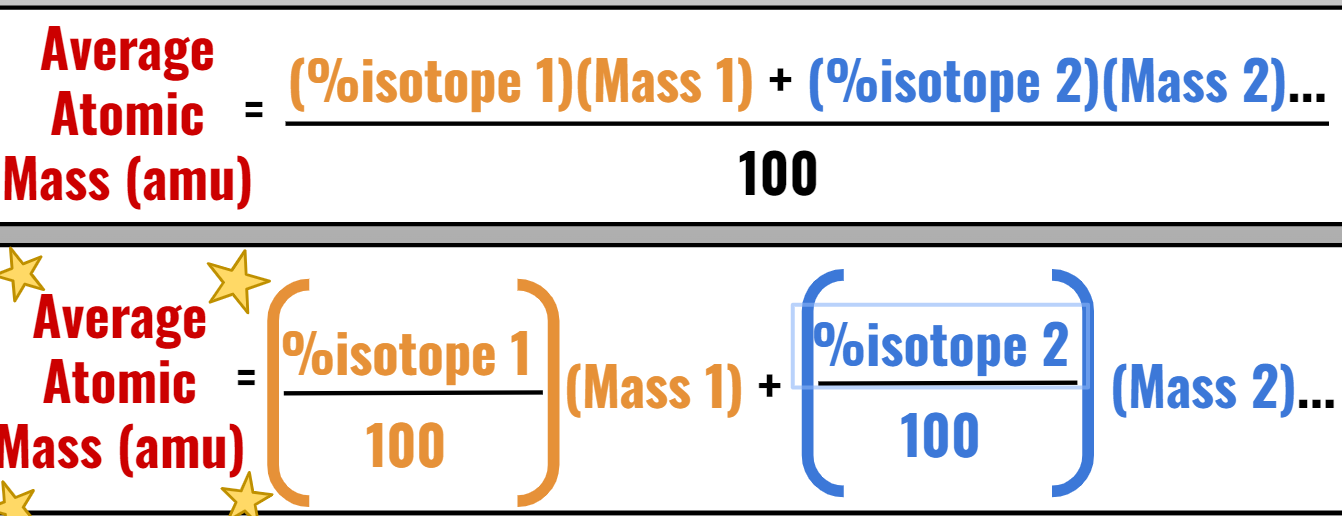
AVERAGE ATOMIC MASS:
Average Atomic Mass definition: weighted average of all the naturally occurring stable isotopes of an element
CALCULATING AVERAGE ATOMIC MASS:
Percent abundance: describes the percent amount of each isotope in nature
Most Abundant Isotope (most common) has the highest percent!
Overall Abundance adds up to 100 %.
Formula:
FOURTH TOPIC: IONS:
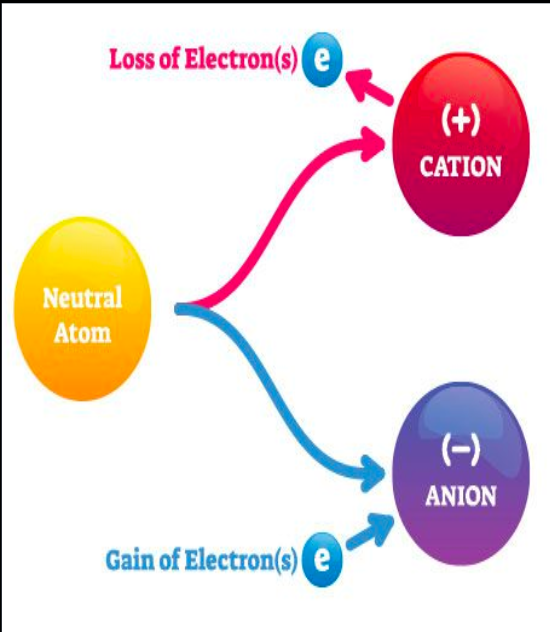
Ions:
Are atoms with a charge
A charge is created because the number of protons IS NOT equal to the number of electrons.
(IONS CANNOT LOSE/ GAIN PROTONS. ONLY ELECTRONS; this is because if you change the # of protons, you change the element)
CATIONS VS ANIONS:
CATIONS:
Cations are ions with positive (+) charges.
Cations are created when atoms lose electrons.
ANIONS:
Anions are ions with negative (-) charges.
Anions are created when atoms gain electrons.
FIFTH TOPIC: BOHR MODELS
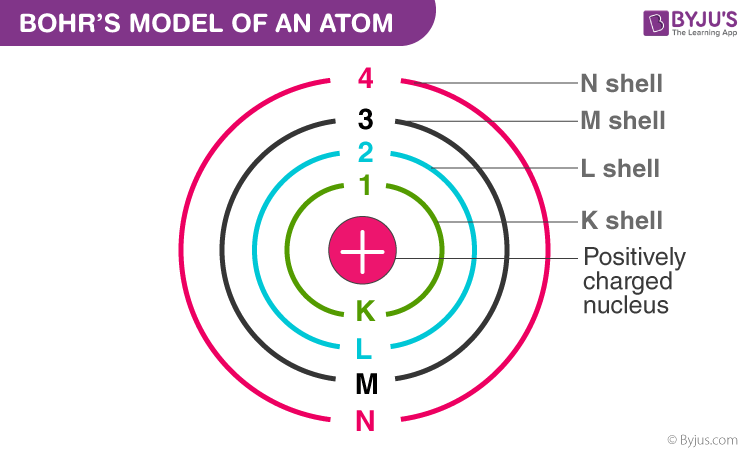
Electron Configuration:
Electrons orbit the nucleus in fixed energy levels called Energy Levels or Energy Shells.
The Electron Configuration of an atom tells us the number of electrons in each shell.
The Electron Configuration of an atom is located on the Periodic Table.
Electron Shells or Energy Levels:
Principle Energy Levels (Shells) that are furthest away from the nucleus contain electrons with the highest energy.
1st Principal Energy Level can only hold a max of 2 e-
2nd Principal Energy Level can hold a max of 8 e-
3rd Principal Energy Level can hold a max of 18 e-
4th Principal Energy Level can hold a max of 32 e-
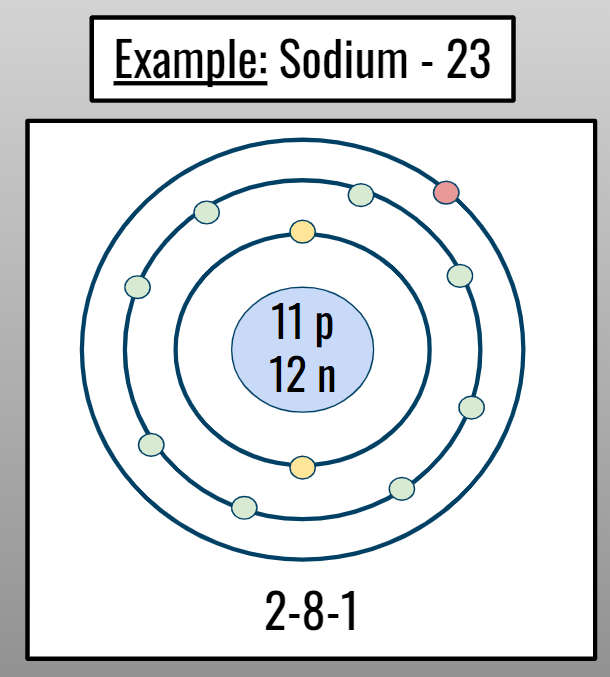
Drawing Bohr Models:
Determine the amount of p+ and n0. Write in the nucleus.
Find the element’s electron configuration
Write or draw the number of electrons in each shell.
SIXTH TOPIC: GROUND & EXCITED STATES
Ground State:
The Electron Configuration for the Ground State is on the Periodic Table.
Electrons in the Ground State are filled from lowest energy level (shell) to highest energy level (shell).
Atoms in the Ground State are stable and have low energy.
Excited State:
Excited State electron configuration is different from the Periodic Table.
An electron is moved from a lower energy level (shell) to a higher energy level (shell)
Atoms in the Excited State are unstable and have high energy.
How to go from Ground → Excited State
Get the electron configuration of an element from the periodic table:
Ex: Neon (2-8)
Bring a electron from a lower shell to a higher shell
(2-8) → (1-8-1), (2-7-1) etc… (REMEMBER YOU CAN’T GO PAST THE MAXIMUM AMOUNT OF ELECTRONS PER PRINCIPAL ENERGY LEVEL OR SHELL!!!)
SEVENTH TOPIC: BRIGHT LINE SPECTRA
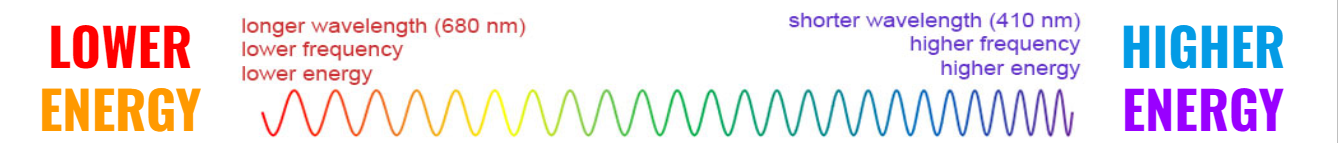
Energy is absorbed when electrons jump from a lower energy level to higher energy level.
(Ground → Excited)
Energy is released when electrons jump from a higher energy level to a lower energy level
(Excited → Ground)
When electrons fall (return) from a higher energy level to lower energy level, energy is emitted (released) in the form of light!
Different electron transitions are related to different COLORS OF LIGHT
Each element will release specific color(s) of light called Bright Line Spectra
No 2 spectra are the same; Bright line spectra are often called an element’s “fingerprint”
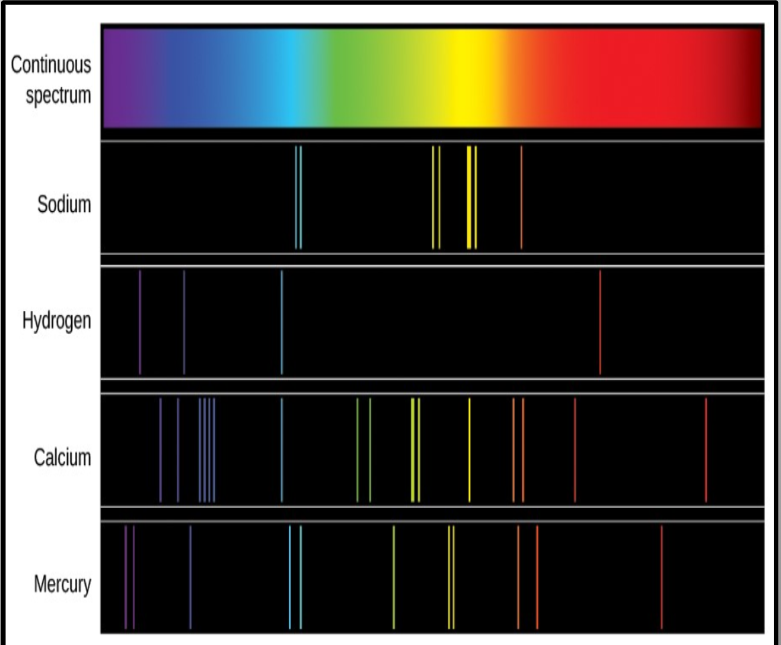
you can tell when a mixture contains specific elements when the lines of the element in the bright line specter appear in the mixture.
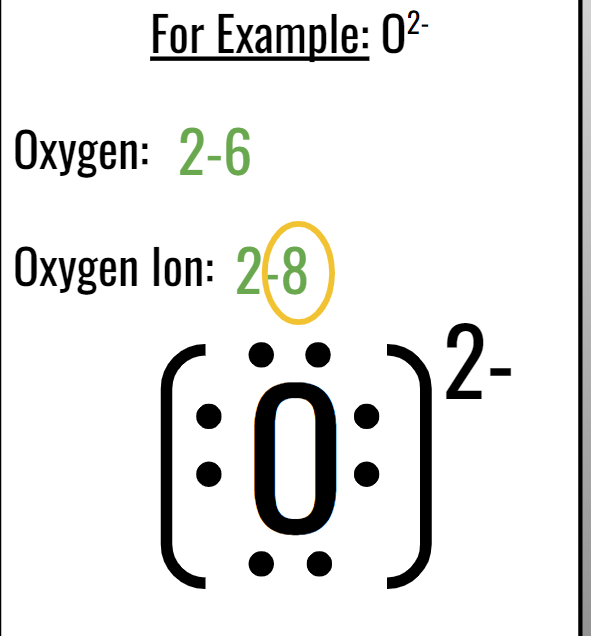
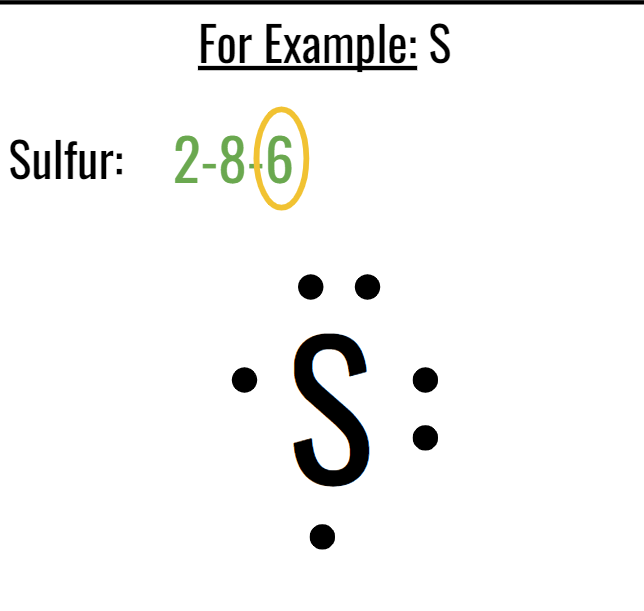
EIGHTH TOPIC: LEWIS DOT DIAGRAMS:
Valence electrons:
Valence Electrons are the electrons in the outermost energy level (shell) of an atom.
The maximum number of valence electrons is 8 e-
Valence electrons are always the last number in an electron configuration
Drawing Lewis Dot Diagrams:
ATOMS:
Write the chemical symbol
Find the electron configuration on the Periodic Table.
Determine # of valence electrons.
Place the valence electrons around the chemical symbol.
(MAKE SURE THEY ARE EVENLY SPACED OUT, DO NOT PLACE THEM RIGHT NEXT TO EACH OTHER, ONLY DOUBLE UP WHEN THERE IS MORE THAN FOUR, GO IN A CLOCKWISE ROTATION)
IONS:
Write the chemical symbol
Find the electron configuration.
Fix electron configuration by looking at the charge.
Determine # of valence electrons.
Place the valence electrons around the chemical symbol.
Add brackets and charge!
(DONT PUT DOTS AROUND CATIONS!)