Chemistry 🧪
Elements
A substance made out of atoms that share the same number of protons and cannot be broken down into simpler substances by chemical methods. Eg- Sodium.
Compounds
Two or more elements that are chemically bonded together (In a fixed proportion). Eg- Carbon Dioxide.
Molecules
2 or more atoms joined by covalent bonds (share electrons). Eg- Water
Atomic Structure
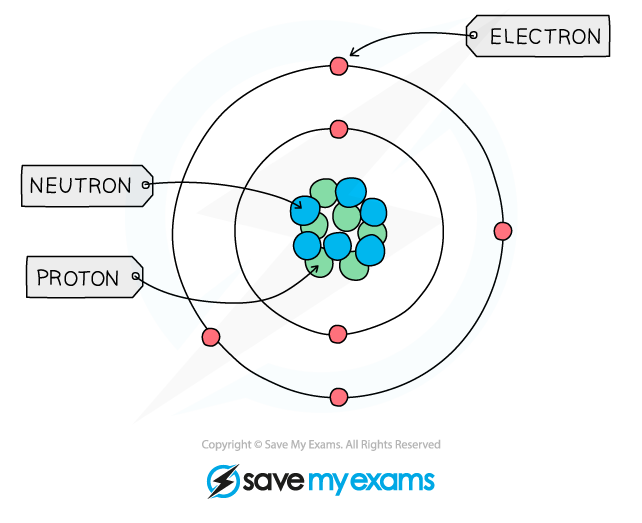
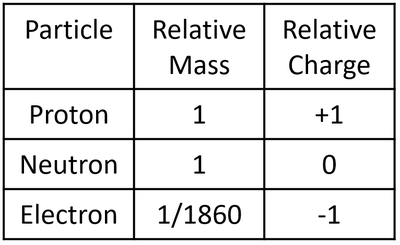
Ionic Bonding
Positive ions= Cations
Negative ions= Anions
Ionic Bonds- Strong electrostatic attraction between oppositely charged ions (Metals and non-metals).
Periodic Trends
Elements in the periodic table of elements are arranged in order of increasing atomic number.
It is made up of rows called periods, and collumns called groups. The position of an element helps determine the electronic configuration.
Period number- number of electron shells
Group number- number of outer shell electrons.
The table moves from metals on the left to non-metals to the right.
Noble Gases
Noble gases are stable, as they have full outer shells.
Properties:
Monoatomic and colorless
Uses:
Neon (in signs), Argon (in lights), Helium (balloons).
Alloys
A mixture of 2 or more metals or a non-metal and a metal.
Alloys are very useful, as they are stronger/more useful than pure metals.

Alkanes
Alkanes are a group of saturated hydrocarbons.
The term saturated means that they only have one carbon-carbon bonds, and there are no double bonds.
Alkanes are generally unreactive compounds, but they do undergo combustion reactions, where they can be cracked down to smaller molecules that react with halogens in the presence of light.
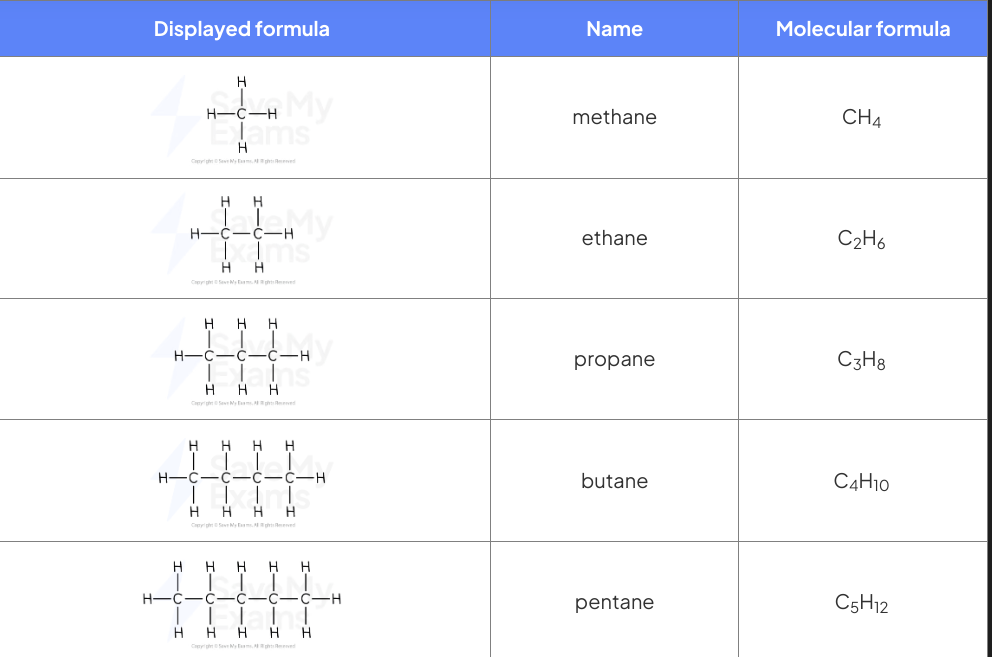
Stoichiomestry
State symbols:
(s)= solid
(g)= gas
(l)= liquid
(aq)= aqueous solution
Reactivity Group 1
Very reactive elements, as they only have 1 electron in their out shells, so they are unstable.
Composition of Air
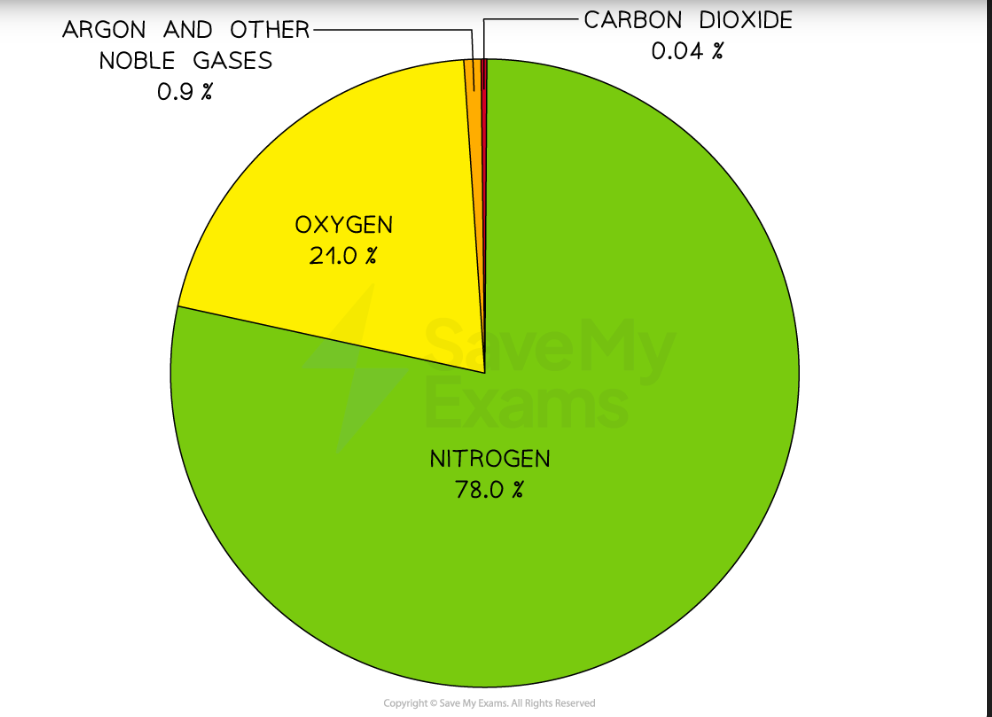
Air Pollution
In addition to the gases that are present naturally in our atmosphere, other gases are also present due to human activities and are classified as air polluntants.
These include:
Carbon dioxide, Carbon Monoxide, Methane, Oxides of nitrogen, and Sulfur dioxide.