AP Bio Unit 1 Study Guide
Water
non living thing that all living things depend on
polar substance
one side (hydrogen atom) is positively charged, the other side (oxygen) is negatively charged
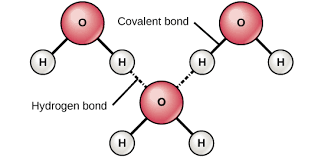
allows for water to form hydrogen bonds
hydrogen bond: a weak bond between the negative and positive regions of 2 separate molecules
Properties of Water
cohesive/adhesive behavior
cohesion: when 2 of the same molecules form hydrogen bonds
adhesion: when 2 different molecules form hydrogen bonds
moderate temperature
has a high specific heat capacity
due to waters cohesive properties
absorbs thermal energy & resists changes in temp.
heat is absorbed when hydrogen bonds break
heat is released when hydrogen bonds form
allows life to thrive by keeping areas near water cool in the summer and warm in the winter
areas remote from water are not able to regulate temp.
universal solvent
due to it’s hydrogen bonds/polarity it is a good solvent in the liquid state
when dissolving an ionic compound each ion is surrounded by a sphere of water molecules
hydration shell
large molecules can be dissolved in water if they have ionic and polar regions
surface tension
measure of how difficult it is to break the surface of a liquid
has a high surface tension
due to hydrogen bond forces between water molecules
caused by cohesion
ice floats
ice is a solid = hydrogen bonds are more packed/ordered
ice is less dense than water
causes it to float
Dehydration vs. Hydrolysis Synthesis
dehydration synthesis: water is produced from OH & H —> creates a bond between monomers (creates covalent bonds)
the covalent bond caused by dehydration synthesis in carbs is called glycosidic linkage
the covalent bond caused by dehydration synthesis in lipids is called ester linkage
the covalent bond caused by dehydration synthesis in polypeptides is called peptide bonds
hydrolysis synthesis: adds water which separates polymers into monomers (breaks bonds)
Macromolecules
comprised of 6 main elements: hydrogen, oxygen, carbon, nitrogen, sulfur, and phosphorus
sulfur
primarily found in proteins
phosphorus
found in nucleic acids and phospholipids
4 main types: proteins, polypeptides, nucleic acids, lipids, and carbohydrates
Carbohydrates
base building blocks for sugar
simplest carbohydrates are monosaccharides (simple sugars)
macromolecules of carbs are polysaccharides
examples are glucose, starch, cellulose, glycogen
serve as fuel and building material
Starch
form of storage
storage of polysaccharide in plants
contains glucose monomers
simplest form is amylose
plants store surplus starch as granules within chloroplasts
Glycogen
storage polysaccharide found in animals
mainly stored in liver and muscle cells
Cellulose
a polysaccharide
major component of plant walls
a polymer of glucose
different glycosidic linkages than starch
Lipids
diverse group of hydrophobic molecules
do not include true polymers
commonly fats, phospholipids, and steroids
store energy, make up cell membranes
Saturated Fats
max. number of hydrogen bonds and no double bonds
typically animal fats
solid at room temperature
Unsaturated Fats
have one or more double bonds
liquid at room temperature
typically plant and fish fats
Polypeptides
unbranched polymers built from amino acids
20 different types of amino acids build up proteins
bonds between amino acids are called peptide bonds (covalent bonds)
proteins are functional molecules comprised of polypeptides
unique linear sequence of amino acids
with carboxyl (c-terminus) end and amino terminus (n-terminus)
Amino Acids
organic molecules with amino (NH2) and carboxyl (COOH) groups
differ in properties depending on their side chains
R groups
amino acids differ depending on their R groups
Proteins
Changes to Functions
physical and chemical conditions can affect the structure
changes in pH, temp., salt concentration
protein can unravel
loss of protein’s natural structure = denaturation
protein is biologically inactive
cannot perform function
Primary Structure
dependent on genetic information;
Secondary Structure
found in most proteins; consists of coils & folds in polypeptide chain
does not include R groups
all within the polypeptide backbone
Tertiary Structure
overall shape of the protein is formed
determined by interactions of the different R groups
Quaternary Structure
when 2+ polypeptide chains form a single macromolecule
Nucleic Acids
store, transit, and transport hereditary information
2 different types: deoxyribunucleic acid & ribonucleic acid
DNA (Deoxyribunucleic acid)
DNA has 4 nucleotides: Adenine, Guanine, Thymine, Cytosine
bonds between matching pairs form hydrogen bonds
nitrogenous bases
complementary base pairing (A—> T, G—> C)
two polynucleotides that form a double helix (always)
phosphate backbones that run from 5’ —> 3’
anti-parallel
one molecule holds many genes
RNA (Ribonucleic acid)
single stranded
complementary base pairing can occur from two different RNA molecules or the same RNA molecule
no thymine —> replaced by uracil
A and U pair
nitrogenous bases
RNA molecules are more variable in form