Spectroscopy Notes
Spectroscopy
Spectroscopy is the study of the interaction between matter and electromagnetic radiation (light).
It involves measuring the absorption, emission, or scattering of light by substances.
The goal is to analyze energy transitions within the material, providing information about structure, composition, and properties.
Common Spectroscopic Techniques:
Spectrometry: quantification and analysis of matter based on interaction with electromagnetic radiation.
Measures emitted light (atomic emission spectrometry) or detects ions (mass spectrometry).
Examples:
Mass Spectrometry (MS): Measures mass-to-charge ratios of ions.
Atomic Absorption Spectrometry (AAS): Measures light absorption by atoms in a vaporized sample.
Atomic Emission Spectrometry (AES): Measures the light emitted by atoms after they are excited.
Nuclear Magnetic Resonance (NMR): Measures magnetic properties of atomic nuclei.
X-ray Spectrometry: Measures the interaction of X-rays with matter.
Spectrophotometry: measures the intensity of light absorbed or transmitted by a sample at different wavelengths.
Involves measuring the absorption spectrum of a sample in a specific region of the electromagnetic spectrum (UV, visible, or IR light).
Examples:
UV-Visible Spectrophotometer: Measures absorption in the ultraviolet and visible regions (200-800 nm).
Infrared (IR) Spectrophotometer: Measures absorption in the infrared region (4000-400 cm-1).
Fluorescence Spectrophotometer: Measures the fluorescence emitted by a sample after excitation by a specific wavelength of light.
Instrument Components
Typical spectrophotometer components:
Light Source: provides radiation to interact with the sample.
Monochromator: selects a narrow band of light from the source and transmits it to the sample at a specific wavelength.
Sample Holder: contains the sample through which light passes.
Detector: captures the light and converts it into an electrical signal.
Data Processor: processes the electrical signal into a readable output (absorbance, transmittance, fluorescence intensity).
Focus On: UV-Vis Spectrophotometer
Analytical instrument used to measure the absorption or transmission of light in the UV and visible regions (190 nm to 1100 nm).
Working Principles:
Absorption of Light: Molecules undergo electronic transitions when they absorb light.
Beer-Lambert Law: Correlates absorbance with concentration and path length.
Types of UV-Vis Spectrophotometer
Single-Beam Spectrophotometer
Uses a single optical path.
Advantages: Simple, cost-effective, easy to operate.
Disadvantages: Requires frequent recalibration and measurement of a reference solution.
Double-Beam Spectrophotometer
Uses two separate beams for the sample and reference.
Advantages: Better accuracy due to simultaneous measurement, minimizes errors from light source fluctuations.
Disadvantages: More complex and expensive.
Beer-Lambert’s Law
Determines how the amount of attenuation depends on concentration and path length.
Where:
is transmittance.
is transmitted light.
is incident light.
is Absorbance
Where:
is the absorbance.
is the molar extinction coefficient (L/mol·cm).
is the path length in cm.
is the concentration (mol/L).
Beer-Lambert’s Law Practice Problems:
A researcher is studying the absorbance properties of a substance in solution. The molar extinction coefficient of the substance is given as 250 L/molȉcm, and the concentration of the solution is 0.100 mol/L. The path length of the cuvette used for the experiment is 1.50 cm. What is the absorbance?
• A 7.50 x 10-5 M solution of KMnO4 has a transmittance of 36.4% when measured in a 1.05-cm cell at a wavelength of 525 nm. Calculate the absorbance and molar absorptivity of the solution.
• In a laboratory, a chemist is analyzing a sample of a compound to determine its molar extinction coefficient. The absorbance of the solution at a given wavelength is measured to be 0.620. The concentration of the compound in the solution is 0.025 mol/L, and the path length of the cuvette is 2.00 cm. What is the molar extinction coefficient of the compound?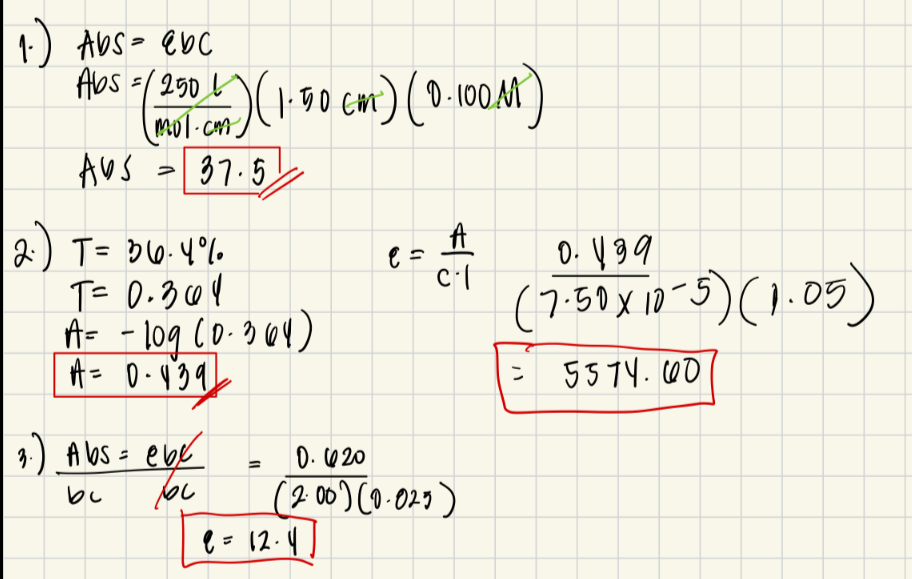
Limitations of Beer-Lambert’s Law
Chemical deviations:
Solution is too dark (exceeds 0.01 M).
Absorbing species undergoes association, dissociation, or reaction with the solvent.
Instrumental deviations:
Presence of stray radiation (or stray light).
Mismatched sample cells.
Quantitative Measurements in Spectroscopy
Light is absorbed differently across wavelengths.
Plotted as spectra (absorbance vs. wavelength).
Each molecule has a unique spectra.
(highest peak)
Indicator of energy transitions occurring in the substance's molecules.
Complementary color of the indicates the solution's color.
Quantitative Spectroscopic Analysis
Set the monochromator to the and observe changes in absorbance with changing concentrations.
Recall:
A student prepared an external calibration curve with four standard solutions of KMnO4. If the unknown gives absorbances of 0.621, 0.641, 0.628, and 0.619, what are the corresponding concentrations?
Concentration (M) | Absorbance |
|---|---|
0.005 | 0.217 |
0.010 | 0.432 |
0.015 | 0.641 |
0.020 | 0.867 |
Quantitative Measurements Notes
The squared Pearson correlation coefficient () is the measure of linearity.
A value closer to 1 means that there is a strong correlation between absorbance and concentration.
Ideally, we prefer , but for the experiment, an is enough.
The higher the slope, the more sensitive the measurement is.