3/31: Carboxylic Acids and Polymers
Introduction to Carboxylic Acid Derivatives and Nitriles
HSO4 in water forms carboxylic acids.
Nitriles are unique as they do not have a carbonyl carbon but behave similarly in reactions when converted to carboxylic acids through hydrolysis.
Nucleophilic Acyl Substitution
Key reaction process for carboxylic acid derivatives.
The carbonyl carbon has a partial positive charge due to electronegativity differences with oxygen.
Less sterically hindered due to sp² hybridization, allowing nucleophiles to approach easily.
Reactivity order: Acid halides > Anhydrides > Esters > Amides.
Converting Derivatives
To convert an acid halide to a carboxylic acid use thionyl chloride or go through carboxylate back to acid.
Grignard reagents can react with esters to yield alcohols:
Methyl ester -> secondary alcohol.
Other esters -> tertiary alcohols.
Reaction with grignards and nitriles leads to imines, which can hydrolyze to ketones (very useful reaction)
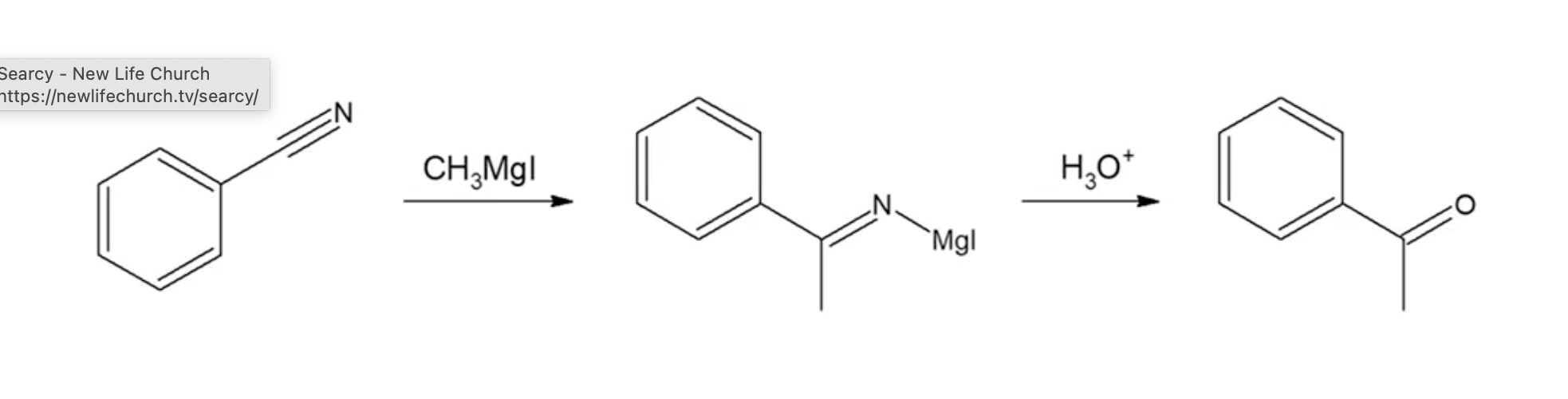
Gilman attacks ester or acid halide only once, forming the ketone instead of alcohol. If acid halide and ester, Gilman is stable and will only attack the most stable position (not ester)
Reactivity of Esters and Reducing Agents
Reviewed two main reducing agents: Lithuim Aluminum Hydride (LAH) and Sodium Borohydride (NaBH4).
Lithium is more reactive and must be used in inert conditions. Ester or amide and reduced to primary alcohol.
NaBH4 is selective for aldehydes/ketones in the presence of esters/amide.
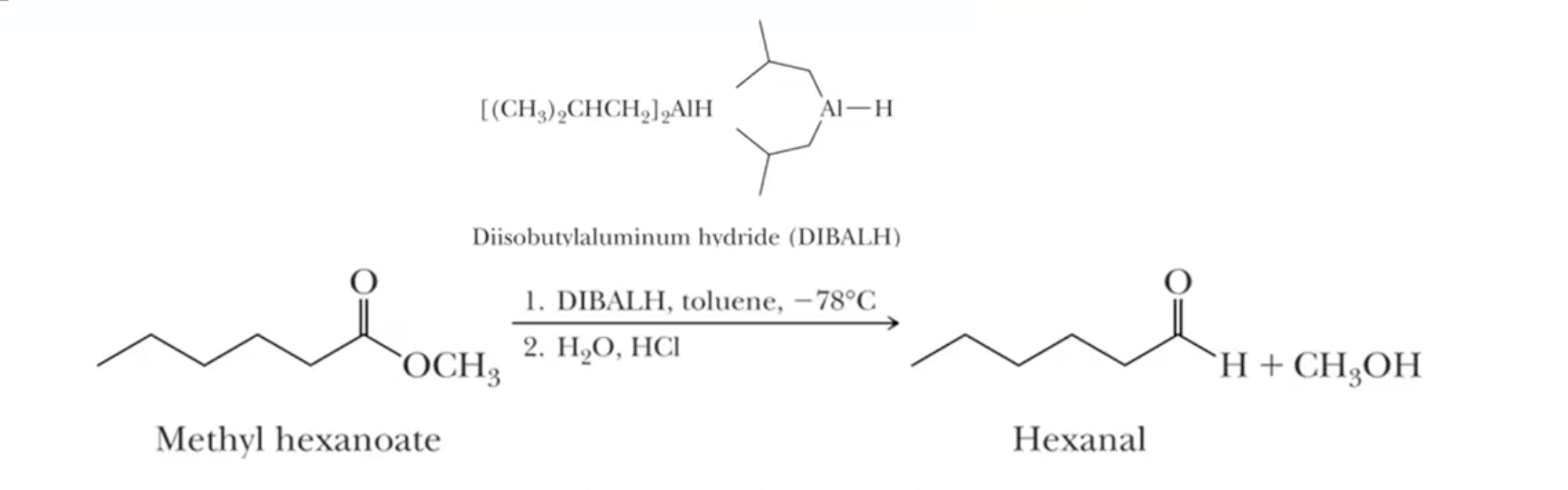
Diisobutylaluminum hydride (DIBAL-H) selectively reduces esters to aldehydes at -78°C, achievable with a dry ice and acetone bath. At room temperature goes all the way to the alcohol.
Primary, Secondary, and Tertiary Amides
Primary amines converted to secondary amides using acid halides, further reduced to tertiary amides.
Highlighted the difference in reactivity between nitriles, alcohols, and carboxylic acids.
Making Nitriles and Challenges
Simplest formation via SN2 reactions with primary/methyl halides or tosylates.
More challenging to introduce cyano groups on aromatic rings.
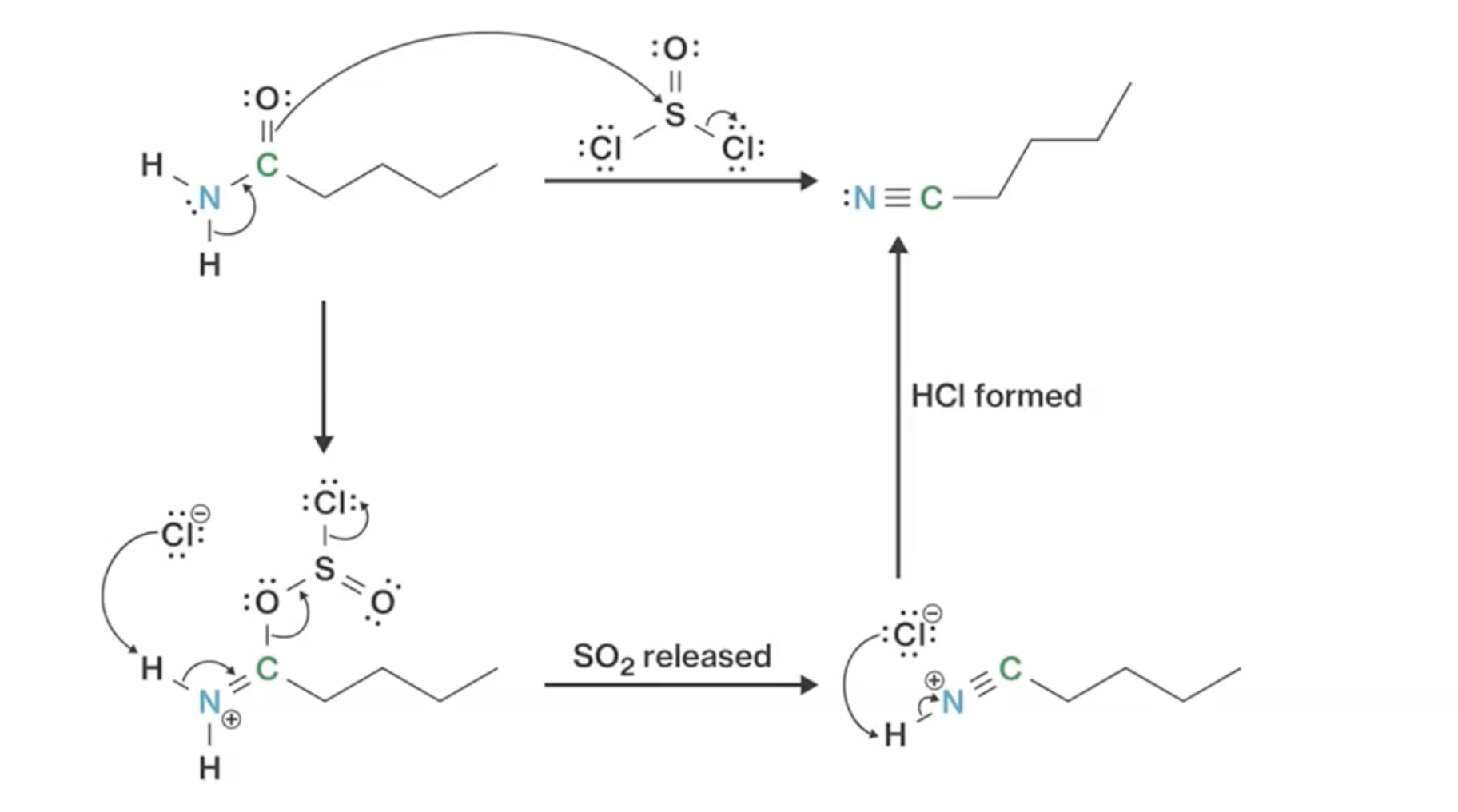
Conversion of amides involves sulfur bond formation, key in the reaction mechanism, pulling protons to form nitriles with sulfur as a better-leaving group.
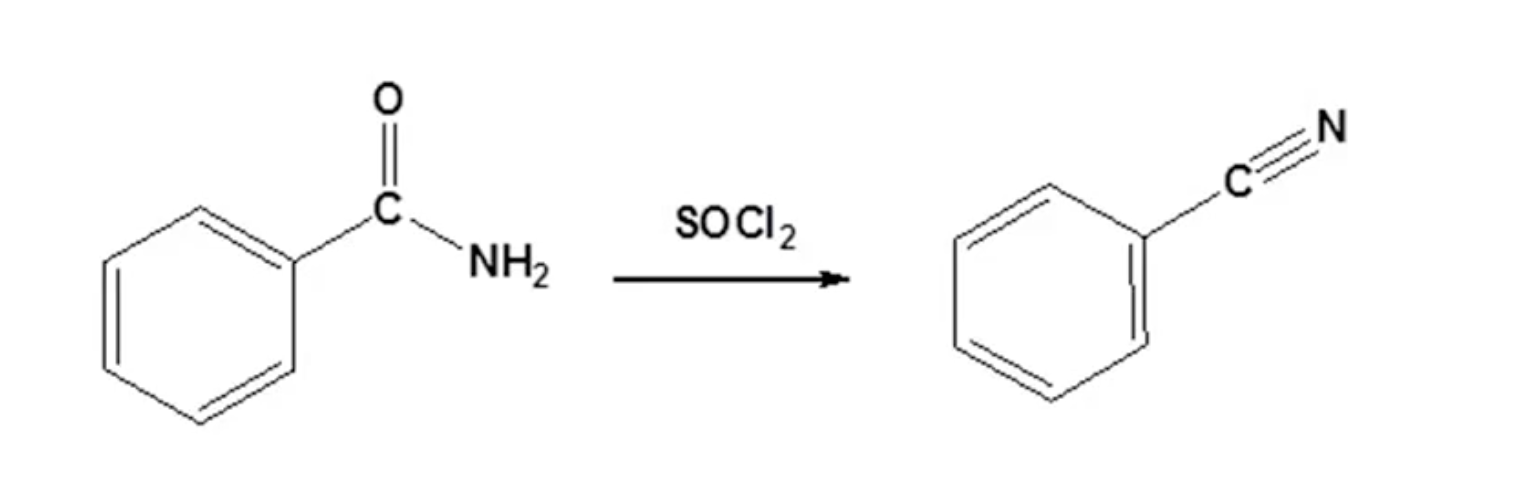
Synthesis of Nitriles Mechanism: sulfur-oxygen bond is very strong, SO2 good leaving group
Introduction to Polymers
Shifted focus on larger organic molecules, specifically polymers formed from small repeating units.
Defined oligomers vs. polymers: Oligomers with less than 10 repeat units, become polymers beyond that.
Connections to natural polymers (cellulose, proteins) versus synthetic polymers (plastics).
Properties and Uses of Polymers
Discussed applications of polymers in everyday life and industry, mentioning the high production of synthetic polymers globally (50 trillion pounds of synthetic polymers manufactured each year).
Polymer: substance composed of macromolecules, molecules with a large number of atoms and high molecular weight. Has tens of thousands of atoms.
Highlighted structural variations (tacticity) and implications for physical properties. Have to consider how monomers are connected as a polymer.
Explored primary polymers (proteins) and reactions creating synthetic counterparts like vulcanized rubber and nylon.
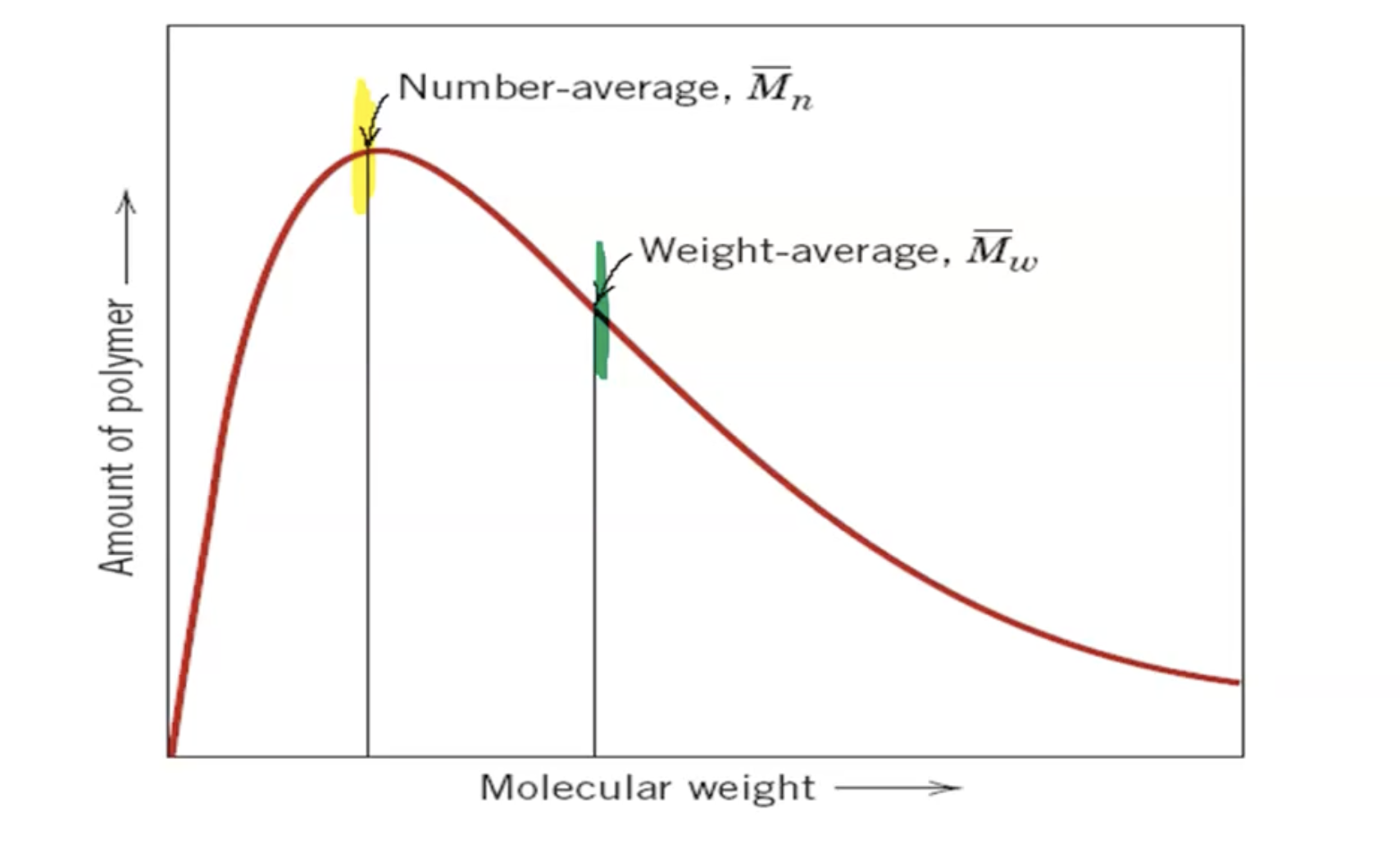
Analysis of Polymer Size and Weight
Not all polymer chains grow to the same length
Average molecular weight is determined by measuring viscosity and osmotic pressure
Chain divided into size ranges.
Introduced concepts of number average molecular weight (Mn) and weight average molecular weight (Mw).
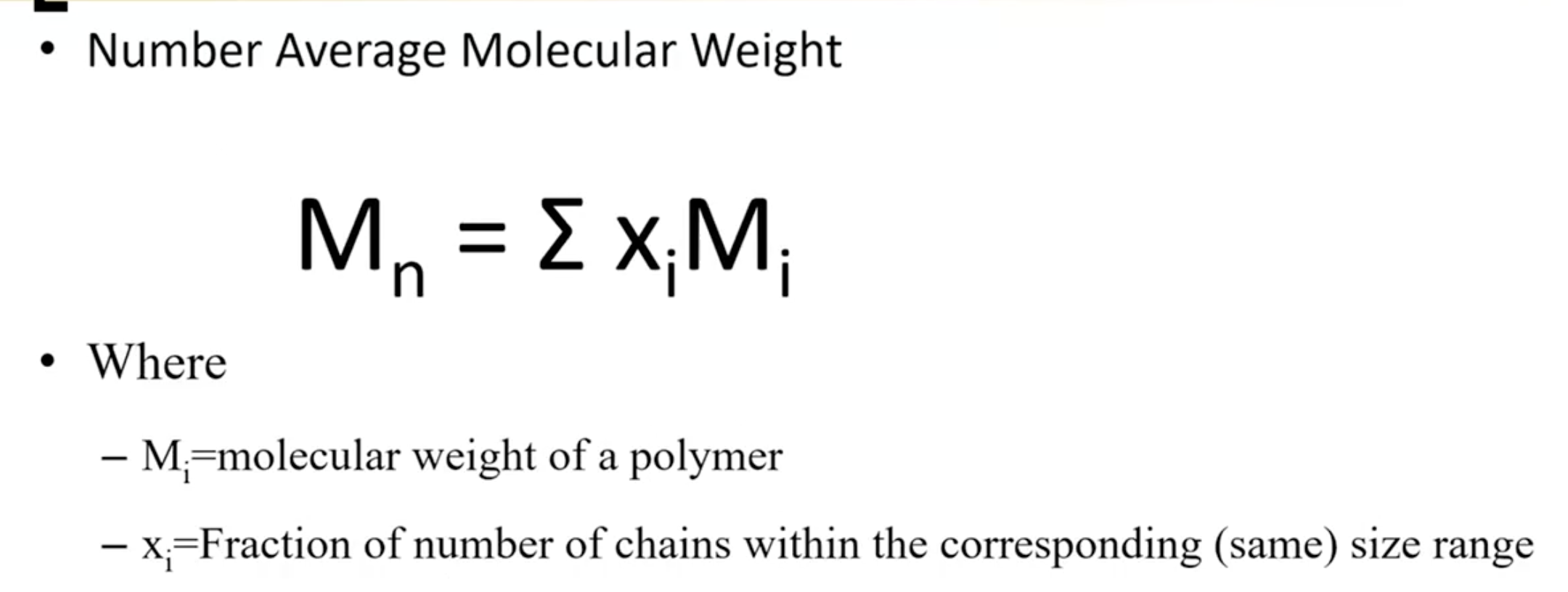

Discussed methods to calculate averages and implications for polymer characteristics, with examples comparing averages.
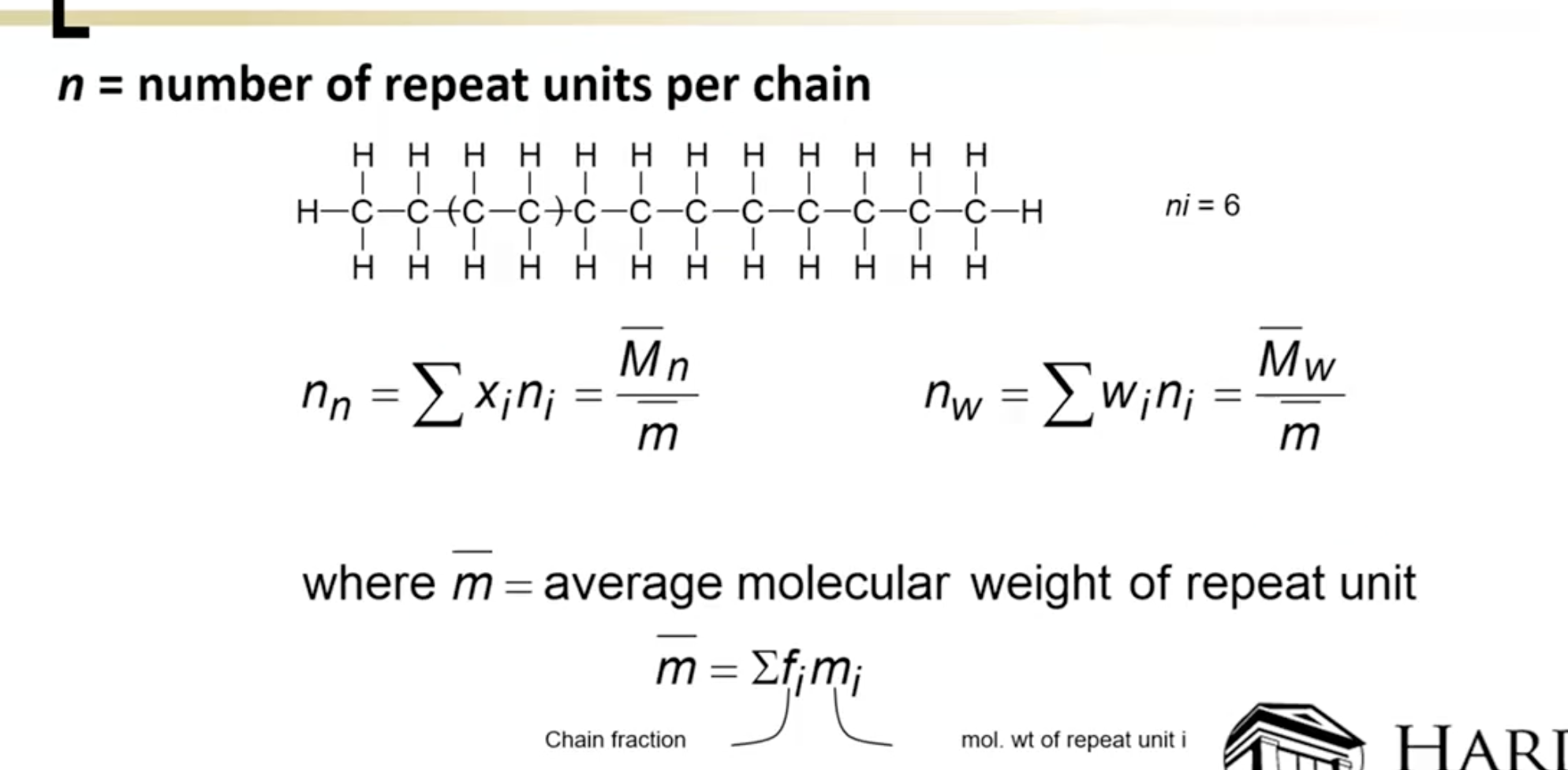
Addressed the degree of polymerization (n value) and the significance of shape and organization of polymer chains in functionality.
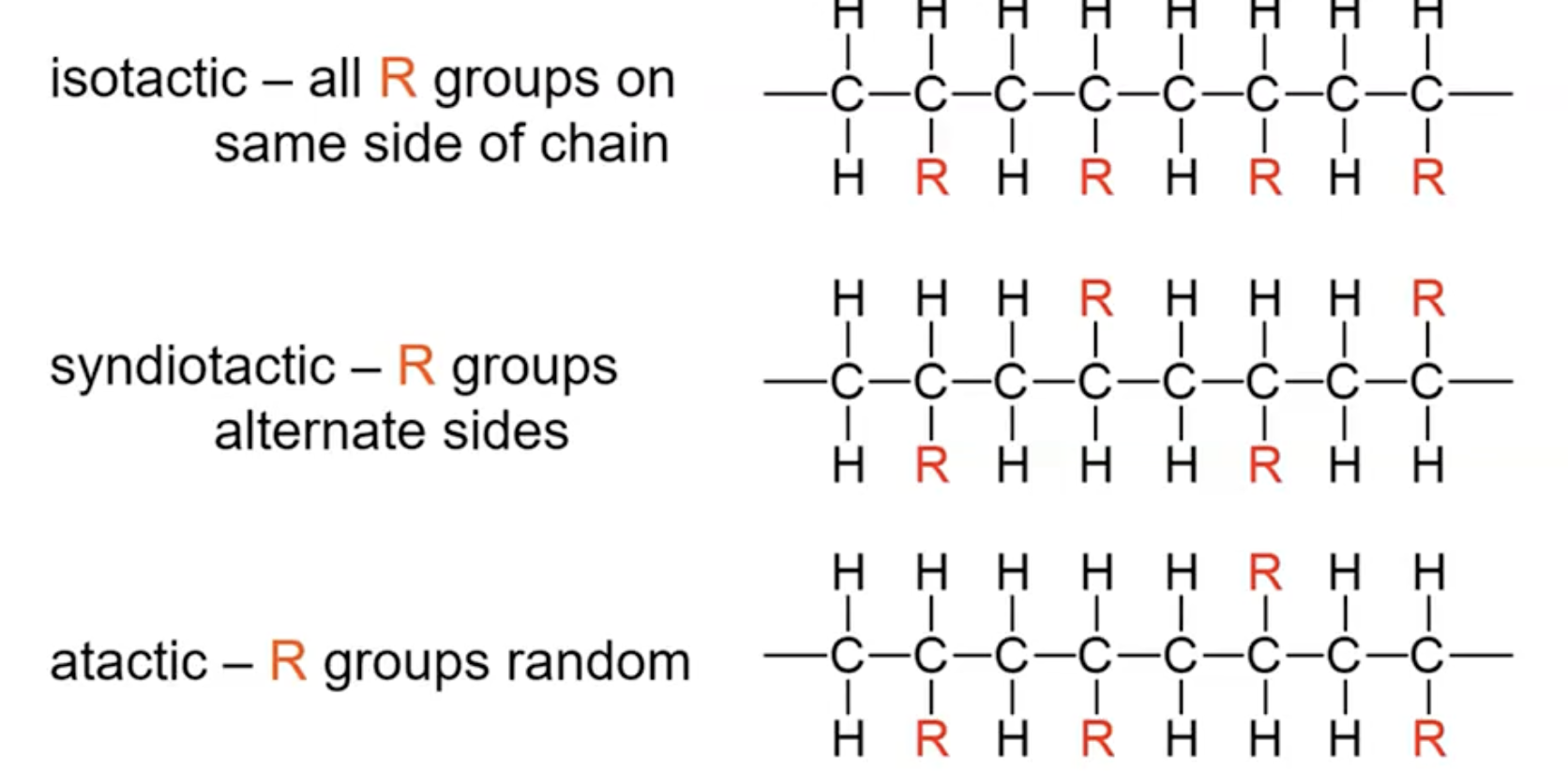
Tacticity: stereoregularity of side chains
Order of tacticity affects its physical properties
What drives a polymer formation is its specific function
Environmental Considerations
Noted implications of synthetic polymer usage on the environment and potential strategies for sustainability.
Emphasized careful monitoring of polymer degradation and recycling methods.
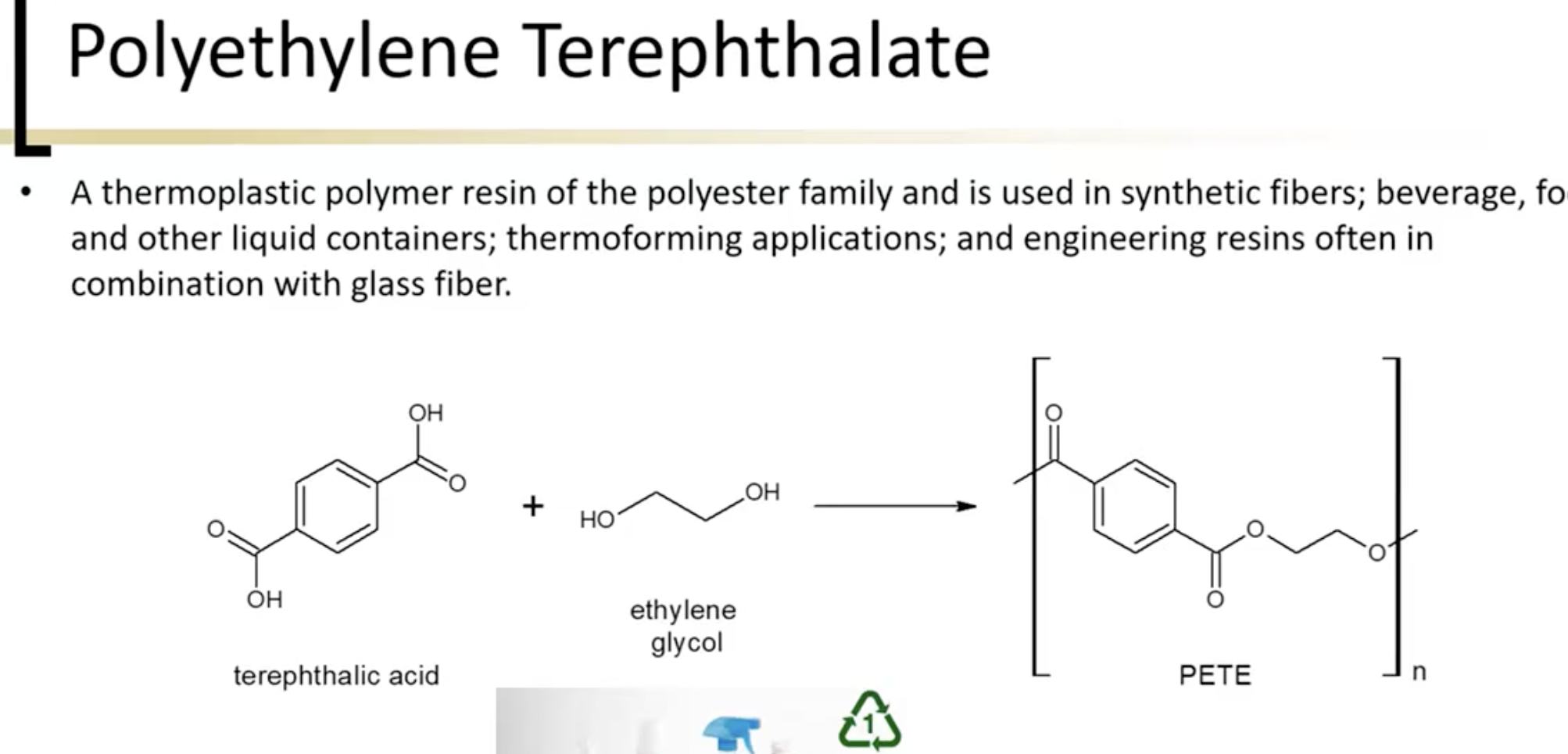
such a good polymer because it’s easily made by fischer esterification and broken by saponification. Good for environment.
All polyamides are polyesters; not all polyesters are polyamides