Toxicology
Paracelsus→ father of toxicology→ discovered that dosage makes the poison a Swiss physician and also said that disease was its self an entity
risk= hazard* exposure
should consider individual differences like timing (age) route of exposure (ie drinking mercury not as toxic as it’s vapors) and toxicity is species dependent
toxicology→ the study of adverse physiological and chemical effects of chemical or physical or biological agents on living systems or their ecosystem
is a multi-discipline thing involving biology, chemistry, pathology, pharmacology, genes and epigenetics where application in analytical, chemical, environmental, forensics, occupational/ regulatory fields
clinical toxicology→ connection between toxic substances and substrate illness
forensic toxicology→ establishes medical or legal aspects of toxicology to find a cause or responsibility of death or exposure to toxin
Heroin is more lipophilic therefore more bioavailable with the acetal groups attached
this makes it more potent then morphine
atrazine → great for farmers and easy to make but its side effects are still debated.
most toxicological studies don’t have conclusions but a benefits vs con list
example: PFOS and PFOA very little data about this nonstick compound this can build up where long-term consequences are unknown
pyridine, nanoparticles and PEG are examples of unknown toxic effects
PEG thought to be non-immunogenic and is found in cosmetics
lead poisoning seen mostly in products with lead (II) acetate and lead oxidizes used for paints also used in gas as a antilocking agent
LD50 or lethal does 50→ the dose required to kill 50% of subjects in experiments → is a dead end but can be good to know for study of effects on systems
also requires species to be mentioned
Mathieu Orfila→ father of forensic toxicology→ found that chemical agents such as arsenic can be used to kill people that can be found via post death report
precaval pott found high amounts of scrotal caner in chimney sweepers due to Polycyclic aromatic hydrocarbons (PAH)
conducted study that allowed legation change that forbid children younger than 8 to drop down the chimney to clean it →1st occupational safety law
Francois Magendic (1783-1855)→ looked at experimental pharmacology and identified the 3Rs of animal work→ replacement, reduction, refinement
drug development cost $26 billion/ drug in 2015 more costly if the drug doesn’t work in the later stages of development
usually testing in initial stages occurs at enzyme and substrate validation →does it have an effect as a medication then you develop lead → drug optimization → testing: preclinical in animal models and stuff →phase 1 clinical trial ( limited to healthy individuals) phase 2 check efficacy as well→ phase 3 is large scale test on population once approved→ phase 4 testing→ market testing
same process used in vaccines
discovery stages can take 6-7 years
a lot of drugs have been withdrawn from the market due to adverse side affects.
1950-2013→ 644 drugs were withdrawn from the market and 462 toxicity related
Toxicology for meds→ stay away from good electrophiles (ie acid chloride) cause it will most likely react with water other things are avoided like aziridine and epoxy compounds cause they’re reactive also include things like nitro groups and aromatic nitrogen
toxicants→ general term for a substance that produce adverse biological effects these can be physical biological or chemical
toxins→ toxicants that are made by living organisms most are proteins but don’t have to be proteins
poisons → toxicant that cause immediate death or illness even with small doses
toxins have different categories:
systematic→ affects many organs or entire body like cyanide which inhibits cellular respiration
organ/ tissue toxicant→ only affect certain organ or tissue ie benzene known carcinogen that affects the kidneys and bones marrow
cellular respiration: happens in cytochrome oxidase being specific uses 4 electrons + 4H+O2 →2 H2O and CN is a nonspecific inhibitor of the enzyme
calculating amount needed to create toxic affect use the following info: total number of cells in body (without micro-organisms) 1013 and 5L of blood in the body ,cyanide lethal does is 1ug/ml blood
lethal concentration= 0.005g of cynide in the blood which is 1.92×10-4 moles of CN in the blood which is (1.92×6.022×1023) 1.16×1020 ions which means there is 1.15×107 ions per a cell
some systematic toxic affects only need 1 proton to change the pH
acute systemic toxicity→ after exposure the affect is immediate
sub chronic → after repeated exposure (days or weeks) can lead to side effects
chronic→ after months or years you seen affects
genetic toxicity → alters gene expression
developmental toxicity → toxic towards embryo or fetus
toxicological paradigm→ toxicokinetic or what do we do the toxicant?→ ADMET(S)
Absorption of the toxicant
distribution of the toxicant
metabolism of the toxicant
excretion of the toxicant
Toxicology
storage of the toxicant
toxicodynamic→ what toxicants do to us
does and exposure → dose is the amount of substance administered at one time units mg/ kg body/ day
tylenol→ species specific toxicity
cucod is a plant rich in starch but also has a compound that can be rinsed off if it’s digested it’s toxic
compound is
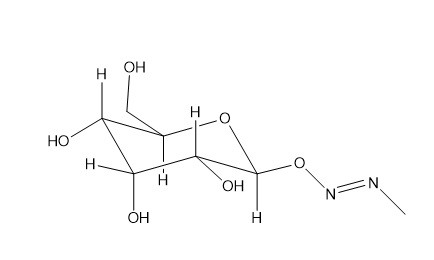 which turns into glucose + methylazoxymethanol→ N=-N-CH3 which is toxic when metabolized in the GI tract
which turns into glucose + methylazoxymethanol→ N=-N-CH3 which is toxic when metabolized in the GI tract
xenobiotic→ chemical substance found within an organism that is not naturally produced or expected to be present within the organism can be drugs or toxicant
exposure to the skin from water, soil, drug, subcutaneous (s.c) or in intramuscular (SM)
Path of xenobiotic:
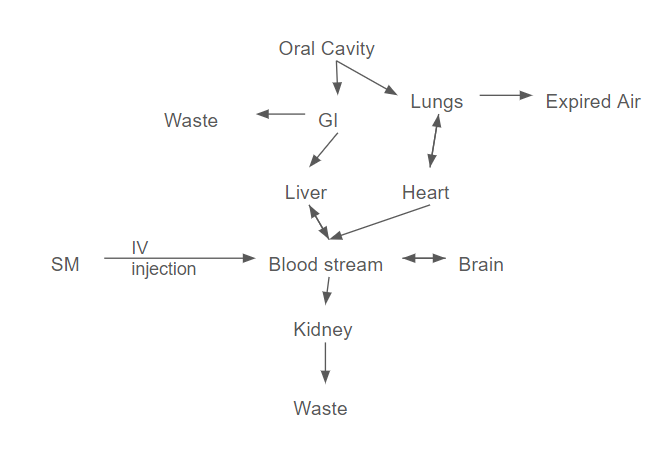
BBB (blood brain barrier) → physical barrier that prevents exposure to xenobiotics from entering the brain that’s semipermeable in the central nervous system
the more bioavailable something is the easier it is to cross the barrier
toxicity can have individual sensitivity
LD50 is great for acute toxicity but to establish any affects before death it’s good to use the affective does which is ED50 and looks at when 50% of people have a response to the xenobiotic
hydrazine is a good nucleophile but it’s also a toxicant
the LD50 is not really a concern but buildup is so other factors must be considered
Combined effects→ can be additive affects (combo affect is seen with the 2+ drugs)or no interactions
synergism→ toxic response is greater than additive effects
Potentiation→ when one drug does not elicit a response on its own but enhances the response to another drug
coalitive→ 2 substances that might lead to a toxic response different from either of the two substances
antagonism→ bind to endogenous receptors but don’t produce a response
ie B12 being the antidote for CN poisoning
effective does → does gives efficient in 50% of subjects
TD50→ produces a toxic response in 50% of subjects
therapeutic index→ measure of TD50/ ED50 (toxic dose/ effective dose)
higher TI the lower chance toxic side affects occur
TI>10 is safe; while TI<2 is narrow therapeutic drug
morphine has a TI of ~10 so it’s considered safe but this is because the ED is low which increases the TI
Disadvantages of TI→ when less then 2 you need constant monitoring cause it’s not technically unsafe
margin of safety→TD1/ ED99 (TD in 1% of subjects/ ED in 99% of subjects) → more strict ranges to tell if a drug is safe
Threshold does→ minimum dose vs, exposure upon which a biological response starts
dose response curve starts at non zero for example CO curve starts at 10% cause hemoglobin has a turnover
toxicity is affected by forms the arsenic or Cr vs dosage exposure, species, age, and sex differences the ability of toxicant to be adsorbed and it’s metabolism and excretion as well as the presence of other chemicals and there interactions
recall DMATS
drugs can also be absorbed in different ways depending on the cell
active transport→ when it goes against the concentration gradient and need E to move against
facilitated diffusion means no energy is needed to move the molecules using pores or channels that allow small hydrophilic molecules to pass through
passive diffusion can pass through membrane using concentration gradient
size and shape of the compound matter it has to be lipid soluble and structurally similar to endogenous compounds
hydrophobic effect think detergent it organizes it self into membranes so nonpolar side doesn’t interact with water
Ficks law: rate of diffusion= k*A(C(inside)-C(outside))/ d where the surface area, d is the thickness of the membrane, k is the diffusion coefficient,
higher K values easier to diffuse
lipophilicity→ affinity of compound to lipophilic environment
partition coeffect → measure of relative affinity of a molecule for the lipid where octanol is used as solvent basically measure Xaq→←Xoctanol
p=[X] oct/[X]aq x
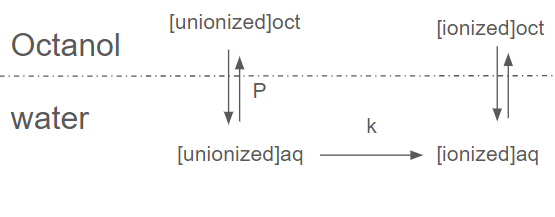
usually expressed as log for neutral things
distribution coeffients→ consider ionization accounts for ionization
higher the lipophilicity the easier it is for compounds to go through the cerebral spine fluid and into the BBB
ionized form low lipid soluble therefore don’t diffuse through membranes easily including the BBB
can use pKa to find this
nitro groups on a compound will make it more acidic
Recall: acid and base dissociation Ka=[H]{A]/ [HA]=> log([A]/{HA]=log Ka-log[H]=>[A]/ [HA]=10ph-pKa
practical definition of pKa→pH where acid is 50% ionized
2 units less than pKa acid unionized
2 units more than pKa base is unionized
amphoteric compounds must look at different components to determine if they are ionized or not
sulfonamides are used to inhibit carbonic anhydrase which is cell membrane by conjugate base with the highest potency at pKa of 7: an ionic species can’t get across cell membrane
makes sense cause free acid is needed to go through the base while being basic is potent
things to consider when crossing membrane:
only neutral species diffuse readily
is there a concentration gradient
can you remove it from across the membrane via circulation ex: the gradient is in place
example of the consideration: benzoic acid in the stomach it’s mostly a free acid ([A]/[HA] (=102-4.2=1/398) while in blood it’s mostly base ([A]/ [HA]=107-4.2=1584) so there is concentration gradient across the membrane which is removed from the barrier via blood so benzoic acid is easily absorbed
example of the consideration: aniline by the same calculations above exists as a conjugate acid (unionized) and removal is unfavored in the blood cause it’s a free base (ionized) therefore there is no concentration gradient and aniline is poorly absorbed
want ionized in stomach (acidic species) and unionized in blood (neutral species)
there is a positive correlation between normalized p and BBB up take rate
real world example of this: free basing →cocaine (salt) hydrochloride+ bicarb (some base) →← free base
when inhaled its easier to get high
douresis→ used to remove acidic or basic compounds by changing it’s ability to cross membrane or be removed by blood
leaky gut syndrome→ membrane are breached but but are not where it’s supposed to be
think celiac disease which indigestible peptide products will form opium mimics
oral dosing→ major route of dosing
major route of dosing where oral bioavailability is given by F% or fraction of does / exposure that makes it to systematic circulation found using area under a curve of [drug] vs. time graph
F%= area under curve after oral dose/ after IV
things that affect bioavailability: chemical properties, chemical stability, 1st pass metabolism
First Pass Metabolism
First pass refers to how the liver breaks down things
things 1st exposed to metabolism mainly in the liver before it reaches the rest of the body
toxicity is dependent on the route of the route of exposure→ most direct won’t go through 1st pass iv>in>im/ sc/ dermal>i.p.>PO
where iv>in>im/ sc/ dermal don’t go through 1st pass
ex: THC is hydrolyzed by the liver makes it more active
toxicants can be stored anywhere:
plasma proteins (albumin(HSA))→ 33-50g/L prevents compound from reaching target ie paraoxon and malaoxin are insecticides in 70’s and 80’s both are based phosphate based →HSA is used as a biomarker
liver and kidneys→ water insoluble metals will be stored in the liver while compounds metabolism becomes reversable in kidney which can cause cancer
fat tissue→ good for highly lipophilic compounds → body weight loss leads to an increase of these in blood plasma
more fat tissue the more likely for potential toxicants in obese people
bones are significant when considering Ca salts like Ca10(PO)6(OH)2→ the Ca can be replace by Pb and OH can be replaced bu F
Lipinski rule of 5: MW>500; log p<5; less than 5 H bond donors and less then 10 H bond acceptors would make a compound more bioavailable
molecular size, polar surface and number of rotatable bonds can be used in addition to these to determine bioavailability
the more negative the delta G is the more bioavailable it is
delta G= delta H-T* delta S
Oxygen toxicity and oxidative stress
Ros is the reason why O2 is toxic but is a lousy reactive mis state mismatch
obligate anaerobes are killed when exposed to oxygen as they deplete their reducing equivalents
earth went from a reductive to oxidative environment in prebiotic world had H2, He, NH3, CH4 and water photosynthesis was used to create the oxidative environment N2 and O2 were available
advantage of oxidative is it can be reduced but this also how you get oxidative stress causing the need for pro and anti-oxidants
pro-oxidants→ generate ROS or inhibit antioxidants
antioxidant →quench ROS species
sources of ROS→ protein damage, DNA damage, lipid peroxidation and signaling purposes
NO is a radical that occurs when Arg is metabolized by nitric oxide synthase (NOS) as a gas it goes through the membrane really easily
Arg via NOS→ NO (passes through cell membrane) activates enzyme turns GRP→CGTP which creates an influx of blood flow or degraded by phosodiesterases which decrease influx of blood flow
ROS has 2 classes: radical and non-radical species
radical species include superoxide (O.-) or hydroperoxyl (HO.)
non radical →peroxide (H2O2); hypochlorous acid HOCl. ozone O3, or ONOO peroxynitrile
most O2 is in it’s triplet state where it’s unreactive due to spin state mismatch but singlet O2 is not the case it’s super reactive
if spin states don’t match like O2 reacting with normal organic compounds a lot of energy is needed
in GS O2 is in the singlet state because it gets excited
the higher reduction potential the more energy is released
kinetic stability→ the delta G that’s available via reduction of O2 is only released if the spin restriction is overcomed typically in the form of more energy
in the singlet state electrons are paired where spin=0 in doublet state 2/3 electrons are paired so spin number of ½ triplet state electrons paired in high energy spin state 1
delta S=) reaction is spin allowed otherwise it’s spin forbidden
ways to render O with spin allowed
3O2+1X→ 3XO where |delta S|=1-(1-0)→spin allowed but a lot of energy is needed
but needs intermediate in triplet state needs to be formed
radical pathways
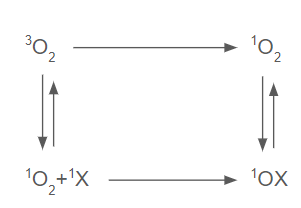
ROS are short lived so how are they studied: conversion of short lived oxygen species allows for the use of EST spectroscopy
like DMPO: whose radical species have a half life of 2×109 micro seconds
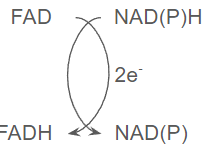
NAD/ NADH transer 2 electrons while FAD and FADH can transfer 2 or 1 electrons at a time
but using NAD is a stronger reducing agent so it’s easier to use for some reactions
FADH does electron transfers one at a time
FAD is flat but FADH is bent cause the ring is not longer aromatic
Typically NAD is coupled with FAD in reaction:
H2O2 detox: 2H2O2+ catalase→2H2O+O2+catalase → single reactant redox reaction
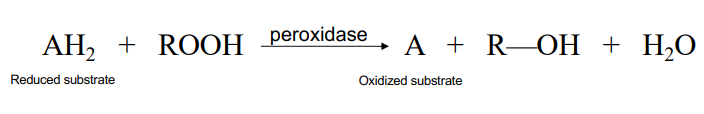
or can use peroxidase
 turn over=kcat=~109 for catalyse → is extremely efficient
turn over=kcat=~109 for catalyse → is extremely efficient
can use superoxide dismutase to get rid of radicals
Fenton chemistry and the fenton reagent (Fe(II)+H2O2)
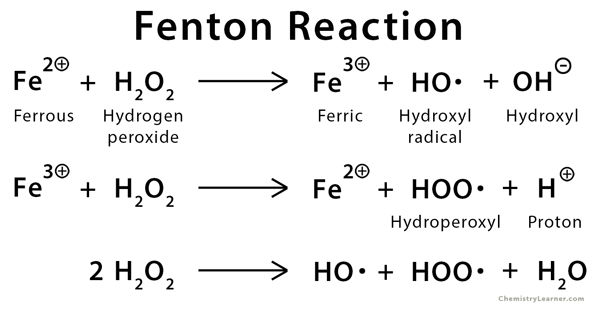
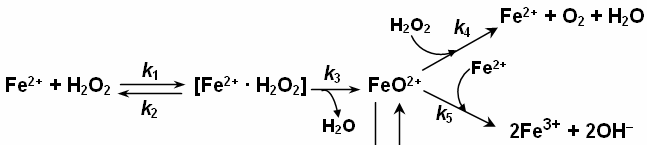
in a biological context→[H2O] in cells is ~0.5mM depending on state of cell with the half life of 0.6s for superoxide the half life is about 20s
peroxynitrite is an alternative explanation that makes more sense as an N radical is more stable than O radical
also the NOO→NOOH is more reasonable pka of this conversion is about 7 and homolysis won’t occur (NO radical and OH radical)
Radical mediated reactions →R. +H2C=CH2 →RCH-CH. where H abstraction can damage lipids or DNA
radical quenched when R+Y radicals come together
Exogenous sources of free radicals: Radiation, chemical reactions that form peroxidation or promote the formation of superoxide, metabolite radicals
UV is a well established radical trigger where HOOH→2HO.
chemicals that led to formation of peroxide:
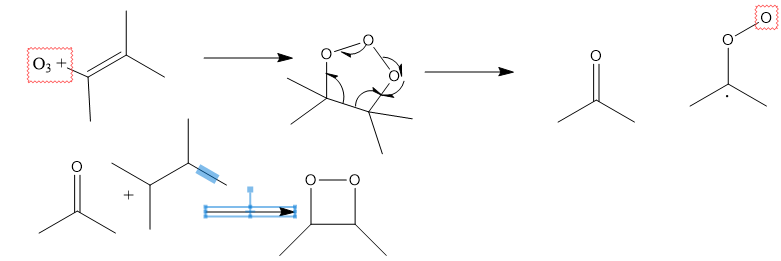
persulfate→ leads to radicals and is used in crystallization in gel
chemicals that promote superoxide formation
class 1: redox cycling reagents undergo 1 electron enzamatic reduction typically using FAD which generates transient radical in turn converts O2 to superoxide
think paraquat
quinones→ highly redox active and are Micheal acceptors which do 2 electron transfer or 1 electron processes
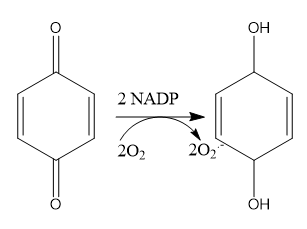 or flavin used to get O- and O. on the quinone
or flavin used to get O- and O. on the quinonemetabolize radicals→ think poyl hologenated alkenes like CCl4 after p450 →Cl+Cl3C. where Cl-C bonds are really easy to cleave
phenols and aminophenols → applies to tynonel where acetanilide were used initially but then was found to be a nephrotoxin because it made
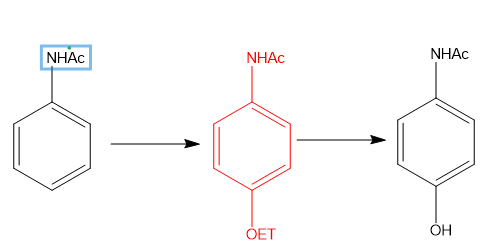 where the core structure is the 1st one without Ac which is unstable and makes quinones
where the core structure is the 1st one without Ac which is unstable and makes quinones
GHS (glutathione) is a radical scavenger it is a tripeptide
good nucleophile that is not broken down by the cell easily cause one of peptides is not in the normal orientation
it can trap electrophiles and radicals
respiratory burst→ defense mechanism that deactivates and kills
toxicity of ROS can used as a defense in the immune system from invasion and aid in engulfing phagocytes which start producing ROS in high concentration
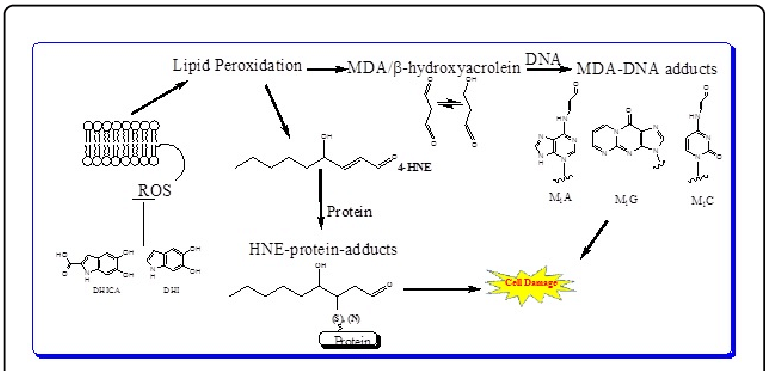
oxidation of lipids→ can led to shorting of fatty acid chains which leads to comprising cell membrane
lipids are used to make bilayers →generally hydrophobic with polar functional groups to allow the formation is and bilayer think FA, glycerol ester, cholesterols and sphingolipids
most interested in poly unsaturated fatty acids or PUFA ie allochronic acid which are used to make horomones, build muscles,
double bonds are almost always in trans conformation with the expection of retinoic acid
PUFA made :
 H are taken at the same face
H are taken at the same facePUFA needs a definitive temperature to change induce a physical (phase) change from ordered to disorder
want to keep this high so membrane is not compromised
most rigid→ FA saturated> trans unsaturated> cis unsaturated
lipid rancidity comes from the hydrolysis gives free fatty acid it’s aroma where as oxidative rancidity generates aldehydes and ketones
incitation of peroxidation: LH+X. →L. +HX so L. is now able to react with O2 as they are both in the triplet state generating COO- so L. +LOOH→LOO. +LH
termination→ 2LOO. →LOOL (radicals react together) or L.+L. →LL
stability of radicals: allyic carbon>benzylic>tertiary> secondary> primary> methyl> vinyl> phenyl
methyl radicals are sp3 hybridized; vinyl and phenol are sp2 and alkyl are sp
the less S character in orbital means the further away from the nucleus and lower electron affinity of half filled orbital making is more stable
generated radicals with PUFA: trans radicals are the most stable so H abstraction will happen there
enol radicals can be formed with O2 creating R-O-O.
what happens next: two radicals combine and are homolyticly cleaved to give alpha, beta unsaturated ketones/ aldehyes and O2
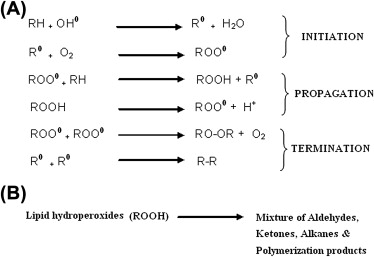
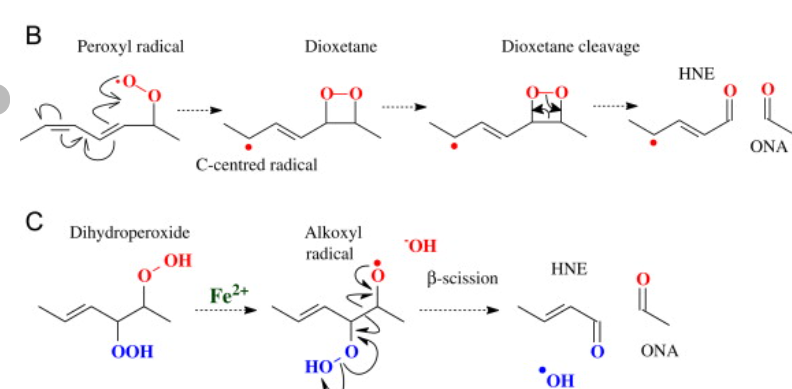
termination products can generate Micheal acceptors
if termination compounds are to accumulate can lead to bad things like DNA damage ie:
antioxidants→ substances at low concentrations compete with other oxidizable substances which delays the oxidation of them think vitamin E (TOH), BHT
chain breaking antioxidants→ donor antioxidant so no more radicals are make like LOO. +TOH→ LOOH+TO. which is stable
sacrificial antioxidant→ LOO. +NO→ LOONO which terminates the radical
good antioxidants should be unreactive in both radical and unradical form ie they decay into relatively harmless things and don’t reaction with O2 in radical form these should also be able to be recycled
catalytic reduction of lipid radicals:
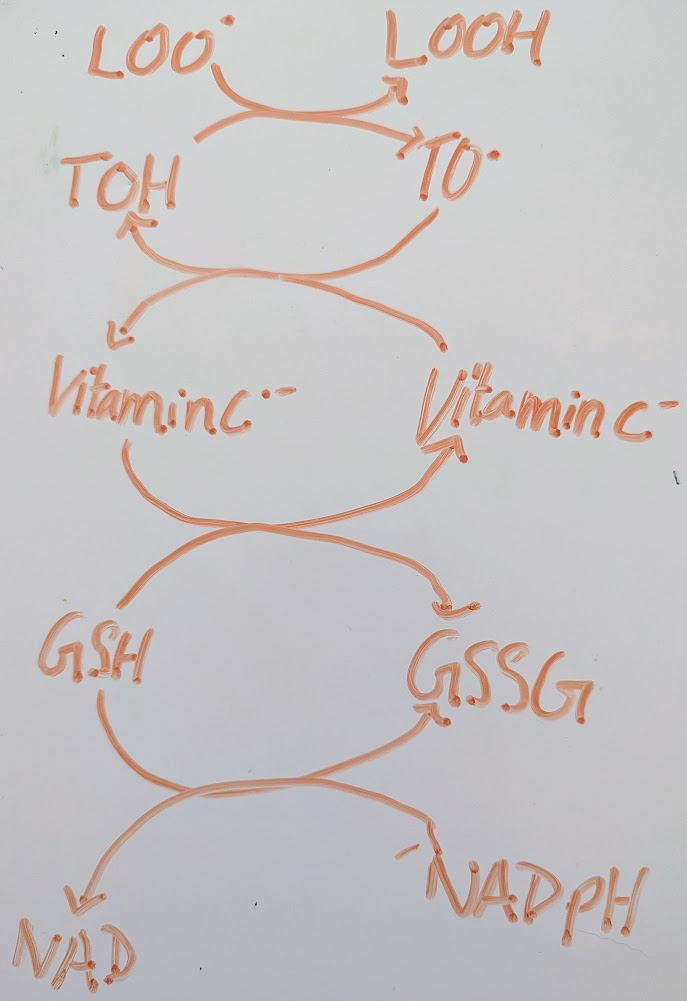 vitamin C is capable of electron transfer but only 1 electron at a time
vitamin C is capable of electron transfer but only 1 electron at a timevitamin E takes in the cell membrane so it can catch the radical
Phase 1 metabolism
metabolism of toxicant doesn’t always lead to detoxification
recall cycasin which is toxic when metabolized by gut microbes
cycasin→ B-glucoside digested by B-glucosidase and makes glucose and HO-C-N=N- radical which is toxic
Richard Williams→ 1st one to purpose phase 1 and phase 2 metabolism
in book he introduced phase 1 and 2 metabolism and the fact that metabolism doesn’t mean detoxification
he introduced borneol to urine and ran an analysis and was able to find the different metabolites
phase 1 metabolism→ polar reactive group is introduced to the molecule think redox, hydroxylation
phase 2 metabolism→ conjugation reactions so that solubility increases and becomes more likely to be excreted
for example benzene is not a toxic until phase 1:
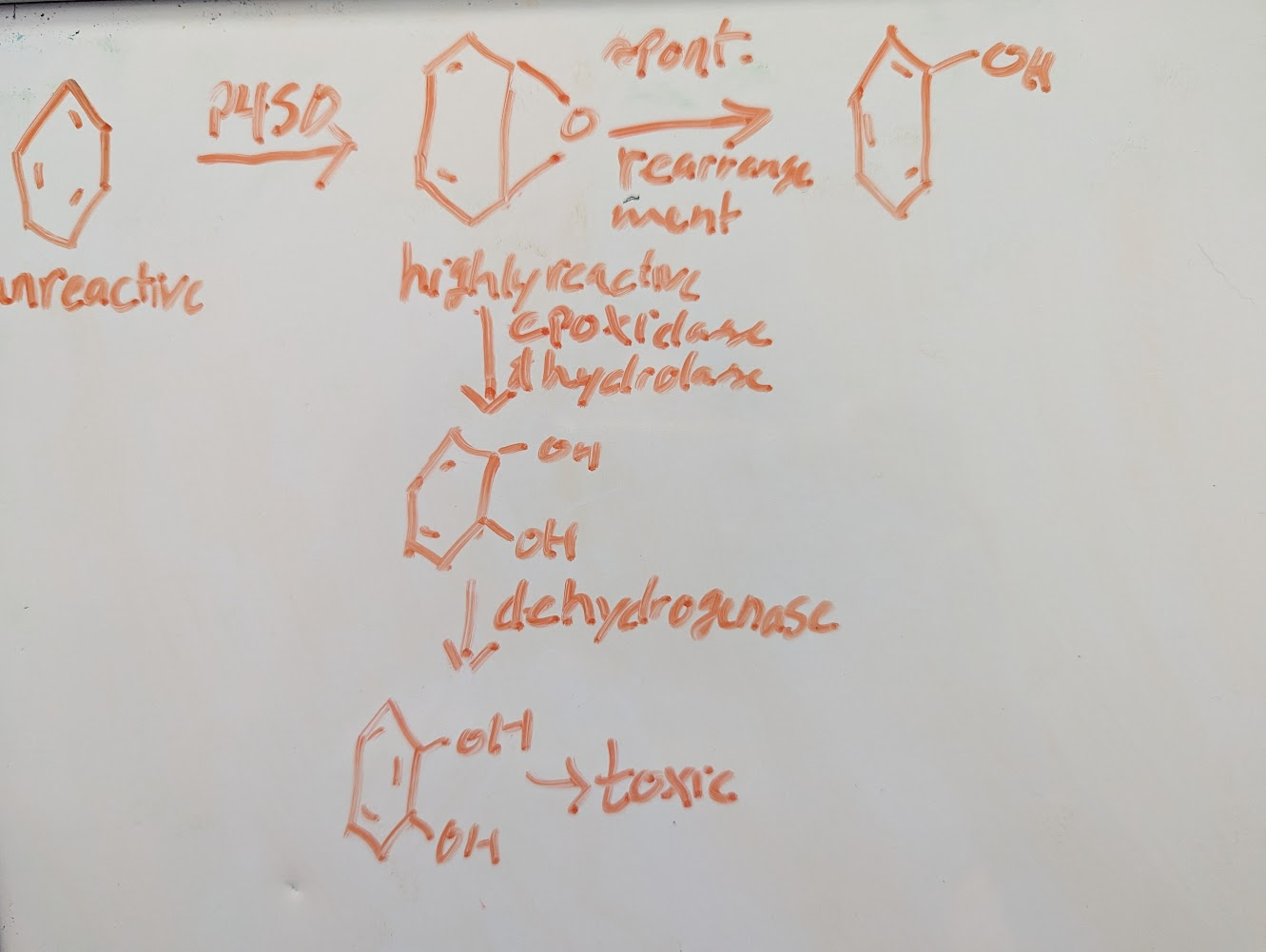
Bernard Brodie →1st insight on how it works:
NADPH+H+RH+O2→ROH+NADP+water
liver microsomes are used get this done
cytochromes→ proteins that have heme which is a 1 electron carrier because of the heme group
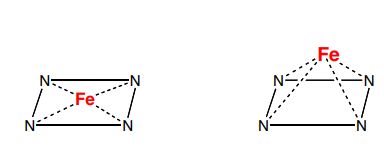 1st is resting state of heme 2nd is active state keep in mind that Fe has two other ligands in 1st on and 1 ligand in 2nd on
1st is resting state of heme 2nd is active state keep in mind that Fe has two other ligands in 1st on and 1 ligand in 2nd onliver microsomes has all phases of metabolites including cytochrome p450
P450 mechanism sample: S2O-24 → SO.- Fe3+ →SO.- Fe2+
p450s can be followed using fluoresces and are good for enzyme assays
p450 monooxygenase adds O to substrate: RH+O2+NADPH→ROH+NAD+ water
Fe does the work: crystal field model shows the breaking of degeneracy of electron orbital with the D and F orbitals due to a static electric field
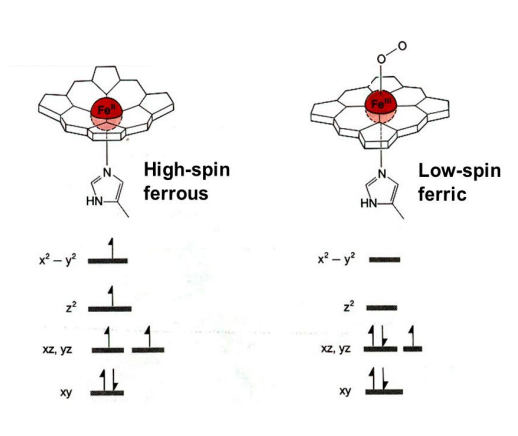
when spin state is higher it’s easier to reduce and binding will reduce the heme
P450 reductase →complex located on ER where NADH transfers 2 electrons to FAD which transfers to FMN and then p450 cause of redox potentials

Hydroxylation of P450 catalytic cycle
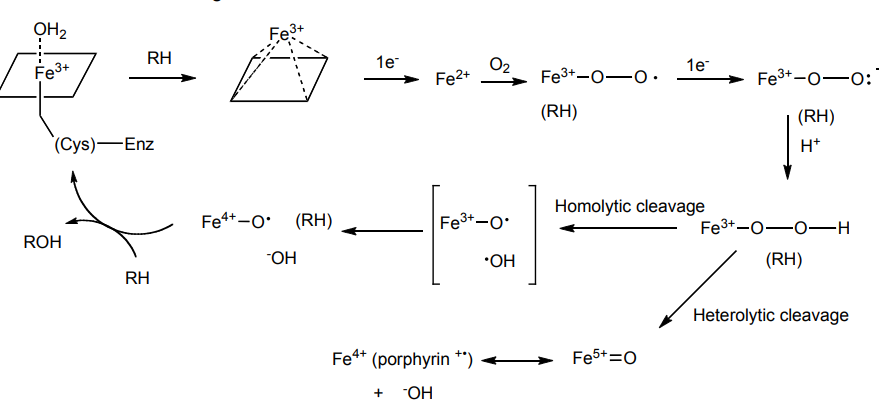
1st step→ low spin→ high spin cause of binding makes reduction easier→ electron transfer chain →generate radical neutral species using acid/ base chemistry → O is radical species cause of Fe electron transfer in res. →oxygen rebound to get products
P450s oxidize catalyse epoxidation, hydroxylation, heteroatom oxidation
2 considerations of p450 enzymes → geometry of ES complex and reaction is radical process so relative reactivity of the accessible position of the substrate
keep in mind most stable radicals cause that’s were it’s most likely to occur
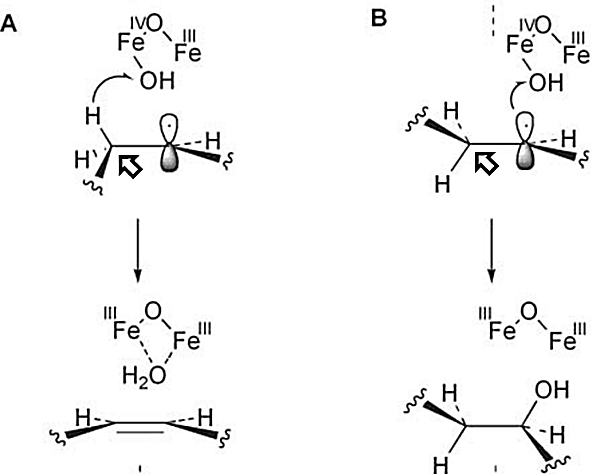
fatty acid hydroxylase looks at terminal opposite to acid W so hydroxylation
P450 can produce toxic species→ parthion→p450 add replaces O with S →paraoxin
W/ W-1 hydroxylation of ibuprofen (advil)→
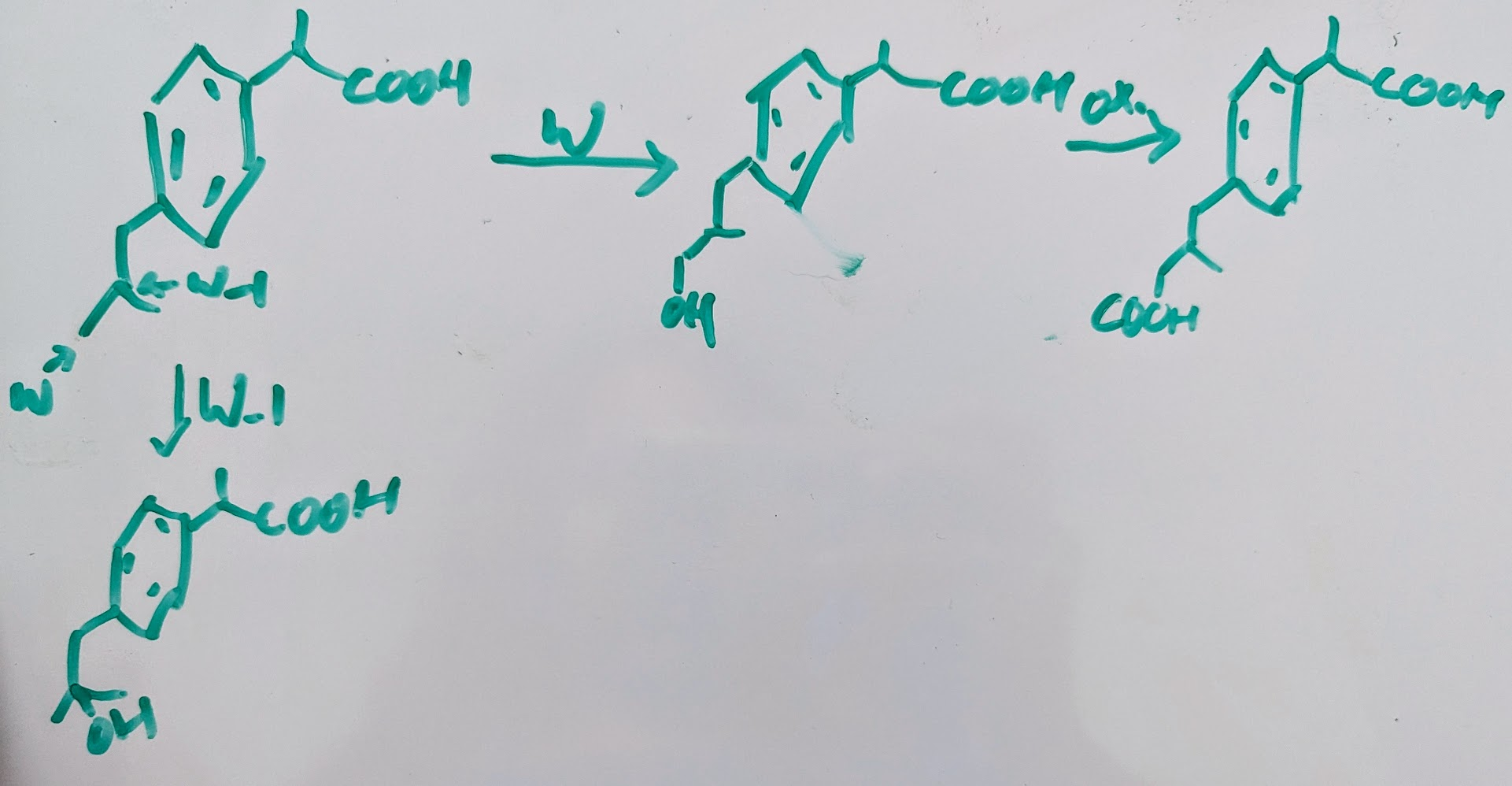
old root beer flavor came from root of sessfras →after metabolism compounds became carcinogenetic where p450 would hydrolyze a bond then sulfatransferase would add SO to that OH making it good LG that reacts with DNA
oxidation of heteroatom→
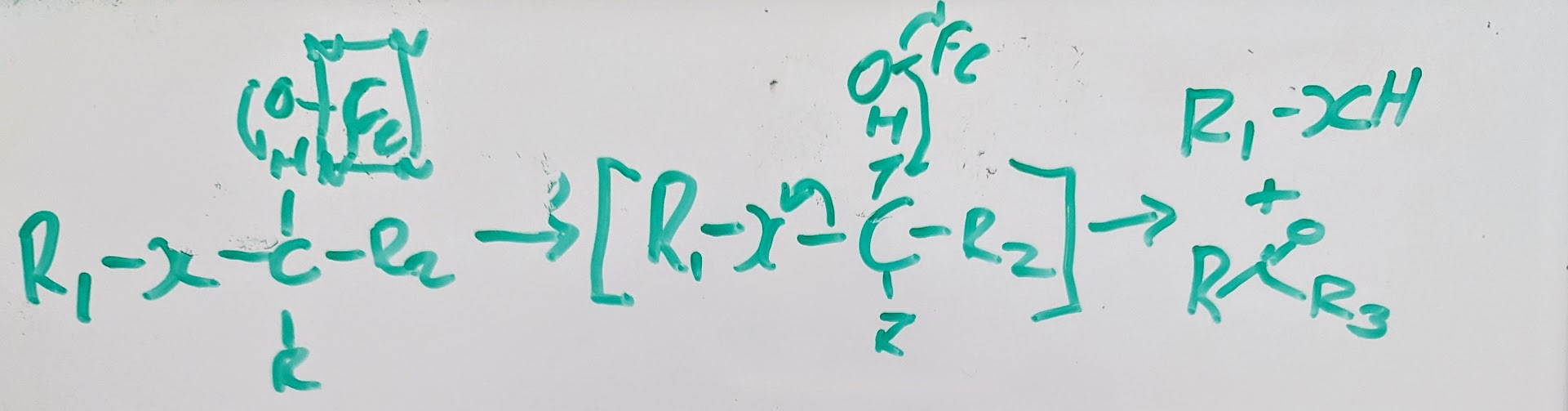
CN systems do a similar thing but for primary amines you’ll get aldehyde and NH3
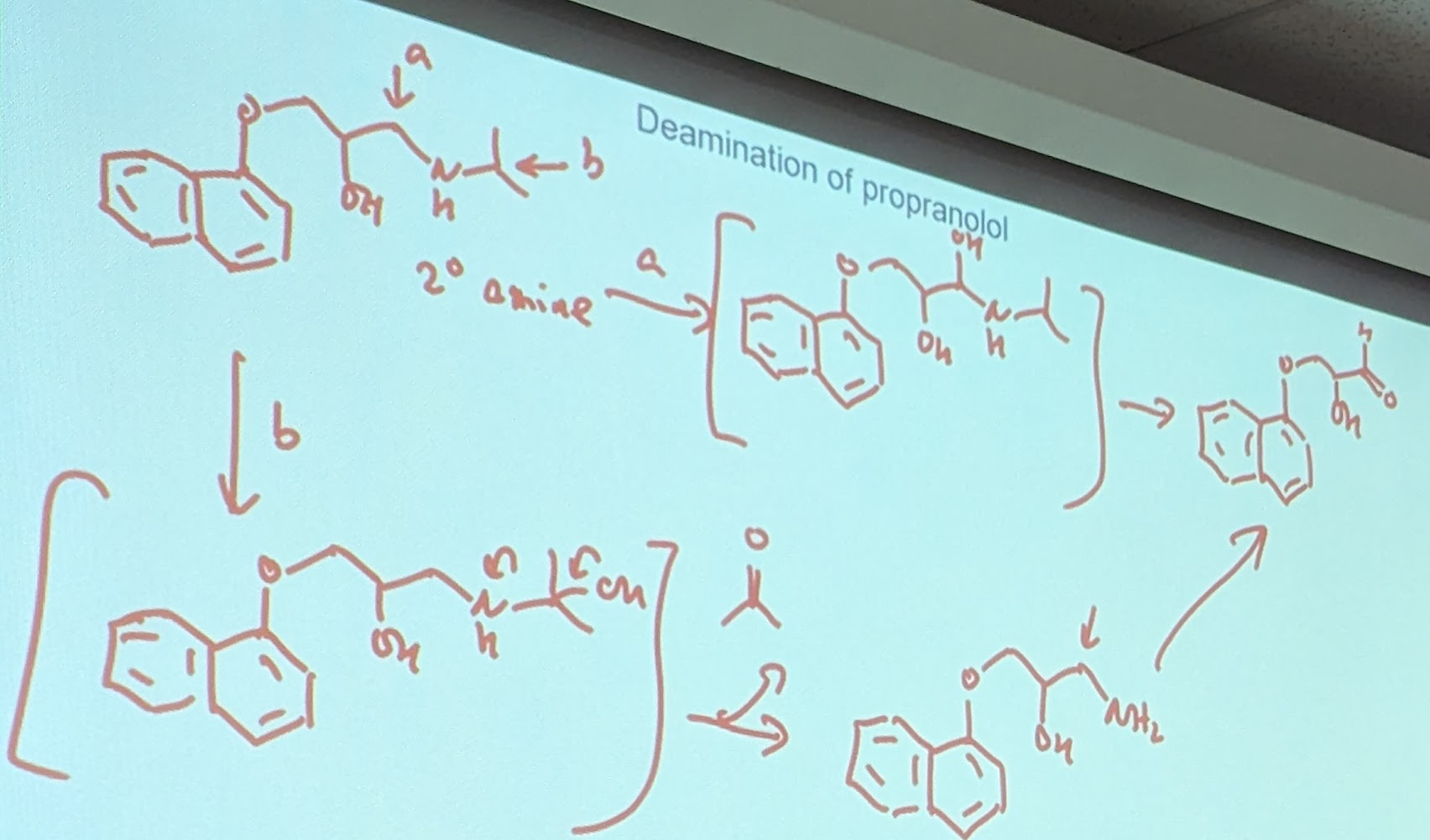
Propanol example is a secondary amine
oxidation of N→ typically happens if nothing can happen
primary amine →R-C-NH2→RCNOH→RCNO→RCNO2
secondary amine: give you R-CN(OH)R’→RCN(-O)-CH2
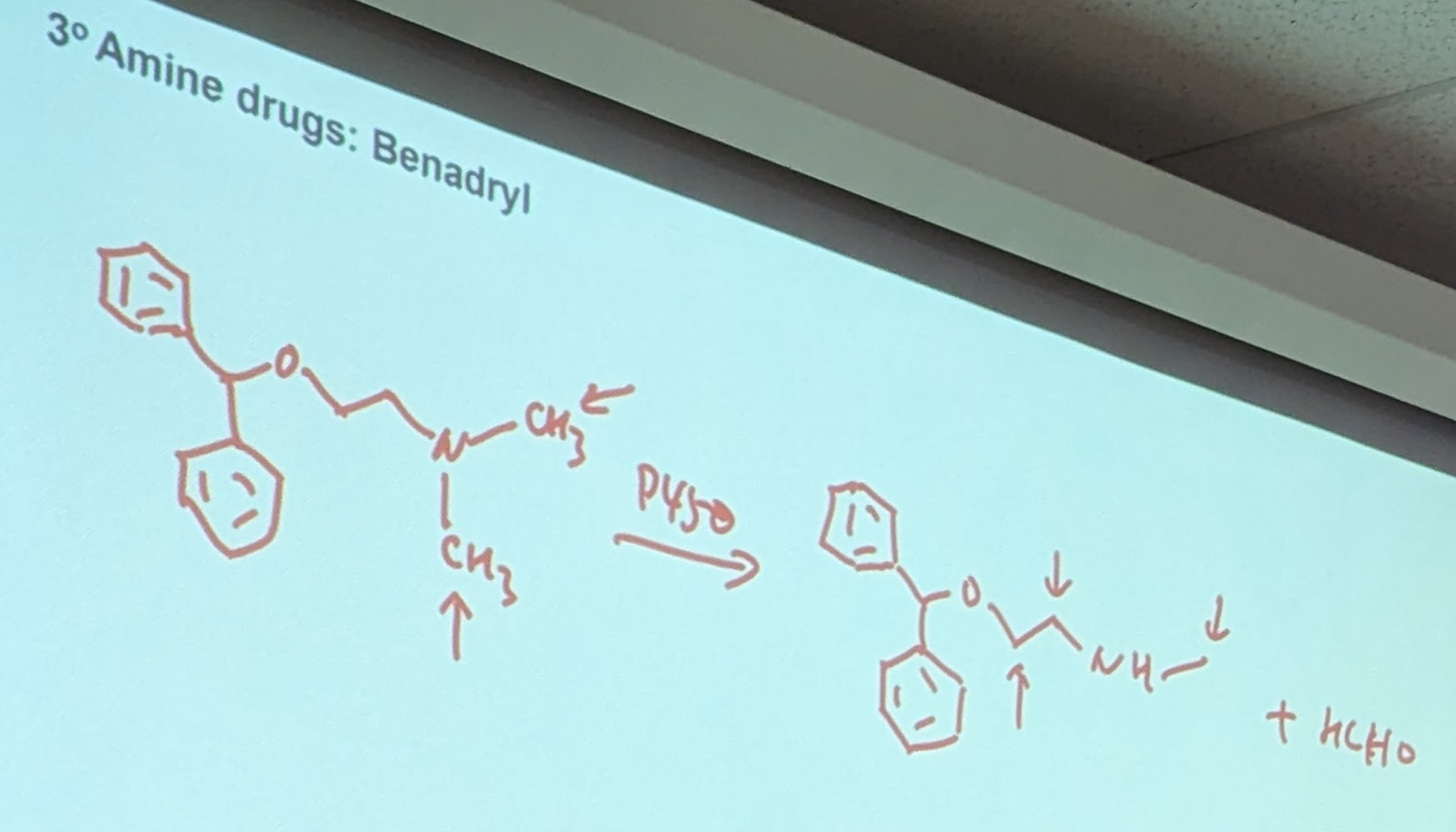
tertiary amine→ RCN(R,R)-O-
amantadine is hard to metabolize cause it’s a primary amine which isn’t easily oxidized
phenacatin →
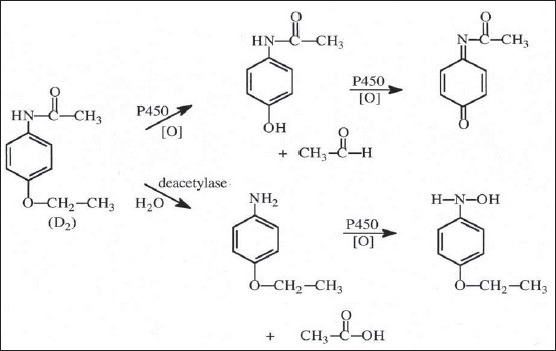
CCl4 carcinogen is different than chloroform as a carcinogen chloroform is less carcinogenetic CCl4 will produce phosgene which is really toxic and release Cl
phenytoxin and toxicity→ found to fetal hydantoin syndrome→ when used it inhibits epoxide hydrolase can decrease side affects
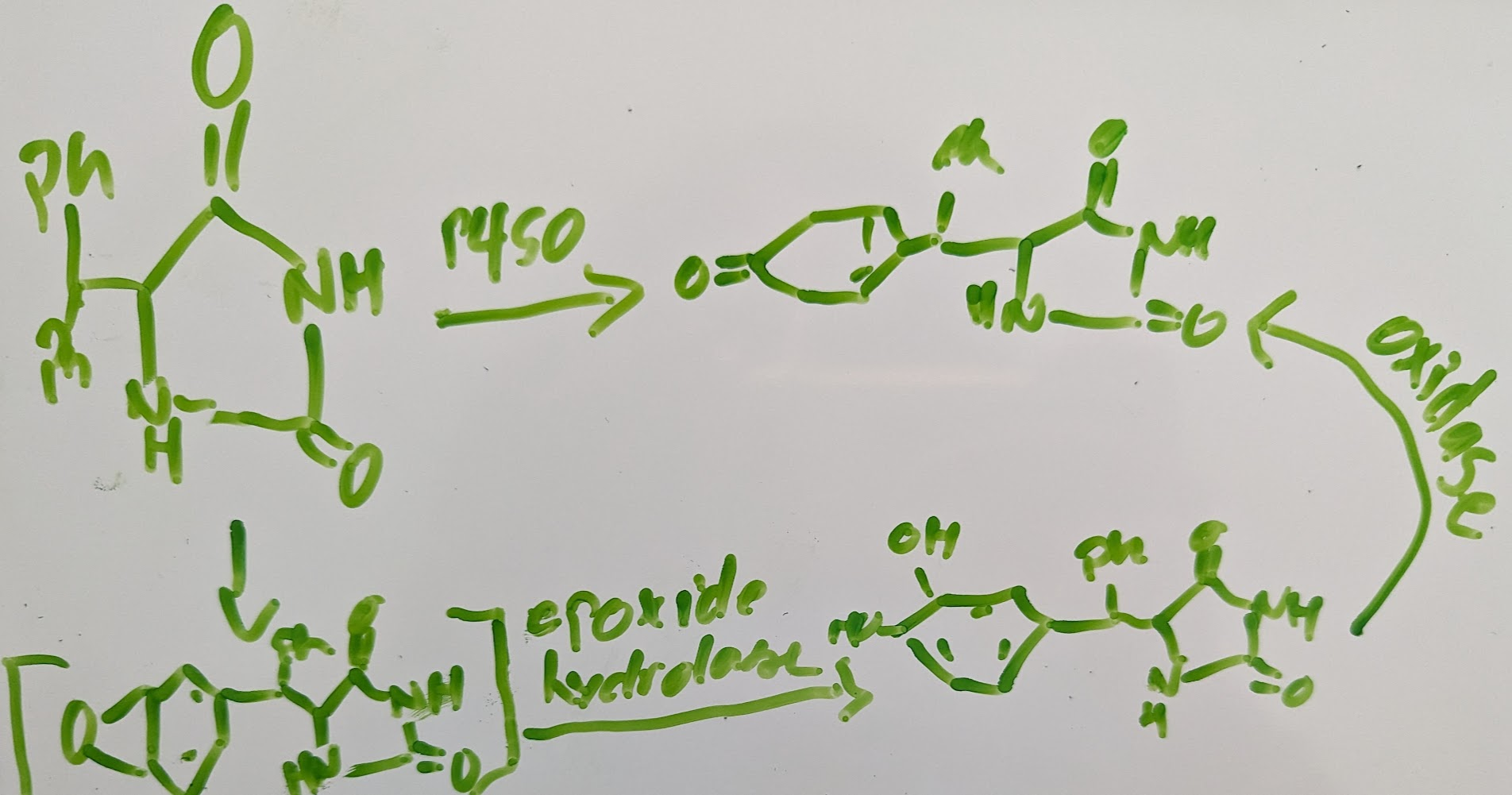
later shown phytoin was bind to potassium ion channels is also through for it to be toxic
codeine→ painkiller shares co structure with morphine
metabolism C-O→ hemiacetal which degrades spontaneous to make morphine
fentanyl can easily cross the BBB binding constant Kd to the u-sprod receptor of 400pm
carfenanil Kd>20pM compared to codeine Kd of 700nM
oxidation of C-S system:
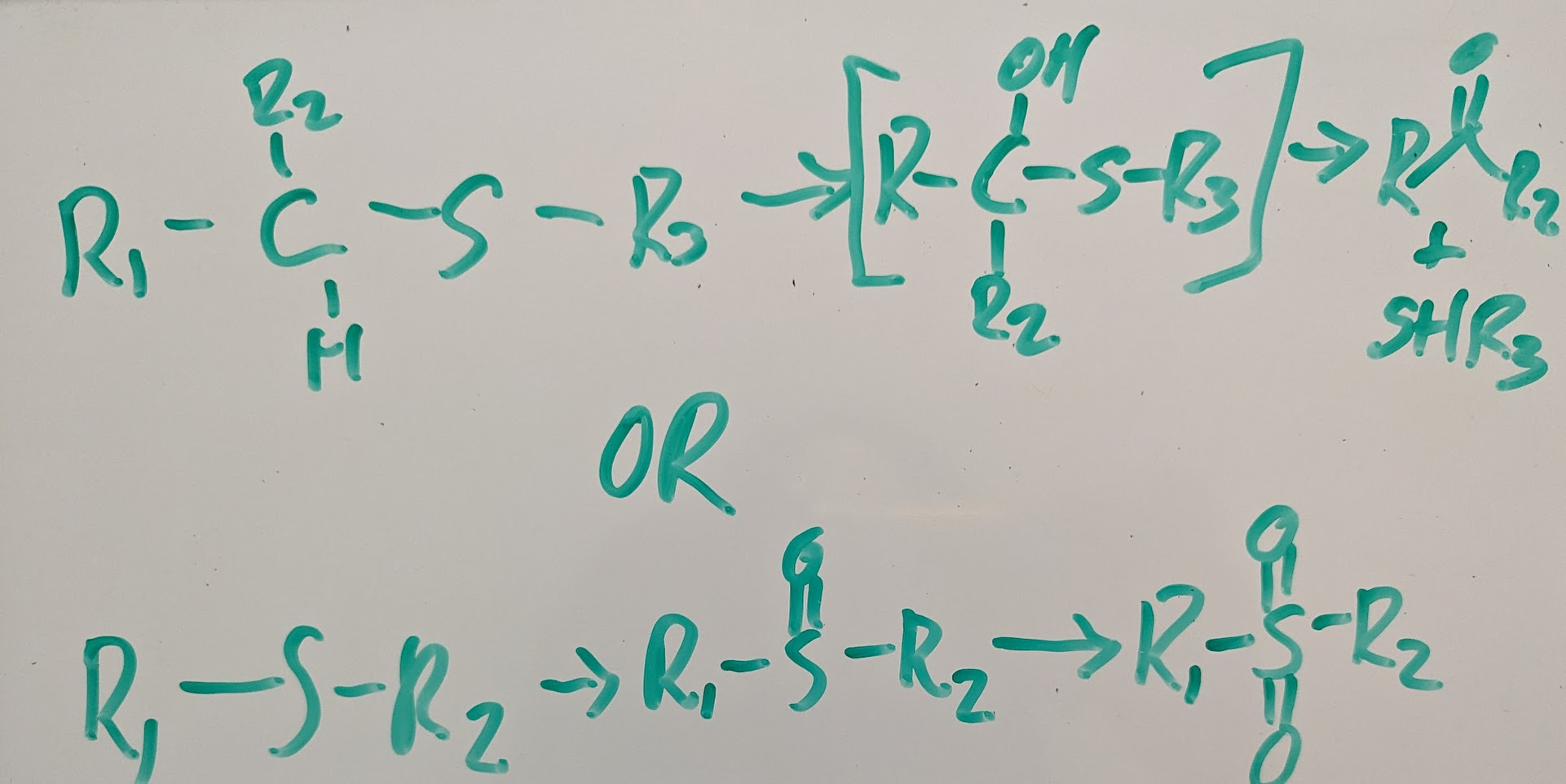
in desulfuration turn RCR=S→ aldehyde
aromatic hydroxylation produces arene which are impossible to isolate cause they so reactive as follows:
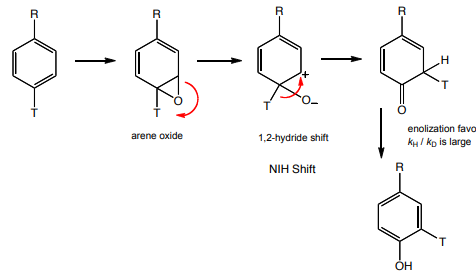
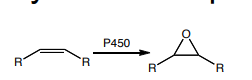
Making epoxide using p450:
when something is electron deficient its hard to put OH there
Drug -Drug interactions→ can induce increase or decrease activity of P450
works on a case by case basis
there have been multiple drugs pulled from the market do to these interactions
p450 inhibition→ used to probe enzymatic mechanism and cancer-preventive agent
ex: celeboxib
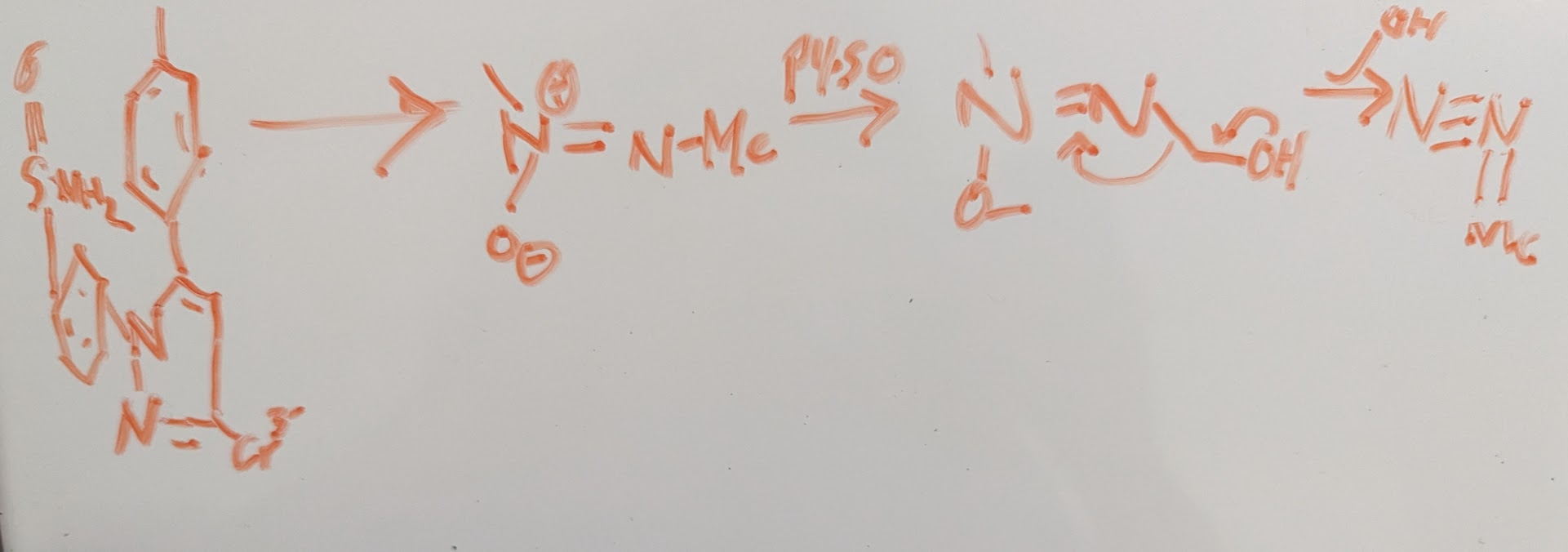 the final product is a dangerous electrophile
the final product is a dangerous electrophile
non sedative antihistamine →seldane (marketed so that you don’t get drowsy) but can cause death when interacting with ketacanazole which is an antifungal and antidandruff agent
ketacanazole inhibits Cyp3A4 so concentration of tertenadine and Na chancels where blocked causing heart attacks
inhibition examples →kaletric cocktail of lopinavir and ritonavir where Lopinavir and ritonavir are HIV protease inhibitors but all inhibitors enzymes that work hydrolyzes lopinavir (p450)→ cyp3A4
induction example: ie the alternative ethanol metabolism
cyp2E1 is induced by more alcohol drinking
cabamazepine and valproic acid:
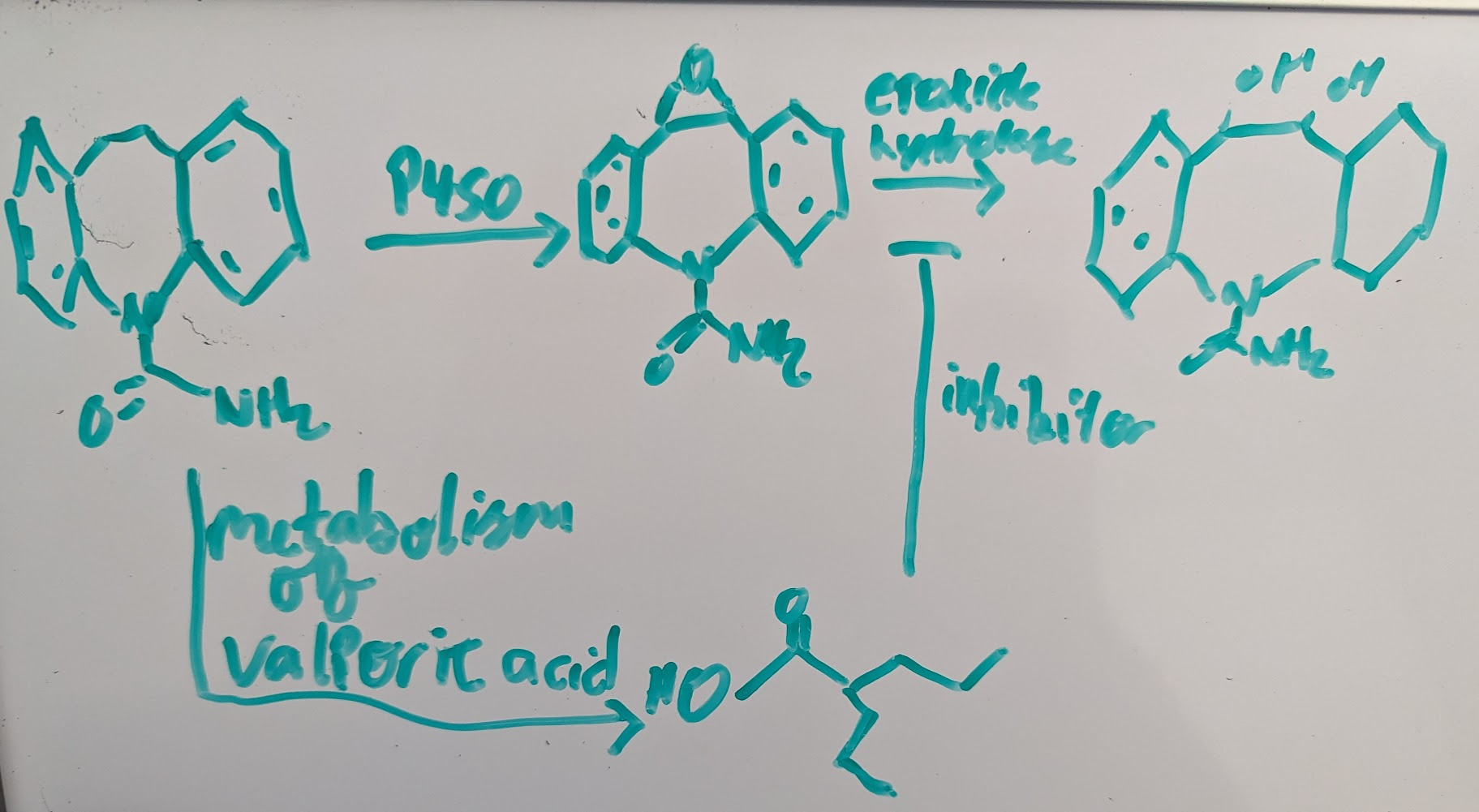
consequences of metabolism→ formation of chemically stable metabolites leading to detoxification, lack pharma logical/ toxicological activities or
generation of short lived reactive metabolites leading to toxicological activation
formation of chemically stable but pharmocolgically active metabolites
Phase 2 Metabolism
formation of conjuncts →which will change physical and chemical properties of primary metabolites
usually, happens when substrate has a chemical handle otherwise it must be processed by phase 1 metabolism
addition of oxidized glucose or glucuronaniadation is ~40% metabolism then sulfotransferase~20%, N-acetyl transferase→~15%
features of phase 2 metabolism→ conjugation increases MW, pKA slightly which lead easier excretion of compounds and is dependent on availability of other substrate
if depleted will have high concentration and low excretion of metabolite
Acetaminophen metabolism:
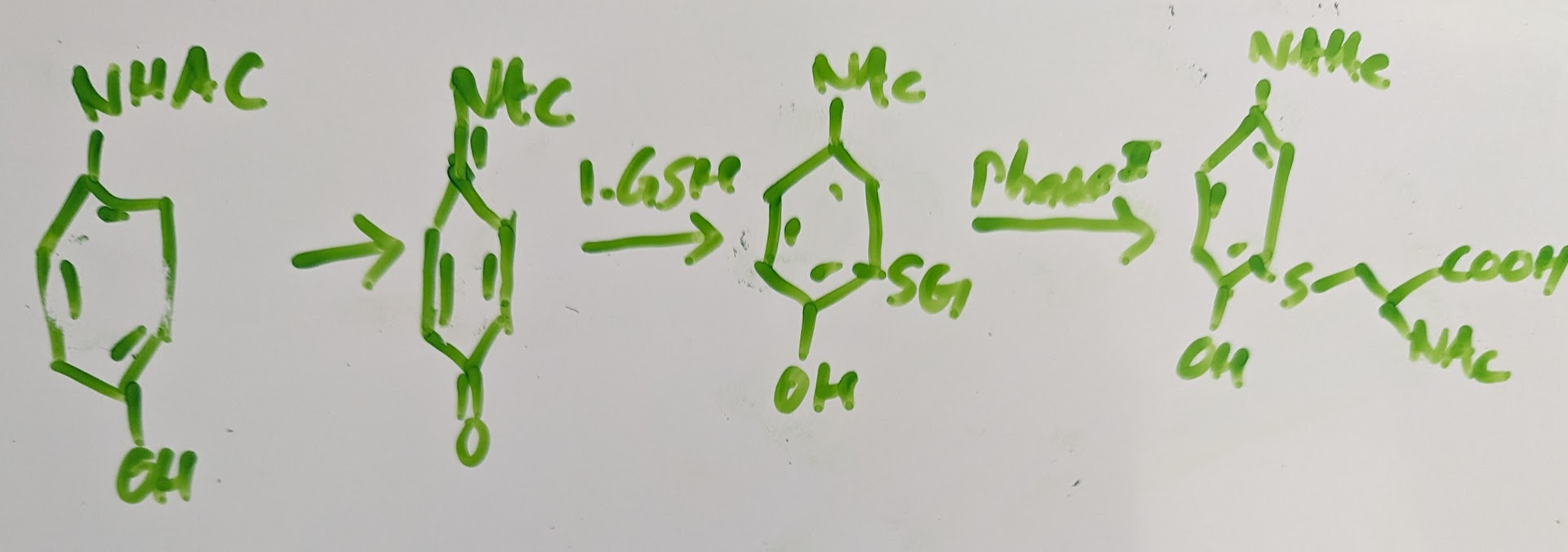 when GSH is overwhelmed it can’t deal with the acetaminophen leading OD
when GSH is overwhelmed it can’t deal with the acetaminophen leading ODOAT( organic anion transporter) used to transport things from kidney to blood
more binary excretion when MW is 300-400 cause anatomy of hepto binary system so phase 2 metabolism helps by adding MW
deconjugation is a thing→ in vivo where you remove things to make molecule smaller
acyl glucuronide → key intermediate → case of zampirae withdrawn due to toxicity caused by anaphylaxis
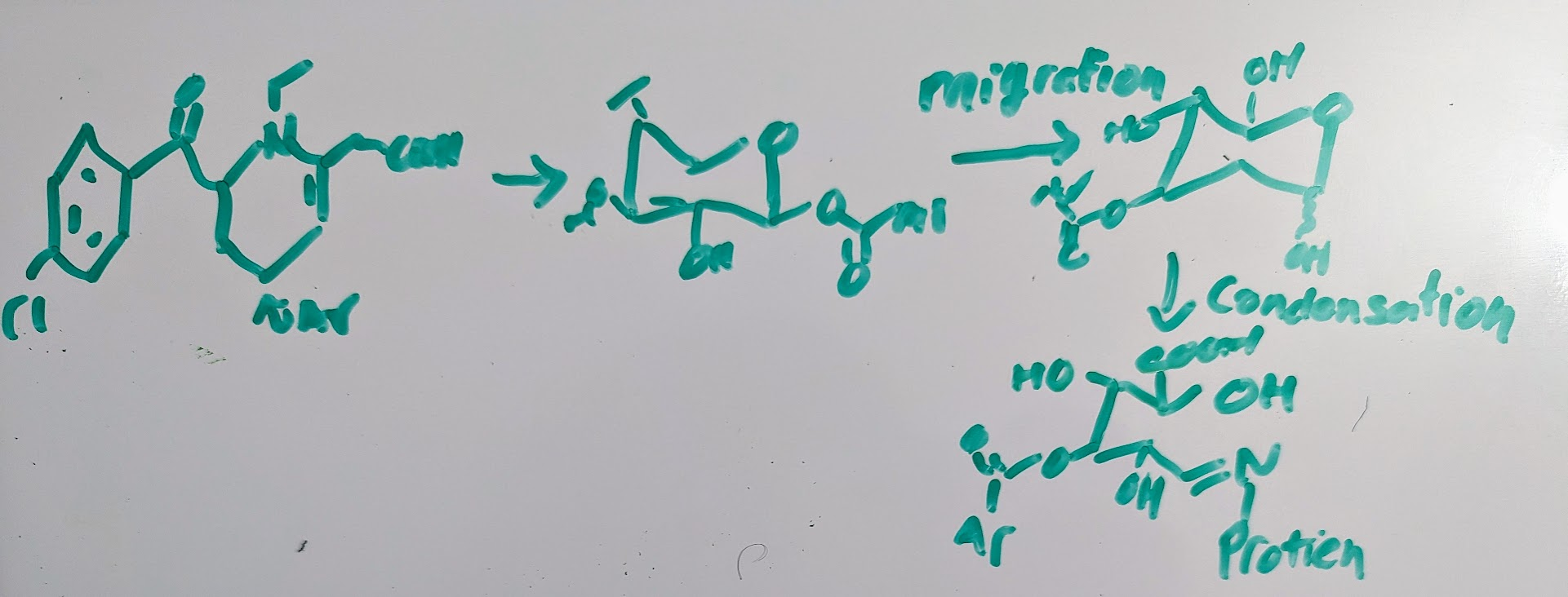
Glutathione (GSH) and Glutathione transferase→ has bond with side chain of glutamic acid and cystine making in enzyme resistant
GSH is made by the following pathway:
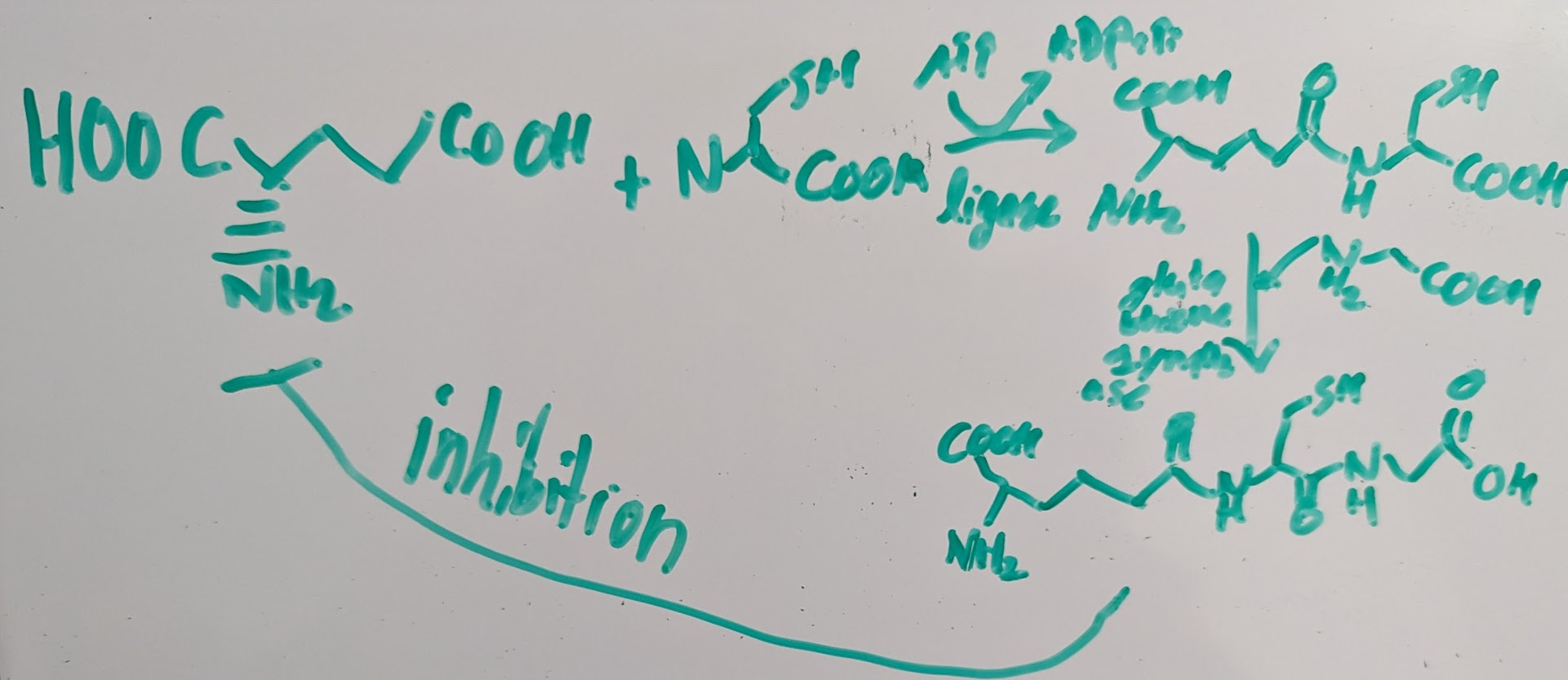
agents to mimic GSH synthesis in process will inhibit sulfate cystine ligase such as B50
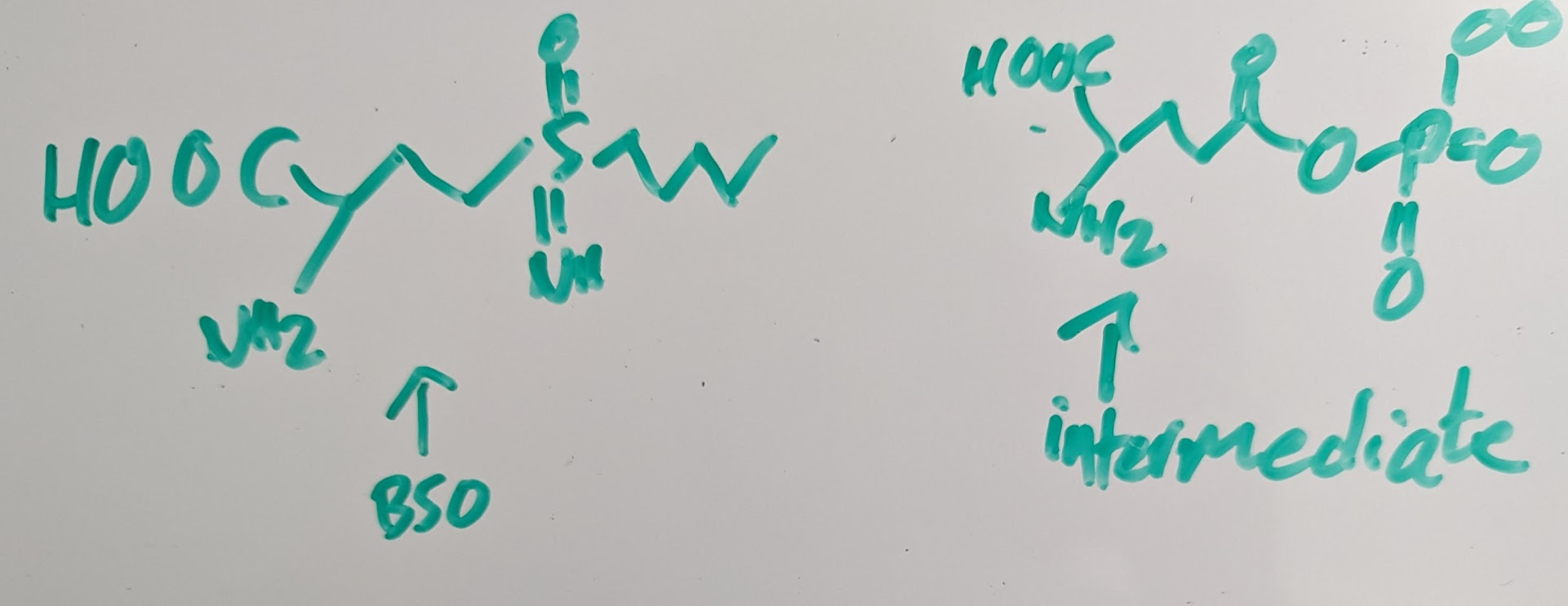
was thought that B50 inhibiting GSH depiction could be used against cancer cause normal GSH causes ROS metabolism and energy is no necessary so the cell can survive but cancer cell have a lot of ROS so they won’t survive
good idea but had a lot of side affects
GSH used as nucleophile which reacts with electrophile demanding covalent adduct: epoxide, quinone, halo alkanes, isothiocyanate (R-N=C=S) and Micheal acceptors
toxicant→ reactive metabolite (electrophile) + biomolecules _>damage to biomolecules if GSH intercepts GSX can lead to detoxification or the creation of toxin
study metabolism of aromatic compounds using Br-Ar showed that after P450 the arene oxide made is trapped by GSH where a glycine is added and converted into a cystine after which the compound leaves in urine
GSX adduct formation can be detoxification which can be enzyme catalyzed with glutathione transferase which protects against xenobiotics
detoxification of mustard agents: seen in warfare in compounds like tear gas (benzyl chloride/ bromide), phosgene and diphosgene, mustard gas, and in pesticides
ex: 1,2 dibromoethane is a pesticide that kills bugs but forms an ozone depleting agent

Styrene was spilled in English Channel sparked research in toxicology but no definitive answer on its safety ie one place says its safe others doesn’t
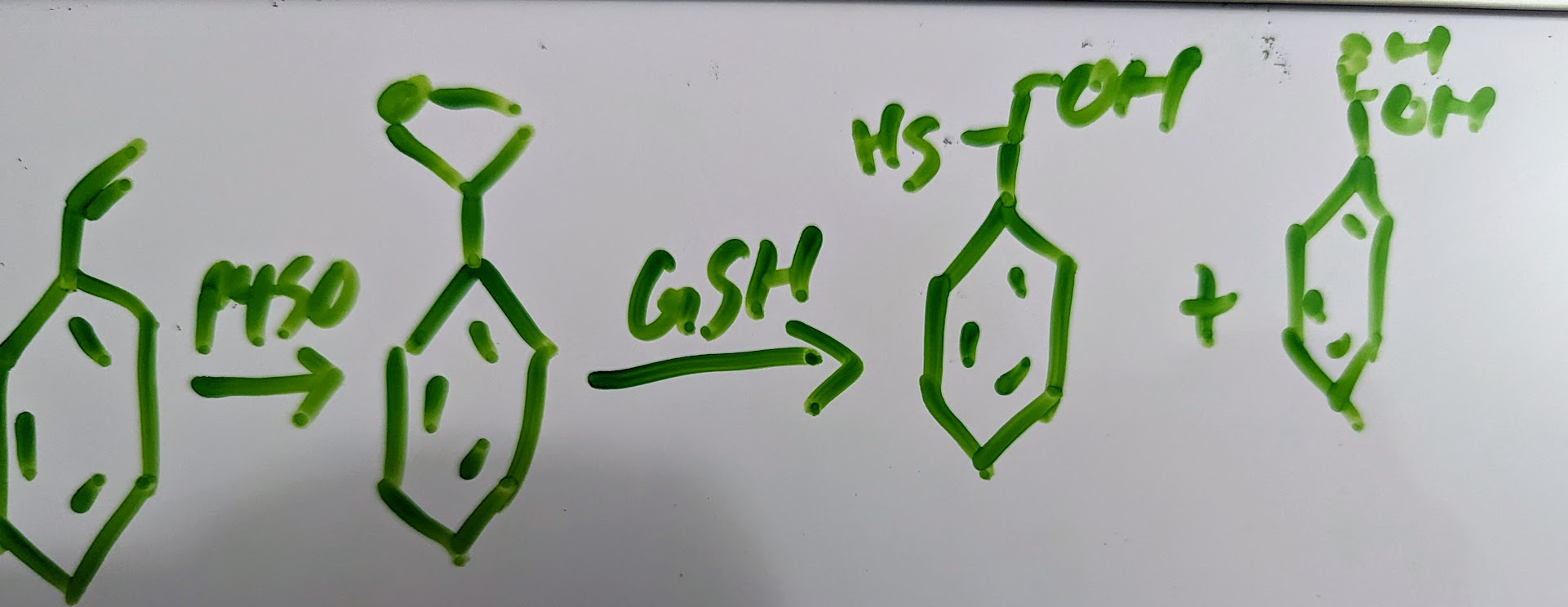
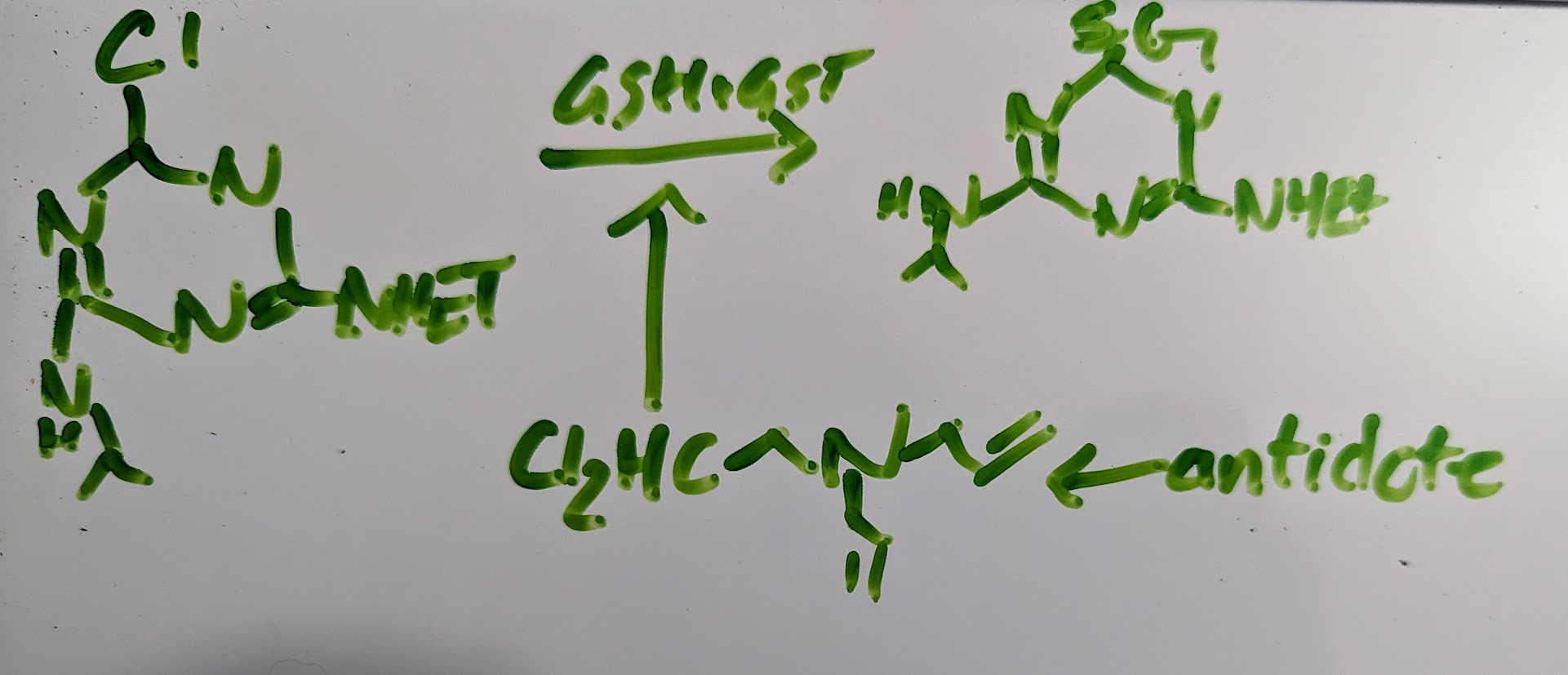
GSH and herbicide atrozin with antidote makes safer for humans as it induces GSH and glutathione transferase expression allowing it to be more selective
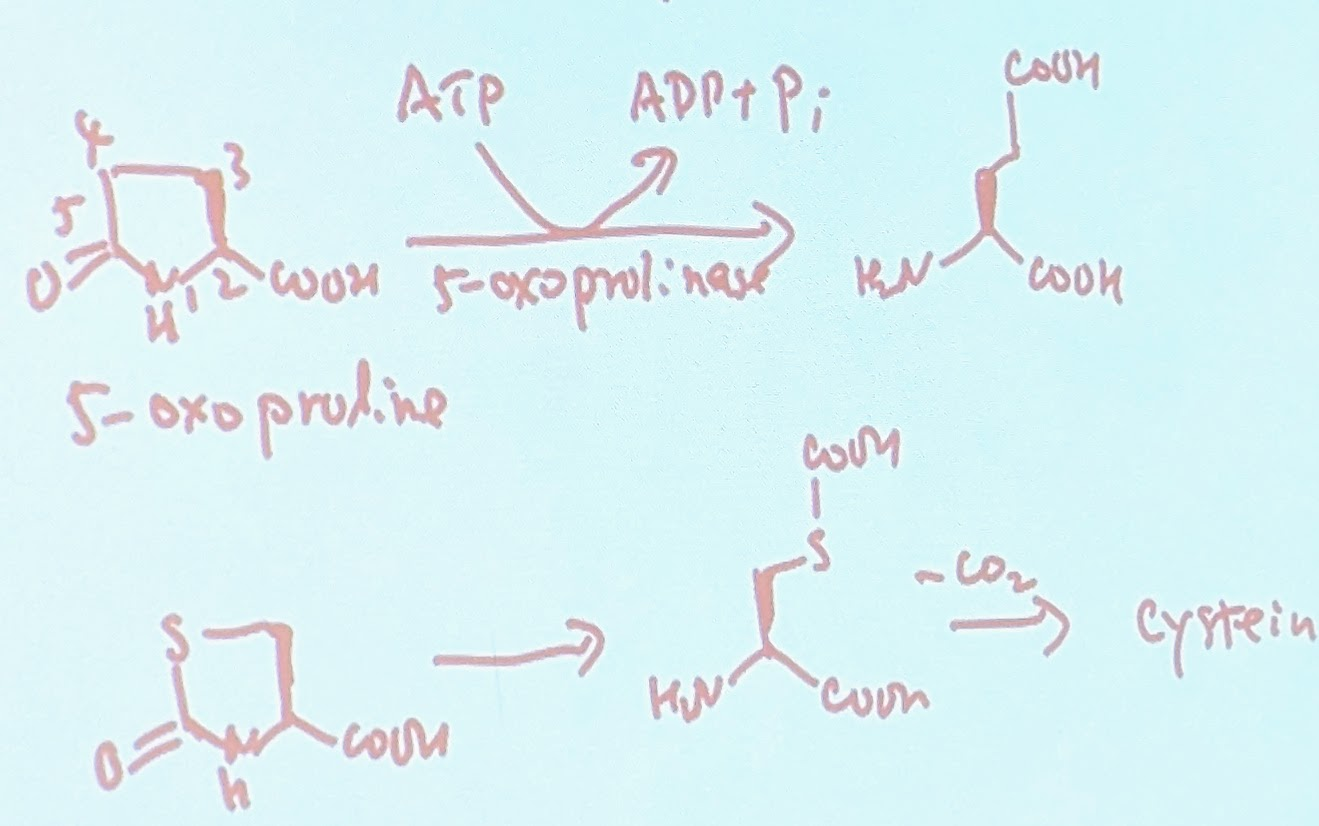
GSH is poorly bioavailable so cell provides cystine as a precursor to GSH but cystine is unstable in terms of redox and has low solubility so clinical studies use N-acetyl cysteine or oxidized proline to induce cystine production ex:
Phase 2 metabolism and Gluconidation→ accounts for 1/3 of all conjugated drug metabolism and can also do androgenesis compounds
aglycon→ non sugar component
glucuronic acid: D Beta:
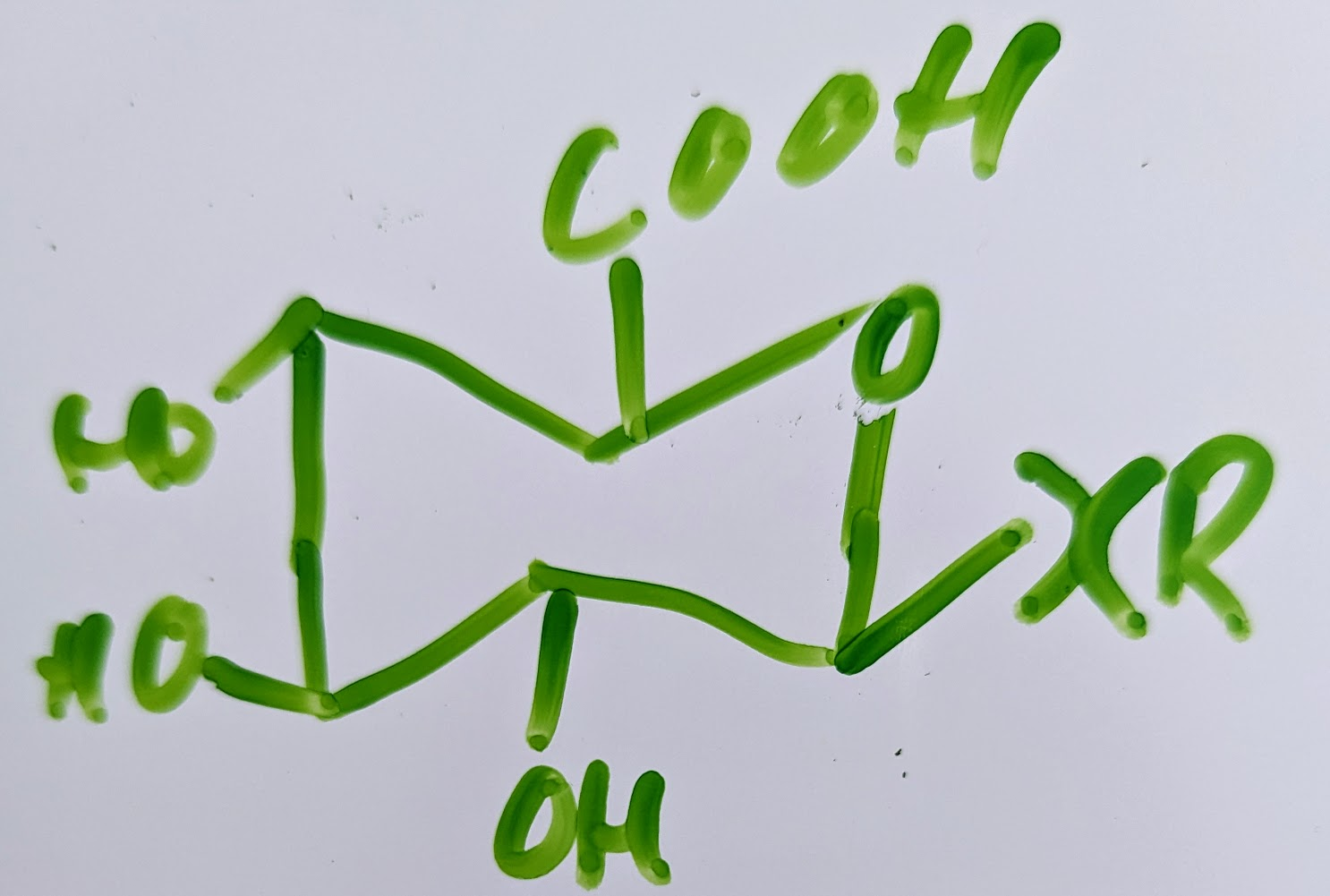 where x is O,S,N,C and R is the aglycan
where x is O,S,N,C and R is the aglycan
making UDP-gluconide
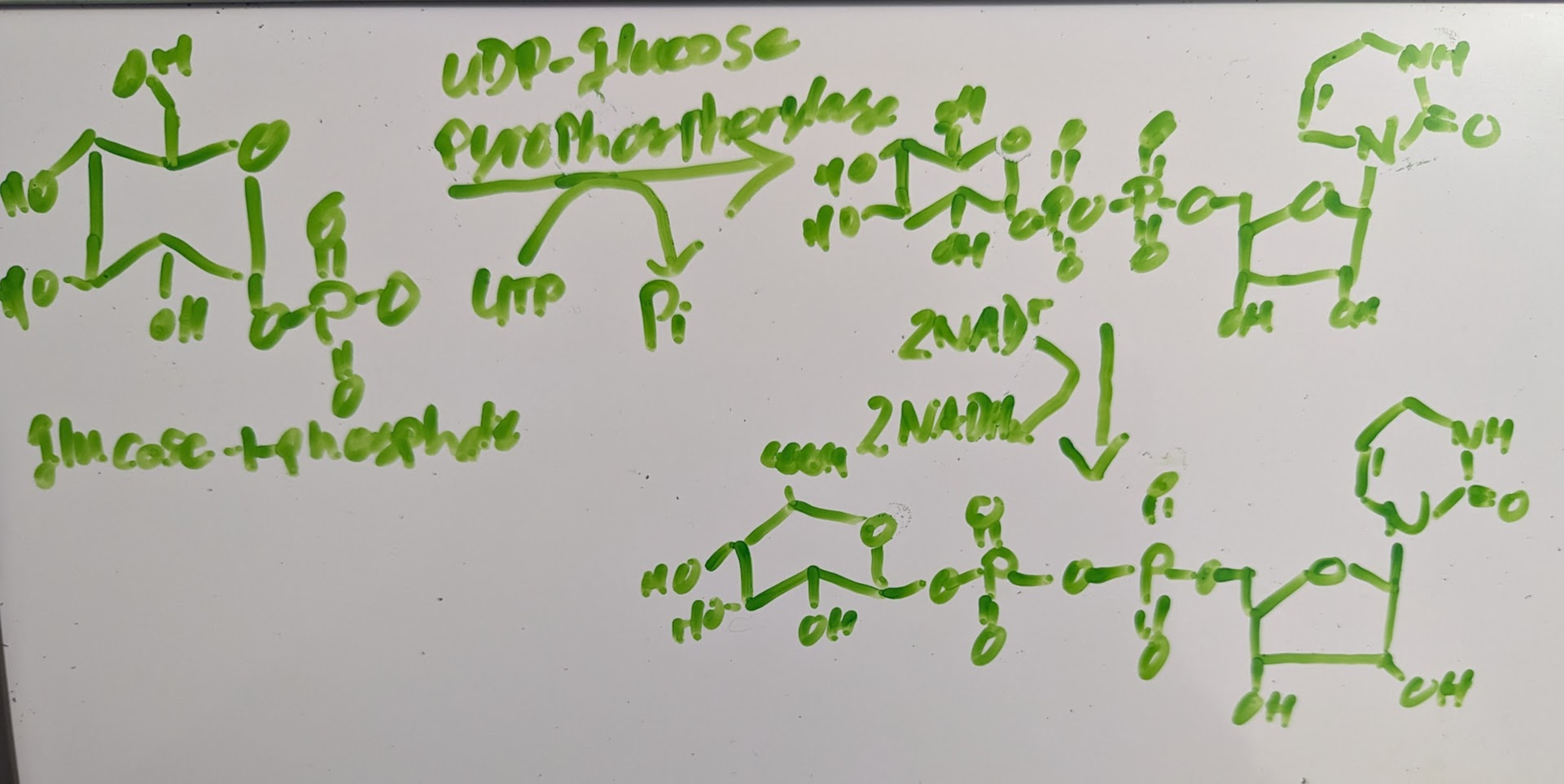
why is glucose used cause it’s able to the chemistry
GSH is a nucleophile so conjugation is between nucleophile and electrophilic xenobiotic GlcA→ nucleophile metabolite or xenobiotic
UDPGTs associated with p450:
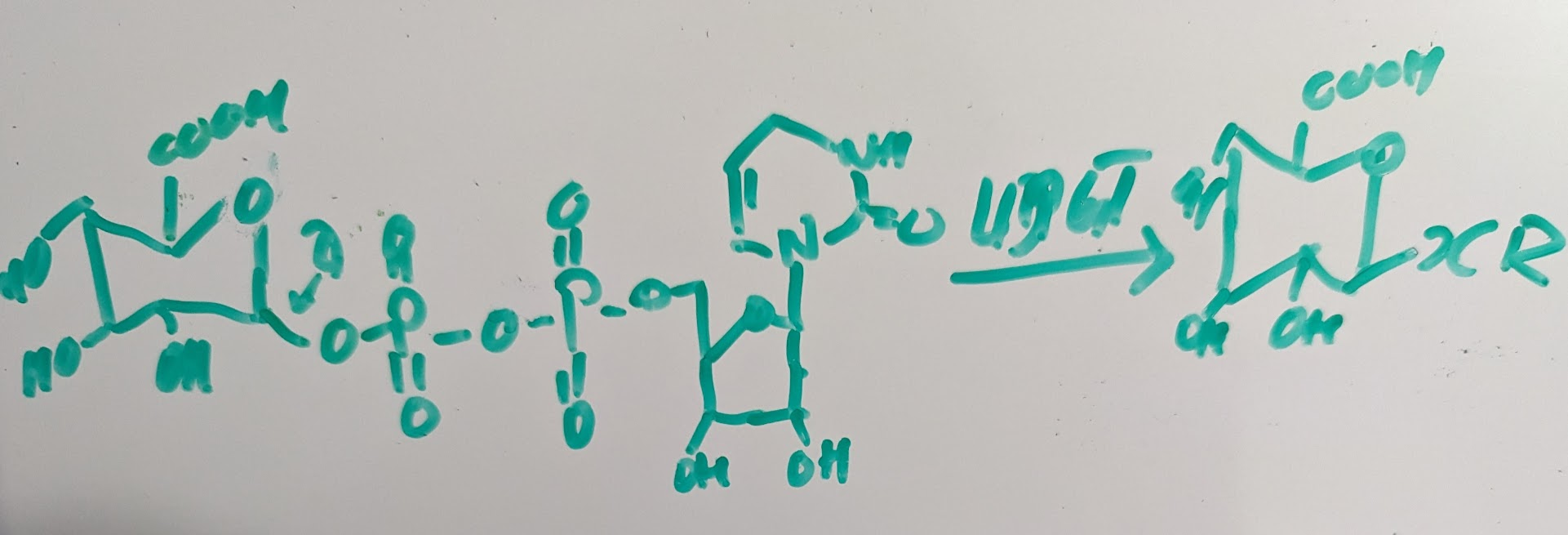 in this process X can be O,S,N, or C it’s just the thing that connects the sugar with the aglycon depending on the atom it is a different metabolite
in this process X can be O,S,N, or C it’s just the thing that connects the sugar with the aglycon depending on the atom it is a different metabolitetypically, Gluconidation is detoxification pathway but this not always the case for morphine 10% of the product will make a more active compound
the glucuronidation of heme heme occurs during red blood cell death to get rid of heme with a turnover rate of >10^6/ day
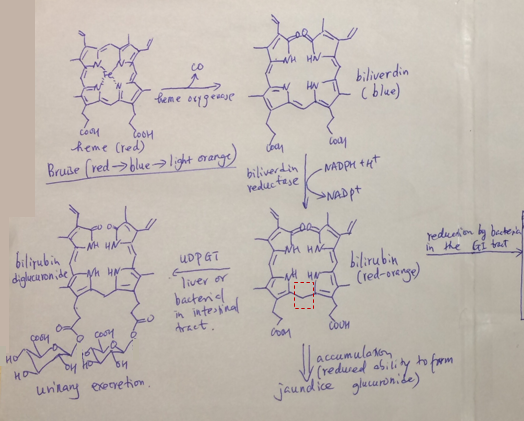 CO2 leaving with the help of heme oxygenase is what turns bruise blue created biliverdin→ bilirubin give it a light orange hue
CO2 leaving with the help of heme oxygenase is what turns bruise blue created biliverdin→ bilirubin give it a light orange hueif bilirubin is glucuronidated it is excreted through urine
if bilirubin is built up in person it gives a person jaundice
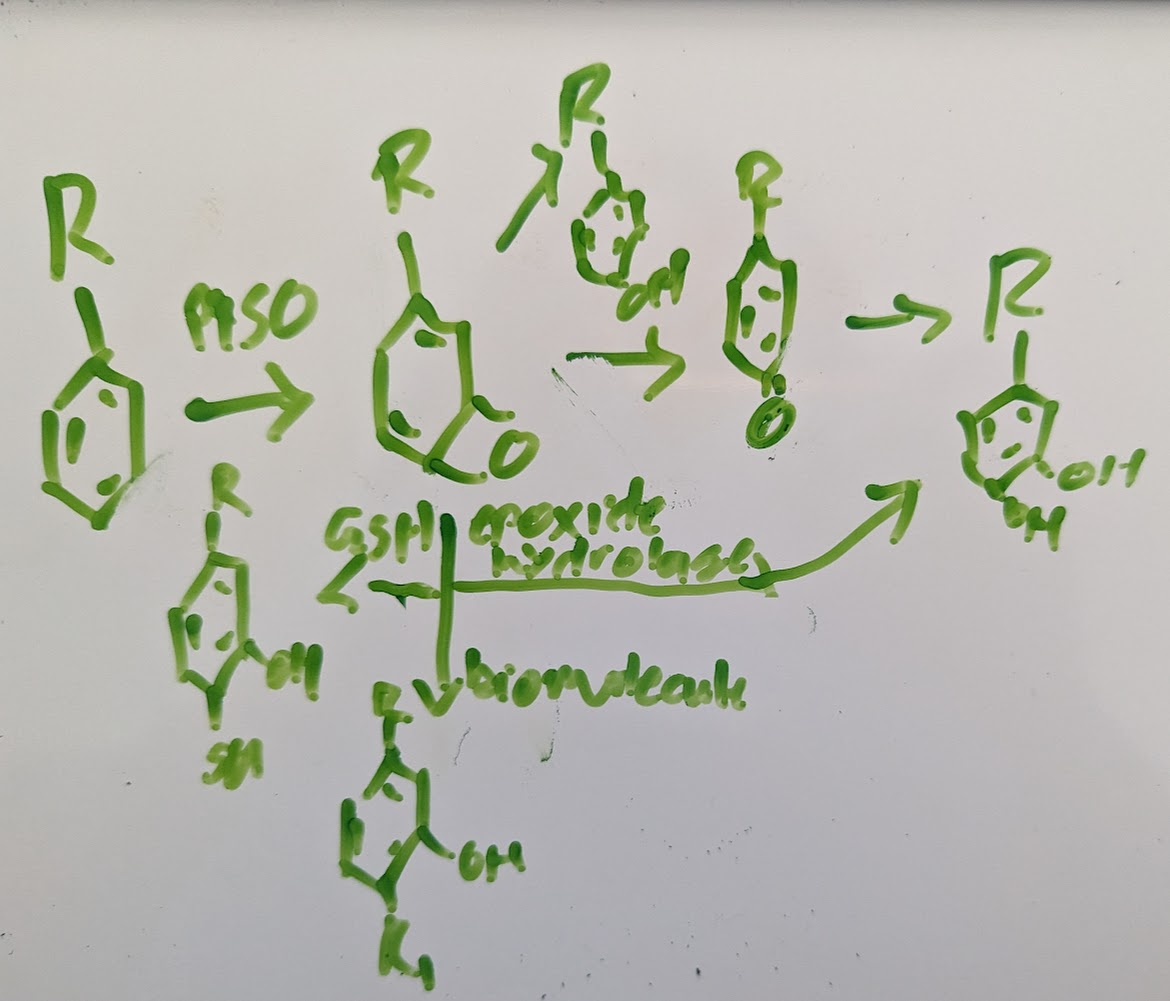
Polycyclic Aromatic hydrocarbons (PAH)
PAHs→ compounds with 2 or more Aromatic rings made with C and H
benzene is a carcinogen and toluene isn’t because toluene once metabolized by p450 can easily go through phase 2 metabolism but benzene becomes a super stable arene oxide which can lead to radical propagation and other issues
nomenclature systesm of PAH→ it’s a combo of trivial and IUPAC naming
find the highest priority group in the compound
number atoms
number edges (bonds between C on outer ring) with letters
example:
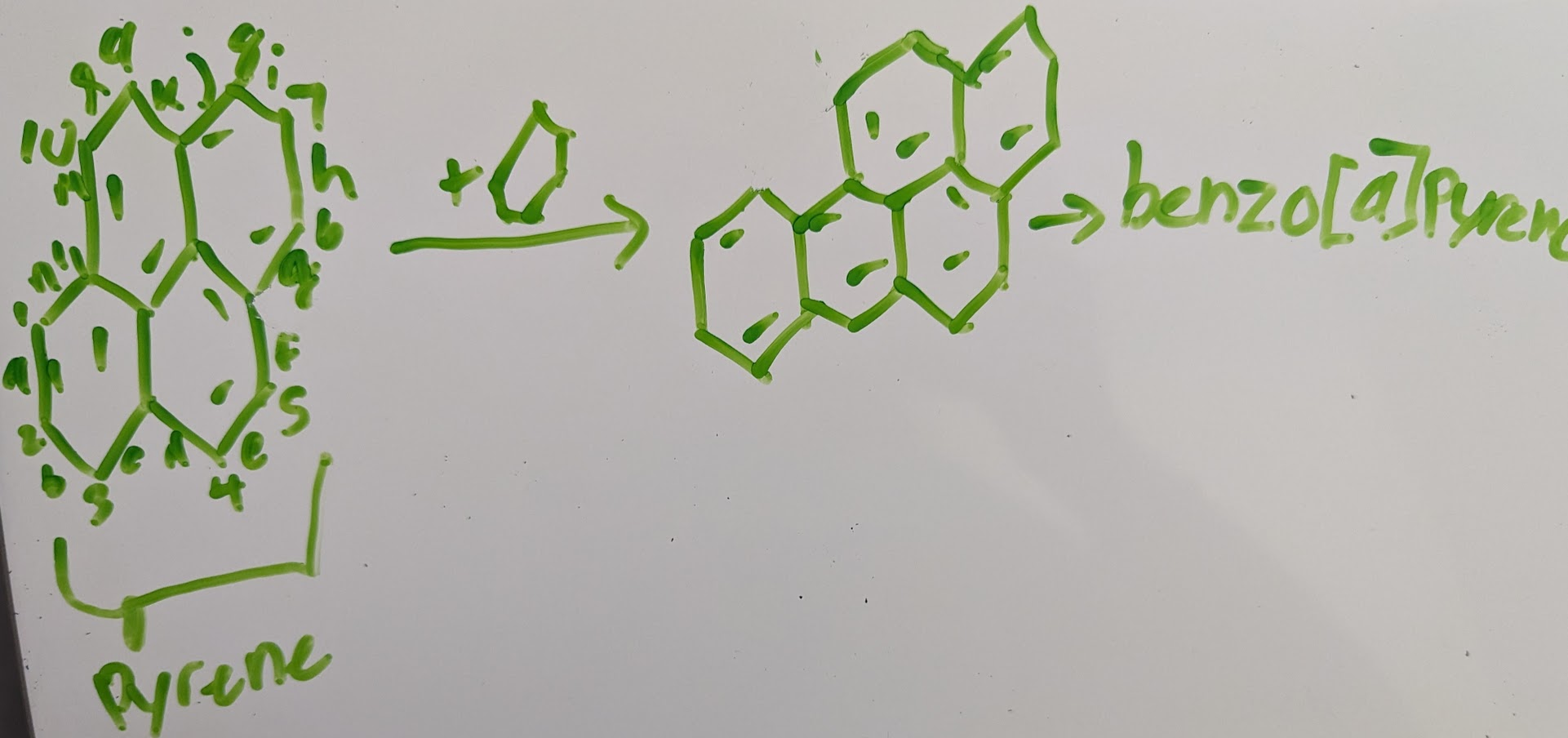
the more rings you add to cycle the more it will start to fold and twist so topography is developed to aid on how the molecule twist and can be used to predict where reactions happen
James and Elizabeth miller → found that most carcinogens are mutagenic after they are metabolized
mutagenicity depends on the extent of conversion to reactive electrophile
example of benzo[a]pyrene:
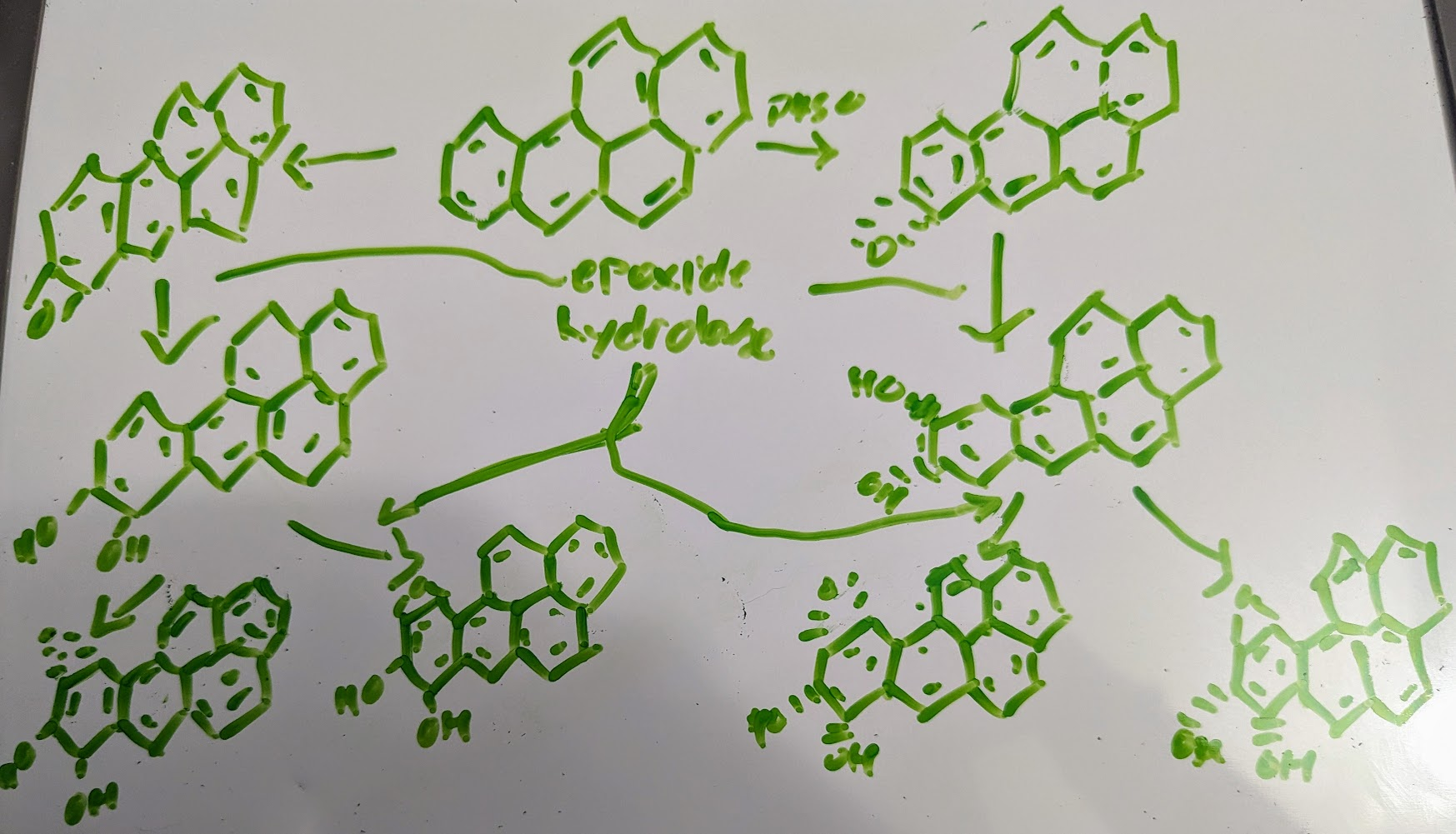 all of the last row are super reactive that you can’t isolate them
all of the last row are super reactive that you can’t isolate them