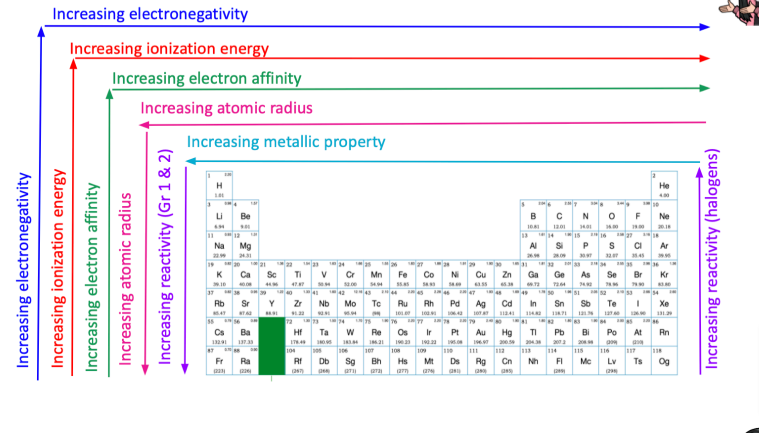
THE PERIODIC TRENDS
FEATURES OF THE PERIODIC TABLE
Period- horizontal arrangement (left and right)
Group- vertical arrangement (up and down)
Patterns are either increasing or decreasing
Definitions of features:
Atomic mass- mass of an atom (protons+neutrons), expressed in atomic mass unit (amu)
Atomic mass v atomic weight-
Atomic mass is the sum of the protons and neutrons present in an atom
Atomic weight is the average of all mass of all naturally occuring isotopes
Atomic radius- half-distance between two nuclei
Formula: radius=distance/2
Metallic property- characteristics attributed to metals (i.e: luster, conductivity)
Electron affinity- measure of the attraction an atom has for an additional electron
Ionization energy- amount of energy to remove an electron from an atom
Electronegativity- ability of an atom to attract elections
DIFFERENCE OF ELECTRON AFFINITY AND ELECTRONEGATIVITY:
Electron affinity: amount of energy released when an atom attracts an electron, a quantitative measurement, the ability of an atom to ACCEPT an electron
Electronegativity: ability to attract an electron TOWARDS itself through a chemical bond, a qualitative measurement
DIFFERENT PERIODIC TRENDS
Atomic mass- Increases left to right (by period) and increases downwards (by group)
WHY?
The periodic table is arranged in increasing atomic number (protons only). Atomic mass adds the protons and neutrons in an element. Most isotopes are close to or equal to the amount of protons, therefore by how the periodic table is arranged, atomic mass increases left to right (following the periods) and downwards.
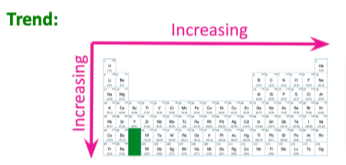
Atomic radius- Increases right to left, and increases downwards
WHY?
The number of shells increase as electrons increase. By going down a group, the electrons increase. Therefore, the shells increase as well, furthering the distance between two nuclei (of the same elements)
Across a period from right to left, the attractive force of between a proton and an electron decreases. This leads to the shells being more further apart, which increases the distance between two nuclei
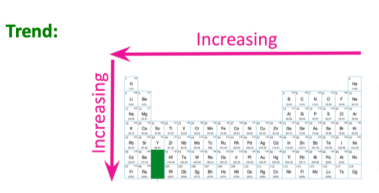
Metallic property- Increasing right to left, increasing downwards
WHY?
We must remember the first groups on the left are the metals (alkali and alkali earth), while all nonmetals are on the right. With this, we can say that the metallic property will definitely increase from right to left
Going down a group has more metal-like properties due to the increased weight and mass helping it have a more solid form, therefore, making it more metallic/ have more metal characteristics.
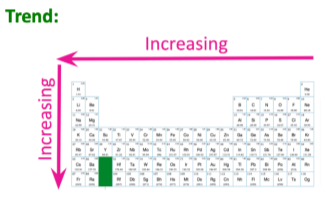
Electron affinity- Increasing left to right, increasing upwards
WHY?
Electron affinity increases from left to right because of the decrease in atomic radius. The less the radius, the more pull the nucleus has for other electrons outside of the orbitals.
Electron affinity increases upwards because going downwards means adding more shells. This increases the atomic radius, which makes it harder fro the nucleus to attract electrons.
Ionization energy- Increasing left to right, increasing upwards
WHY?
Ionization energy increases when atomic radii/radius decreases. This happens because when you go right to left, fewer electrons are attracted to the nucleus. This lack of attraction makes it easier for the electron to escape.
Ionization energy increases upwards because the less electrons in an orbit means the pull of the nucleus on the remaining electrons is stronger. This makes it harder for the electron to escape.
Electronegativity- Increases left to right, increases upwards
WHY?
Electronegativity increases upwards because less shells means more attraction between the electrons and the nucleus. This leads the nucleus to be able to attract more electrons.
Electronegativity increases left to right because if the valence shell of an atom is less than half full, it requires less energy to lose an electron than to gain one. Conversely, if the valence shell is more than half full, it is easier to pull an electron into the valence shell than to donate one.
Summary: